Patterns of Use for an Enhanced Nociceptive Trigeminal Inhibitory Splint
Andrew Blumenfeld, MD; Steven D. Bender, DDS; Barry Glassman, DMD; Andres Pinto, DMD, MPH
Commentary by Howard E. Strassler, DMD
Professor and Director of Operative Dentistry
Department of Endodontics, Prosthodontics and Operative Dentistry
University of Maryland Dental School
Baltimore, Maryland
Abstract
To date, a survey of the factors associated with use of a nociceptive trigeminal inhibitory splint has not been conducted or published. An independent research firm was commissioned by National Dentex Corporation to conduct an online survey of dentists who had previously purchased such devices, commercially known as the Nociceptive Trigeminal Inhibition Tension Suppression System (NTI-tss). The goal of this survey was to assess reasons for NTI-tss use and adverse events related to NTI-tss use in a large group of NTI-tss providers. The survey was developed using a Delphi process to rank relevant items and was broadcast to a total group of 5,807 general dental practitioners and specialists in one day. Responses were analyzed with descriptive statistics (means, medians, percentiles, scatter plots, and frequency distributions). A total of 587 surveys (10.11%) were completed by providers who placed an average of 160 (SEM ± 13.8, 95% CI 133, 187, median of 60) devices. Of responders, 97% were general dentists (n = 567); 16% (n = 92) had more than 10 years of experience using the NTI-tss; 43% (n = 250) had more than 5 years of experience; 35% (n = 208) between 1 and 5 years; and 6% (n = 37) had less than 1 year. Fifty-six percent of responders (n = 322) used the device equally for headache and/or orofacial pain management and tooth/restoration protection, while 26% (n = 147) used it primarily for pain management. The remaining 18% used the device primarily for tooth protection or to treat other indications. This provider-based survey reported a high degree of success with the appliance for treatment of orofacial pain and favorable patient outcomes among current users of this appliance.
The grinding and clenching of teeth, commonly termed bruxism, can occur during sleep as well as during wakefulness.1 These parafunctional episodes are categorized as either idiopathic or iatrogenic and may be the result of a variety of causes, including medications, medical disorders (including Parkinson’s disease, tardive dyskinesia, myoclonus, tic, depression, and anxiety), and natural orofacial events (such as swallowing, somniloquy, and sighing).1 Awake bruxism is reported to be present in 20% of the adult population. (Table 1).2 The prevalence of sleep bruxism peaks in the 25–44 year age range and declines with aging. The incidence is 5.5% among 15–18 year olds, 9.2% among 19–24 year olds, 10.5% among 25–44 year olds, and 8.8% among 45–64 year olds.3
A survey of published research indicates that bruxism has been linked to a variety of conditions. Approximately 40% of patients suffering from bruxism complain about orofacial pain (OFP), masticatory myalgia,4 morning jaw rigidity,5 headache,6,7 and migraines.8,9 Patients known to experience sleep bruxism are four times more likely to report jaw pain and movement limitations than those who do not self-report bruxism.10,11
Mitrirattanakul et al12 cited OFP disorders, including temporomandibular disorders (TMD), to be highly prevalent and a significant health problem. In a 1989 survey of 45,000 households, Lipton et al13 reported that approximately 22% of the adult population experienced at least one of five types of orofacial pain during the preceding 6 months. Although dental pathology was the most common type of OFP, temporomandibular joint (TMJ) pain was experienced by 5.3% of adults, and face or cheek pain by 1.4% of adults.4,13 Manfredini et al14 discovered a significant association between TMD and bruxism in a study of 212 patients with TMD.
