Postextraction Hemostasis for Medically Compromised Patients
Alloplastic bone graft substitute reduces pain and risk of bleeding events
Robert J. Abbiati, DDS
As the techniques employed for traditional bone grafting have improved, the use of cortical and cancellous freeze-dried bone allograft of various particle sizes has become the standard of care. Although effective techniques include the use of freeze-dried bone allograft with platelet-rich plasma (PRP) and the use of collagen membranes or decalcified bone matrix that is cut into thin membranes (ie, flexo-membranes), unfortunately, these techniques are not always effective when a patient presents with a significant infection or is medically compromised. When an emergency patient presents to a dentist's office with a large infection, packing an infected extraction site with cortical and cancellous bone particles is ill advised because the infection normally breaks down the bone grafting material, leading to a poor prognosis. Furthermore, medically compromised patients who suffer from cardiac issues compound this problem with the use of blood thinners such as warfarin, clopidogrel, or apixaban. Dental practices that focus on patients aged 65 and older deal with blood thinner usage, suboptimal patient systemic health, and significant immune system deficiencies on a regular basis. In the presence of systemic health problems, PRP treatment and cortical and cancellous bone grafting techniques are less effective and fail to mitigate the patient's immediately present infection, pain, and ultimate bleeding risk.
For the treatment of medically compromised patients who suffer from failing teeth and active infections where traditional methods of bone grafting or collagen plugs cannot adequately and effectively stop profuse bleeding, the use of alloplastic bone grafting technology following extraction can not only provide hemostasis for postoperative bleeding but also reduce patient pain, inflammation, complications, and healing times by sealing exposed bone and preventing tissue invagination into the open wound.
This article presents a technique involving the immediate packing of a synthetic grafting material into an actively infected or otherwise compromised extraction site, complemented by the use of a CO2 laser for severe bleeding risks and the use of dexamethasone sodium phosphate for the control of inflammation and pain.1 The aim of this technique is to help medically compromised patients achieve wound closure with less pain and without experiencing a major bleeding event, not to achieve significant, long-term bone formation among this patient population.
Clinical Technique
In this technique, the teeth are extracted using luxators in order to separate the bone from the tooth while minimizing damage to the buccal plate, adjacent teeth, and lingual bone. The luxator is run down the periodontal ligament to create space (rarely ever is an elevator required) until the tooth exhibits visible movement and can be extracted using forceps. After the tooth is extracted, the infection is cleaned by curette and rinsed with saline, and an x-ray scan is taken to ensure that no root fragments or other debris were left behind. Surgical suction is utilized to remove any excess blood, and then an alloplastic bone graft substitute (GUIDOR® easy-graft® CLASSIC Bone Grafting System, Sunstar) is placed.2 It is recommended to utilize the large system (ie, 500 to 1,000 µm granules in a 0.4 mL volume) to completely fill the cavity.3,4 This synthetic bone graft comes in a two-piece kit where the synthetic polymer coated β-tricalcium phosphate (β-TCP) granules are combined with the BioLinker® polymer activator in the provided delivery syringe.3,4 Once combined, excess BioLinker should first be expelled before placing the material into the extraction socket. While the product is dispensed into the extraction socket, any remaining excess BioLinker is removed via adjacent surgical suction, and then the practitioner can mold the material to the socket before it hardens.3 After the hardening process is completed, which only takes about 1 minute, the osteoconductive bone grafting material almost completely prevents bleeding (minor oozing may occur), even in high-risk patients.5 The BioLinker seals the bone, which helps minimize the patient's pain, and pain control is further enhanced by an injection of 4 mg/mL of dexamethasone sodium phosphate that is given immediately following the extractions. After the grafting material is molded into the socket, wet gauze is placed in the mouth and removed after 30 minutes, followed by the placement of more wet gauze for another 30 minutes. No additional hemostatic agents are needed unless the margins of the wound are still bleeding significantly. At this time, the margins of the wound are treated with a 9.3 µm CO2 laser (Solea® Version 3, Convergent Dental), which effectively ablates the tissue and eliminates any additional bleeding risk.1 Patients are instructed to eat ice cream immediately after treatment, followed by a period of soft foods to avoid disrupting the synthetic bone graft, and prescribed 1 week of antibiotics as well as pain medication on an as needed basis.
