Amalgam Replacement Using Composite Core Buildup
A bulk-fill approach saves time while providing a long-lasting result
Apolinar Madrigal, DDS | Manisha Patel, DDS
Dental amalgam fillings have been utilized for more than 150 years in the mouths of millions of patients across the world.1 Although safety questions have been raised regarding its mercury content, this mix of metals has proven long-lasting and resistant to masticatory forces, while also being affordable to those from all socioeconomic classes. However, with improvements in the color and attributes of modern filling materials, the breakdown of amalgam due to bacterial infiltration presents an opportunity to provide an esthetic update that is free of controversy.2
The following case report details the steps taken to replace a failed Class II amalgam restoration with a composite core buildup and a zirconia crown. The dual-cure composite material used for the core buildup facilitated a bulk-fill, stackable delivery.3 This minimally invasive approach delivers a long-lasting restoration that is resistant to microleakage, and it does so in a fraction of the time required for a layered core buildup approach.
Case Report
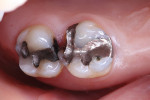
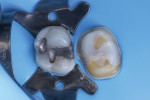
A 34-year-old female patient presented with recurrent decay on tooth No. 3 (Figure 1). It was determined that this Class II amalgam restoration was no longer viable in the patient's mouth. The patient consented to the proposed treatment plan, which included the removal of the existing amalgam, a core buildup utilizing composite material, and restoration with a zirconia crown.
First, an impression of the failed restoration was taken to aid in the fabrication of the temporary crown. A quadrant posterior tray (The Gripper®, DenMat) was lined with medium viscosity impression material (Splash!Max, DenMat). After impressing tooth No. 3, a rubber dam was placed, and the amalgam was removed. Deep decay was visible on the distal aspect of the tooth. In preparation for the composite core buildup, a bonding agent (Tenure 4G®, DenMat) was applied to the outer dentin, followed by the application of a bond enhancer (Tenure® S, DenMat) using sponges ((Dab-Eze®, DenMat).
Due to the extent of the decay, as well as its proximity to the pulp, a self-adhesive, base liner capping material (Bio-Cap®, DenMat) was applied to the distal surface of the tooth and light-cured for 30 seconds (Flashlite Magna® 4.0, DenMat). This was followed by selective enamel etching (Etch ‘N' Seal®, DenMat), which, when used in conjunction with the Tenure bonding system, forms a dentin hybrid layer that is necessary to strengthen the core buildup.
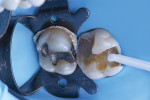
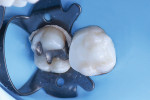
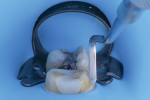
A matrix was placed on the distal aspect of the tooth, and the composite core buildup material (Core Paste® XP, DenMat) was applied via syringe using an intraoral tip (Figure 2). White was selected instead of the enamel color so that the material could be easily distinguished from natural tooth structure. This proves beneficial when shaping the composite for the final crown and aids in discernment should future dental work be required. After placement, the composite material was light-cured for 30 seconds. Figure 3 shows the completed core buildup ready for shaping.
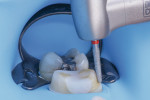
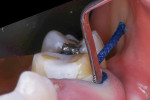
Next, the material was shaped utilizing a bur kit (Chamfer Preparation Logic Set, Kerr) (Figure 4). The composite core buildup material cuts like dentin, easing the task of shaping. Once all of the edges were smooth, a margin trimmer (DM2, Hartzell Instruments) was used to remove excess enamel and smooth the surface at the margin (Figure 5). This step ensures an exceptional marginal seal, which is paramount to the long-term success of the final restoration.
With the crown preparation complete, the buildup was checked once more to ensure that the edges were rounded and no undercuts were present (Figure 6). This assurance helps eliminate the occurrence of pressure points under the final crown. After the clear margins were verified, the tooth was prepared for the final lab impression.
A two-cord retraction technique was utilized prior to capturing the impression for the final restoration. A size 000 cord (Knit-Pack™, Premier) was packed into the sulcus around tooth No. 3, followed by a size 0 packing cord (Figure 7). After 5 minutes, the size 0 cord was removed, and with the size 000 cord remaining in the sulcus, a light body impression material (Splash!Max, DenMat) was extruded intraorally. A Gripper Impression Tray lined with medium body material was then impressed into the light body material. This impression technique, combined with the use of light and medium body impression materials, provides exceptional detail to the laboratory to aid in fabrication of the final restoration.
While the laboratory was fabricating the final zirconia crown, a temporary crown (Perfec
temp10®, DenMat) was placed and the amalgam on tooth No. 2 was removed and replaced with a light-cured composite (Nuance®, Den
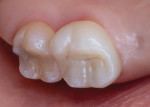
Mat). Later, the patient was recalled to the office for removal of the temporary crown and cementation of the final restoration. An adhesive (Tenure Uni-Bond®, DenMat) was applied to the crown preparation, the enamel was selectively etched (Etch ‘N' Seal®, DenMat), and the zirconia crown (LumiZir®, DenMat) was cemented utilizing a self-etching material (Infinity® SE, DenMat). The patient was happy with the final result (Figure 8), which should exhibit strength and lifelike esthetics for years to come.
About the Authors
Apolinar Madrigal, DDS
Private Practice
Turlock, California
Manisha Patel, DDS
Private Practice
Mooresville, North Carolina
References
1. US Food and Drug Administration. About Dental Amalgam Fillings. FDA Website. www.fda.gov/medicaldevices/productsandmedicalprocedures/dentalproducts/dentalamalgam/ucm171094.htm. Updated December 5, 2017. Accessed June 14, 2018.
2. Dodes JE. The amalgam controversy. An evidence-based analysis. J Am Dent Assoc. 2001;132(2):348-356.
3. Bortolotto T, Melian K, Krejci I. Effect of dual-cure composite resin as restorative material on marginal adaptation of class 2 restorations. Quintessence Int. 2013;
44(9):663-672.