As many as 65% of patients with sleep bruxism suffer frequent headaches.11 In both migraine and tension-type headache, tenderness of the pericranial muscles is a common complaint in headache sufferers.15 In one study, patients with tension-type headaches were found to contract their temporalis muscles during sleep an average of 14 times more intensely than controls who were asymptomatic, indicating a potential cause of pericranial tenderness.16 Furthermore, it was found that the presence of pericranial muscle tenderness was evident upon palpation in many headache patients (migraine and tension-type headache) while it was not present in controls.17 That migraine is primarily caused by vascular phenomena is no longer tenable. In fact, migraine etiology is now considered to include a dysfunction of the trigeminal nerve.18
According to the World Health Organization, migraine headache is one of the top 20 most-disabling diseases.19,20 It has been estimated that the prevalence of migraine is 12% in the US general population with more women affected (18%) than men (6%).21,22 The 1999 American Migraine Study II23 estimated that up to 28 million Americans experience migraine. One study has shown the incidence of migraine among the population of western countries to be 10%–12%.24 The estimated cost of migraine in the United States is $11 billion, which includes hospitalization, medications, and lost productivity. Migraine sufferers have been found to have significantly higher healthcare costs than controls.20
It is estimated that $80 billion is spent annually in the United States related to chronic pain, with as much as 40% directed toward the diagnosis and treatment of OFP disorders.25,26 OFP can have a significant impact on a patient’s quality of life. Twenty percent of patients indicate that OFP interferes with their daily activities, and 10% of those individuals report a direct impact on their ability to work.27 In TMD patients, the impact was higher if their condition was caused by musculoskeletal issues rather than temporomandibular disc derangements.28 Wolf et al determined that chronic OFP leads to hopelessness, resignation, and lack of faith, factors which impact compliance and outcomes and should be considered during treatment and management of patients with these diagnoses.29
While no definitive approach to treating chronic orofacial pain, chronic headaches, or habitual bruxism has been determined, a variety of preventive measures, such as bio-behavioral therapy, pharmacotherapy, and dental splint therapy have been used successfully.1
A Nociceptive Trigeminal Inhibitory Splint
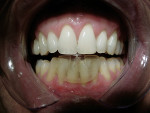
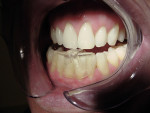
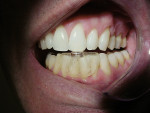
It has been demonstrated that oral dental splints decreased the number of sleep bruxism episodes and associated arousals by 40%, while reducing symptoms of jaw pain and dysfunction.30 In another study, contact on only the anterior teeth has been shown to significantly reduce biting forces.31 In 1998, the FDA approved the marketing of a specially designed oral appliance for the prevention of TMD caused by jaw clenching.8 Three years later, in 2001, the same device was approved for the prophylactic treatment of medically diagnosed migraine pain.32 The approved device, commercially known as the Nociceptive Trigeminal Inhibition Tension Suppression System (NTI-tss), covers only the anterior incisors (either maxillary or mandibular) and is prescribed for use during sleep in most cases. Unlike other anterior bite splints, this device includes an “enhanced discluding element,” an extension on the device that prevents any cuspid or posterior tooth contact with the device or the opposing dentition during all jaw movements (Figure 1, Figure 2, and Figure 3).
By eliminating any canine or posterior tooth occlusion and allowing for immediate and continuous incisal guidance on the “enhanced discluding element,” the device inhibits the jaw elevator muscles from reaching maximum clenching forces.8 In another short-term study, the device produced a strong inhibitory effect on EMG activity in jaw-closing muscles during sleep.33 Shankland reported at the conclusion of the pivotal FDA clinical trial that 82% of the participants using the NTI-tss device had an average 77% reduction in migraine events after 8 weeks of use, and 16% experienced an 85% to 100% reduction of headache events.8
Patterns of Use Survey
Dentists currently use the device for a variety of indications, including tooth wear, chronic headache, and OFP. Because no broad provider-based report of factors associated with the use of an enhanced anterior deprogrammer (the Nociceptive Trigeminal Inhibition Tension Supression System [NTI-tss]) device has ever been conducted or published, the principal investigator of this study determined the necessity of a comprehensive survey. Consequently, National Dentex Corporation, the manufacturer and a licensed distributor of the NTI-tss Plus, funded the project and commissioned an independent research firm to conduct an online survey of dentists currently using the NTI-tss device and therapeutic protocol. The objectives of the survey, Factors Related to Patterns of Use of the NTI, were to:
• identify patterns of use of an intraoral nociceptive trigeminal inhibition splint.
