Improved Tissue Management With Less Steps
Hemostatic gel efficiently achieves retraction for conventional and digital impressions
Srid Janyavula, DMD, MS | Peet van der Vyver, BChD, MSc, PhD
Impression quality continues to present the dental laboratory with significant challenges. According to the results of a study that assessed the quality of impressions for fixed partial dentures that were sent to commercial laboratories, 89% had one or more noticeable errors.1 Voids and tears at the finish line are cited as the most common errors. The use of hydrophilic, high-tear-strength impression materials and proper tissue management can help to effectively improve outcomes.
Prior to taking an impression, good tissue management protocols are required for full- or partial-coverage crowns, and the gingival tissues need to be adequately displaced to expose the finish lines on prepared teeth.2 The tissues must be safely managed while a sulcular width of 0.15 to 0.20 mm is created.3 The primary factors in defective impression margins are related to the inadequacy of the gingival displacement technique used3 and poor fluid control prior to taking the impression. There are several mechanical, chemical, and surgical methods employed by clinicians to retract the gingival tissue and control fluids, but the most common technique involves the use of a physical retraction device (eg, retraction cord, paste) coupled with a hemostatic agent.
Today, several hemostatic agents are used by clinicians, including epinephrine, ferric sulfate solutions, and aluminum chloride. All of these materials have distinct mechanisms of action. Epinephrine acts by vasoconstriction, but it can result in adverse cardiac effects and is generally contraindicated for hemostatic applications. Ferric sulfate solution forms an iron complex with blood, causing hemostasis; however, the solution is acidic, so it can cause postoperative sensitivity and temporarily stain soft tissues and teeth.4
The most commonly used active ingredient for hemostasis in dentistry is aluminum chloride, which is the main component in many retraction liquids, gels, and pastes. Aluminum chloride solutions (ie, up to 25% w/w) are astringents that function by causing minor shrinking of the soft tissue, thereby limiting the space for capillaries and restricting blood flow. When used in combination with retraction cord, aluminum chloride can achieve adequate hemostasis and gingival displacement. However, care must be taken to completely rinse the material from the tissue because aluminum chloride interferes with the setting mechanism of impression materials.
The typical impression taking technique begins with the selection of a retraction cord of adequate size, which is then soaked in a liquid aluminum chloride solution. The cord containing the aluminum chloride solution is manually packed into the sulcus and left for several minutes until adequate tissue retraction is achieved. The cord is then removed, and the impression is taken or the scan is captured.
Clinicians will often encounter additional bleeding, termed secondary bleeding, after initial hemostasis and cord removal. The causes of secondary bleeding are mechanical and physiological. Simply soaking the cord can leave its surfaces relatively dry and textured, and the astringent agent tends to drive fluid from the tissue, causing dryness relative to its normal state. These two factors set up a clinical situation wherein the tissue can be traumatized upon cord removal and result in secondary bleeding, requiring the process to be repeated.
Clinical Technique
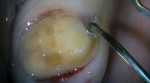
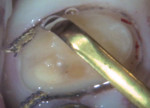
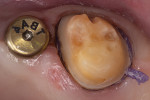
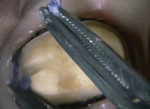
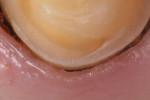
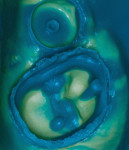
This situation can be addressed by using a 25% aluminum chloride hemostatic gel (Hemoban® Gel, Dentsply Sirona). Because the material is a gel, the viscosity allows it to stay where it is placed in the sulcus (Figure 1). In addition, it facilitates the immediate placement of a dry retraction cord because it lubricates the cord and the sulcus simultaneously. The gel completely coats the cord as it is placed, reducing friction and tissue trauma (Figure 2). If the clinician wishes to employ a double cord technique, a second coat of gel is applied to the first cord, and then the second cord is placed immediately over it (Figure 3). Effective hemostasis and retraction are achieved in 5 minutes, then following cord removal, an air-water spray is used to rinse the excess gel from both the preparation and the tissue (Figure 4). This technique significantly reduces secondary bleeding because the properties of the gel provide more lubrication when compared with those of soaked retraction cord. This method of placing the 25% aluminum chloride hemostatic gel into the sulcus and packing dry cord through the gel can reliably enhance marginal visibility (Figure 5), especially with conservative preparation designs. The selection of impression material has a tremendous impact on clinical results. High tear strength and hydrophilicity are two critically important material characteristics that are required to reduce the incidence of voids and tears at the margin. When a high-strength impression material is used in combination with this technique, the clinician can predictably capture every detail quickly and reliably (Figure 6).
About the Authors
Srid Janyavula, DMD, MS
Private Practice
Philadelphia, Pennsylvania
Peet van der Vyver, BChD, MSc, PhD
University of Pretoria
South Africa
Private Practice
Sandton, South Africa
References
1. Samet N, Shohat M, Livny A, et al. A clinical evaluation of fixed partial denture impressions. J Prosthet Dent. 2005;94(2):112-117.
2. Aimjirakul P, Masuda T, Takahashi H, et al. Gingival sulcus simulation model for evaluating the penetration characteristics of elastomeric impression materials. Int J Prosthodont. 2003;16(4):385-389.
3. Donovan TE, Chee WW. Current concepts in gingival displacement. Dent Clin North Am. 2004;48(2):
433-444.
4. Conrad HJ, Holtan JR. Internalized discoloration of dentin under porcelain crowns: a clinical report. J Prosthet Dent. 2009;101(3):153-157.
For more information, contact:
Dentsply Sirona Restorative
singleunitrestorations.com
844-848-0137