Case Report 1
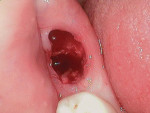
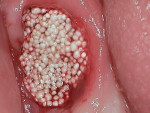
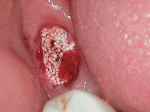
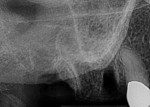
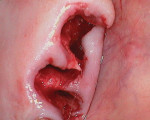
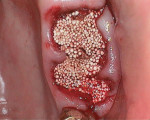
An elderly male patient presented with pain and infection associated with his molars, root tips, and failing root canals as well as the presence of gross decay. He was taking blood thinners daily and had an increased risk of cardiac issues and bleeding events, a reduced capacity for healing due to systemic health issues and age, and a high bacterial load from years of poor oral hygiene and dental neglect. Following extraction (Figure 1), hemostasis was readily achieved using the alloplastic bone graft substitute (Figure 2). One day after treatment, the extraction site was stable, and the patient reported no postoperative pain (Figure 3).
Case Report 2
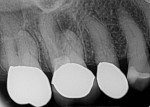
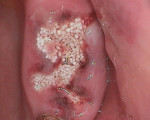
An elderly female patient presented with a large periapical lesion on teeth Nos. 2 and 3 (Figure 4). She refused to undergo endodontic treatment and requested that her teeth be extracted instead (Figure 5 and Figure 6). In addition, she had several areas of failing dentistry involving old crowns and fillings but did not want to restore these areas and again requested extractions. Despite the large periapical abscess and heavy bleeding that accompanied the extractions with an attached globular mass, hemostasis was readily achieved using the alloplastic bone graft substitute (Figure 7), and 1 day later, the patient reported that she hadn't experienced any significant bleeding events and had no postoperative pain (Figure 8).
Conclusions
Employing an alloplastic bone graft substitute is an effective treatment modality when treating patients who are at higher risk for bleeding, suffer from an active infection, or are medically compromised with a reduced capacity to heal. When coupled with polymer-coated granules, the BioLinker contained in this system is effective in shortening healing times, reducing pain, providing hemostasis control, and encouraging osteoconductivity and bone formation in an infected extraction site.2 Patients also benefit greatly by having the teeth extracted using luxators, which does not elevate or break any of the surrounding bone; receiving an injection of 4 mg/mL of dexamethasone sodium phosphate; and having the bone graft substitute placed at the tissue level, which prevents tissue invagination.2,3 In combination, these techniques prevent bleeding events from developing, reduce pain, encourage bone formation in infected extraction sites, and allow systemically compromised patients to be treated both timely and at a reduced risk of complications-outcomes that are not provided by traditional cortical and cancellous bone grafting techniques.2,5
About the Author
Robert J. Abbiati, DDS
Diplomate
International Dental Implant Association
Private Practice
Marco Island, Florida
References
1. Fantarella D, Kotlow L. The 9.3-µm CO2 dental laser: technical development and early clinical experiences. J Laser Dent. 2014;22(1):10-27.
2. Leventis MD, Fairbairn P, Horowitz RA. Extraction site preservation using an in-situ hardening alloplastic bone graft substitute. Compend Contin Educ Dent. 2014;35(4 Suppl):11-13.
3. Schmidlin PR, Nicholls F, Kruse A, et al. Evaluation of moldable, in situ hardening calcium phosphate bone graft substitutes. Clin Oral Implants Res. 2013;24(2):149-157.
4. Neumeyer S, Neumeyer-Wühr S. The use of polylactide-coated ß-TCP: closure of oroantral communications. Implants. 2010;(4):32-36.
5. Saito H, Shiau HJ, Prasad H, et al. Evaluation of a poly(lactic-co-glycolic) acid-coated ß-tricalcium phosphate bone substitute for alveolar ridge preservation: case series. Clin Adv Periodontics. 2017;7(4):190-194.
For more information, contact:
Sunstar Americas, Inc.
us-professional.gumbrand.com/guidor
877-484-3671