• determine future use of the device.
• identify whether the device is used prophylactically for pain control, or as a treatment modality.
• ascertain whether it is used for short- or long-term therapy.
• identify adverse issues or reasons for discontinued use.
• determine dentist success/satisfaction with the device.
• determine preferences for device fabrication techniques (direct intraoral fabrication or indirect laboratory fabrication).
Materials and Methods
The investigators initially suggested questions based on discussions of commonly expressed concerns by dentists about the use of an NTI-tss splint, including potential adverse effects. The final questions and suggested response choices for the online survey were developed using a structured Delphi process to rank relevant items, assessment of agreement, and subsequent revision of ranking among three of the authors, including two dentists who limit their practices to the treatment of headache and orofacial pain, and a neurologist. A draft version of the survey was circulated among the authors, who contributed additional comments. The refined version of the survey was submitted to the survey consultant (Mega Media, Inc.), which made suggested changes in the wording of questions, response options, and item branching, all designed to enhance comprehension. This draft was then posted online for the authors to view and comment. Final changes were suggested to Mega Media, Inc., and the survey (37 questions) was prepared for broadcast to an initial field test of 597 randomly selected e-mails of NTI providers obtained from two distributors, National Dentex Corporation and TMD Courses, Inc. Each company supplied e-mail addresses of current NTI device users who had purchased or ordered devices between February 1, 2009, and March 31, 2010.
Potential recipients were notified by e-mail that they would receive an invitation to participate in the survey. This notification was intended to encourage recipients to “white list” e-mail from Mega Media, Inc. and increase survey participation. Each participant was provided a link to the survey, with a unique and anonymous identifier. Participants were permitted to submit only one survey response. Fifty-five responses were received for a response rate of 9% for the field test.
The field test was considered successful (based on previous experience with online surveys, the expected response rate was 5%), and the remainder of the e-mail invitations was released in waves of 1,000 over the course of one day. A total of 6,333 e-mails were sent to general dental practitioners and specialists. Of those, 526 were undeliverable for a total receivership of 5,807. Participants were again provided a link to the survey with a unique, anonymous identifier and were permitted to submit only one survey per response. Responses were analyzed with descriptive statistics (means, medians, percentiles, histograms, and frequency distributions).
Results
Of the available 5,807 surveys, a total of 587 (10.11%) were completed by participants. A survey was considered completed if participants reached the last question and exited the survey. Providing answers to all 37 questions was not an inclusion criteria for considering the questionnaire completed. Providers placed an average of 160 (SEM ±13.8; 95% CI 133,187; median of 60) devices in their practices since starting use of the NTI-tss appliance. Time in practice of responders and years of experience with the device are presented in Table 2. Overall, 65.8% of responders had more than 20 years of clinical practice experience. The NTI-tss was used mostly by general dentists, with 58% of responders using the device for more than 5 years.
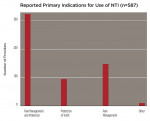
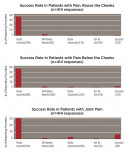
Fifty-six percent of responders (n = 322) used the device equally for headache and/or OFP management and tooth/restoration protection, while 26% (n = 147) used it primarily for pain management (Figure 4). Within these categories, 72% (n = 337) recommended using this particular appliance more than 80% of the time, whereas 87% (n = 409) used it for both short-term diagnosis/treatment and long-term therapy/prevention. When asked to categorize the anatomic pain profile of patients with NTI, 48% of treated patients had pain predominantly “above the cheeks” (migraine, tension, other primary headaches; temporalis pain); 34% were patients with pain “below the cheeks” (facial, tooth, pterygoid and masseter muscle, or neck pain); and 21% had pain predominately located in the TMJs. Four hundred and fourteen providers rated their perceived success with the use of the device. In pain-above-the-cheeks patients, practitioners rated their success with NTI treatment as 90% (n = 371). In patients with pain below the cheeks, 88% (n = 366) of cases were mostly successful, and in patients with joint pain, 74% (n = 306) of cases were mostly successful (Figure 5).
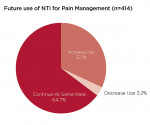
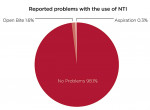
Four hundred and fourteen providers responded to a question about future use of the nociceptive trigeminal inhibition splint for pain management. They were queried about whether they planned to continue using the NTI for pain management at the same rate as in recent years, or planned to increase or decrease its use. Thirty-two percent (n = 133) stated they would increase their use for this indication (Figure 6). Approximately 97% stated they would maintain (n = 268) or increase (n = 133) their levels of NTI use. Of 511 responses to a question regarding fabrication techniques, 34% (n = 172) exclusively use direct fabrication, 31% (n = 160) use the laboratory-fabricated method, and 35% (n = 179) use both methods. The number of patients reported to have developed a clinical observation of an anterior open bite subsequent to use of the device was reported as 1.6% of 78,711 cases (Figure 7). Of patients reporting aspiration of the device (0.3%), no events were documented by radiograph or other means.
Discussion
This is the first published study of a large group of dentists using an anterior nociceptive trigeminal inhibitory splint in clinical practice. The survey sample of 587 was composed mostly of general dentists with 5 or more years’ experience using the device. The majority of respondents reported using the device for both the treatment of OFP and for the protection of teeth and restorations. Just over one quarter of the sample (26%) use the device primarily for pain management. A large number of respondents (87%) reported using the device for both short- and long-term treatment. Of those respondents (n = 36) using the device for short-term use only, just over half of them (n = 19) indicated they restricted treatment time to avoid possible changes in occlusion. One common concern often expressed by dentists about the use of a device that only covers anterior teeth is that an anterior open bite might result. Yet in this survey, the incidence of anterior open bite subsequent to the use of the device (based on 512 responses) was 1.6%. Previous reports on adverse events are limited by a small sample.34 This study, the largest to date, provides substantial provider-based evidence of the NTI’s clinical benefit and minimal occurrence of adverse events. Many dental providers base their overall opinion on the NTI splint on a perception of adverse events. This study helps to clarify the prevalence of these events. Further investigations of its efficacy and possibly related adverse events through randomized clinical trials with large patient populations and clearly defined criteria are needed.
Limitations of this study would include the potential for inconsistency in the diagnostic criteria used by the respondents for certain headache types as well as specific definitions of various TMDs. In addition, respondents were not queried as to other therapeutic modalities being used along with the NTI-tss device. In future studies, determinants of success and specific outcomes should also be well defined. Objective reporting of the splint’s effectiveness may provide additional insight. Inclusion of a provider sample that already was using the appliance may have contributed to data skewness. However, this would not have affected the results concerning serious adverse outcomes associated to the use of these appliances, such as aspiration.
Conclusions and Clinical Implications
This is the first report that describes patterns of use associated with the NTI-tss device in clinical practice. The survey sample was composed mostly of general dentists with 5 years or more experience using the device. Overall, the respondents reported a high degree of success with the appliance for treatment of OFP. In addition, more than half of the responders used the appliance for both pain control and protection of teeth and restorations. The rate of complications, including safety (risk of aspiration) and development of anterior open bite, was very low in this large cross-sectional sample. Overall, this provider-based survey reported a high level of satisfaction and favorable patient outcomes among current users of this appliance.
In summary, based on these findings, the NTI-tss device may be a viable treatment option for some patients with OFP and primary headache pain, either as a substitute for, or as adjunctive therapy to preventive medications and/or physical therapy modalities.
Commentary
It has been estimated that 45 million people in the United States exhibit signs and symptoms of bruxism when sleeping or awake. This number includes all age and gender groups. This grinding or clenching of the jaw can contribute to and is associated with premature tooth wear/attrition, restoration or tooth fracture, orofacial pain and temporomandibular disorders, migraines, and temporal headaches upon awakening. This parafunction has been categorized as being either idiopathic or iatrogenic due to medications, medical conditions, emotional stress, depression, anxiety, or even natural orofacial events. One clinical treatment that has successfully managed the signs and symptoms of bruxism is an occlusal treatment appliance. Which appliance design to use is sometimes difficult to determine.
The authors of this article present the etiology and problems associated with bruxism in a comprehensive, easy-to-understand way. One appliance that has been popular and successful in treating bruxism has been the Nociceptive Trigeminal Inhibition Tension Suppression System (NTI-tss). Using an online survey methodology of clinicians who are using the NTI-tss to treat symptomatic patients to assess the reasons for its use and adverse experiences related to its use, the authors were able to better describe how the device is used and the clinical success of the device for these uses. The majority (97%) of the clinicians were general dentists with more than 65% of the responders using the device for more than 5 years. Based upon the responses, the NTI-tss is a highly successful device for treating headache, orofacial pain management, restoration and tooth preservation. If you have patients with signs and symptoms of bruxism, use of the NTI-tss appliance to provide your patients with a successful treatment outcome should be considered.
Disclosures
Andrew Blumenfeld, MD, has acted as a consultant and received consulting fees from Keller Laboratories, Inc., and Therapeutic Solutions International, Inc.
Barry Glassman, DMD, is Director of Education and Professional Relations for Therapeutic Solutions International, Inc.
Andres Pinto, DMD, received an honorarium from National Dentex Corporation for statistical analysis.
References
1. Huynh N, Manzini C, Rompre PH, et al. Weighing the potential effectiveness of various treatments for sleep bruxism. J Canadian Dent Assoc. 2007;73(8):727-730b.
2. Lavigne GJ, Khoury S, Abe S, et al. Bruxism physiology and pathology: an overview for clinicians. J Oral Rehabil. 2008;35(7):476-494.
3. Maurice M, Ohayon K, Li K, et al. Risk factors for sleep bruxism in the general population. Chest. 2001;119:53-61.
4. Israel HA, Scrivani SJ. The interdisciplinary approach to oral, facial and head pain. J Am Dent Assoc. 2000;131(7):919-926.
5. Aloe F. Sleep bruxism neurobiology. Sleep Science. 2009;2(1):40-48.
6. Molina OF, dos Santos J Jr, Nelson SJ, et al. Prevalence of modalities of headaches and bruxism among patients with craniomandibular disorders. Cranio. 1997; 15(4):314-325.
7. Lavigne GJ, Kato T, Kolta A, et al. Neurobiological mechanisms involved in sleep bruxism. Crit Rev Oral Biol Med. 2003;14(1):
30-46.
8. Shankland WE 2nd. Migraine and tension-type headache reduction through pericranial muscular suppression: a preliminary report. Cranio. 2001;19(4):269-278.
9. Shevel E. Craniomandibular muscles, intraoral orthoses and migraine. Expert Rev Neurotherapeutics. 2005;5(3):371-377.
10. Goulet JP, Lund JP, Montplaisir J, et al. Daily clenching, nocturnal bruxism, and stress and their association with TMD symptoms. J Orofac Pain. 1993;7:89.
11. Dao TT, Lund JP, Lavigne GJ. Comparison of pain and quality of life in bruxers and patients with myofascial pain of the masticatory muscles. J Orofac Pain. 1994;8(4):350-356.
12. Mitrirattanakul S, Merrill RL. Headache impact in patients with orofacial pain. J Am Dent Assoc. 2006;137(9):1267-1274.
13. Lipton JA, Ship JA, Larach-Robinson D. Estimated prevalence and distribution of reported orofacial pain (OFP) in the United States. J Am Dent Assoc. 1993;124(10):115-121.
14. Manfredini D, Cantini E, Romagnoli M, et al. Prevalence of bruxism in patients with different research diagnostic criteria for temporomandibular disorders (RDC/TMD) diagnoses. Cranio. 2003;21(4):279-285.
15. Bendtson L. Central sensitization in tension-type headache—possible pathophysiological mechanisms. Cephalalgia. 2000;20
(5):486-508.
16. Clark GT, Sakai S, Merrill R, et al. Waking and sleeping temporalis EMG levels in tension-type headache patients. J Orofac Pain. 1997;11(4):298-306.
17. Louis I, Oleson J. Evaluation of pericranial tenderness and oral function in patients with common migraine, muscle contraction headache and “combination headache.” Pain. 1982;12:385-393.
18. Dodick DW, Silberstein SD. Migraine prevention. Pract Neurol. 2007;7:383-393.
19. Hu XH, Markson LE, Lipton RB, et al. Burden of migraine in the United States: disability and economic costs. Arch Intern Med. 1999;159(8):813-818.
20. Hawkins K, Wang S, Rupnow M. Direct cost burden among insured US employees with migraine. Headache. 2008;48(4):553-563.
21. Bigal ME, Lipton RB. The epidemiology, burden, and comorbidities of migraine. Neurol Clin. 2009;27(2):321-334.
22. Schwartz BS, Stewart WF, Lipton RB. Lost workdays and decreased work effectiveness associated with headache in the workplace. J Occup Environ Med. 1997;39(4):320-327.
23. Lipton RB, Stewart WF, Diamond S, et al. Prevalence and burden of migraine in the United States: Data from the American Migraine Study II. Headache. 2001;41(7):646-657.
24. Breslau N, Rasmussen BK. The impact of migraine: epidemiology, risk factors, and co-morbidities. Neurology. 2001;56(6 Suppl 1):S4-S12.
25. Fricton JR, Schiffman E. Epidemiology of temporomandibular disorders. In: Fricton JR, Dubner R, eds. Orofacial pain and Temporomandibular Disorders. New York: Raven Press; 1995:1-14.
26. Schiffman E, Fricton JR. Epidemiology of TMJ and craniofacial pain: an unrecognized societal problem. In: Fricton JR, Kroening RJ, Hathaway KM, eds. TMJ and Craniofacial Pain: Diagnosis and Management. St. Louis and Tokyo: Ishiyaku EuroAmerica Inc.; 1988:1-10.
27. Wong MC, McMillan AS, Zheng J, et al. The consequences of orofacial pain symptoms: a population-based study in Hong Kong. Community Dent Oral Epidemiol. 2008;36
(5):417-424.
28. John MT, Reissmann DR, Schierz O, et al. No significant retest effects in oral health-related quality of life assessment using the Oral Health Impact Profile. Acta Odontol Scand. 2008;66(3):135-138.
29. Wolf E, Birgerstam P, Nilner M, et al. Nonspecific chronic orofacial pain: studying patient experiences and perspectives with a qualitative approach. J Orofac Pain. 2008;22(4):349-358.
30. Dube C, Rompre PH, Manzini C, et al. Quantitative polygraphic controlled study on efficacy and safety of oral splint devices in tooth-grinding subjects. J Dent Res. 2004;
83(5):398-403.
31. Hattori Y, Satoh C, Seki S, et al. Occlusal and TMJ Loads in subjects with experimentally shortened dental arches. J Dent Res. 2003;82(7):532-536.
32. Shankland WE. A method of preventing migraine and tension headaches. Compend Contin Educ Dent. 2002;23(2):105-108.
33. Baad-Hansen L, Jadidi F, Castrillon E, et al. Effect of a nociceptive trigeminal inhibitory splint on electromyographic activity in jaw closing muscles during sleep. Oral Rehabil. 2007;34(2):105-111.
34. Magnusson T, Adiels, AM, Nilsson HL, et al. Treatment effect on signs and symptoms of temporomandibular disorders—comparison between stabilization splint and a new type of splint (NTI). A pilot study. Swed Dent J. 2004;28(1):11-20.
About the Authors
Andrew Blumenfeld, MD
Director of The Headache Center of Southern California
Encinitas, California.
Steven D. Bender, DDS
Director of the North Texas Center for Head, Face & TMJ Pain
Plano, Texas.
Barry Glassman, DMD
Director of the Allentown Pain Center
Allentown, Pennsylvania.
Andres Pinto, DMD, MPH
Director of the Oral Medicine Center
Member of the Center for Clinical Epidemiology and Biostatistics
University of Pennsylvania School of Dental Medicine
Philadelphia, Pennsylvania.