Re-Treatment of Iatrogenic Implant Failure
A simplified approach for guided bone regeneration and soft-tissue augmentation
Giorgio Tabanella, DDS, MS
For more than 3 decades, guided bone regeneration (GBR) was a complex technique that only expert surgeons could execute predictably.1 It was further complicated by the risk of wound opening and membrane exposure associated with the titanium-reinforced membranes and titanium mesh used to support the GBR material.2-6 However, implant dentistry has advanced and now offers new and more predictable forms of therapy with minimally invasive protocols for tissue regeneration. Today, the use of resorbable collagen membranes has reduced the risk of wound opening,7 and they are better tolerated by the healing tissue than the previously used titanium membranes. Resorbable collagen membranes also provide additional benefit to clinicians and patients because they do not require a removal surgery. Accordingly, resorbable collagen membranes have made the performance of GBR procedures more practical for a larger group of general practitioners.
Such simplified protocols using resorbable collagen membranes may also be applied to complex clinical cases in which trauma to teeth, periodontitis, or iatrogenic damage result in a ridge defect that precludes straightforward implant therapy. With biomimetic restorations being the ultimate goal of the clinician, esthetic challenges may be even more pronounced when tissue reconstruction needs to be performed in areas adjacent to teeth with a reduced periodontium, because the impaired blood flow, as a result of the disease, can reduce the biologic potential of GBR.8 This article presents a simplified approach for GBR and soft-tissue augmentation in a complex clinical case.
Case Report
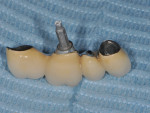
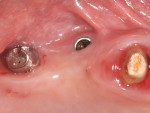
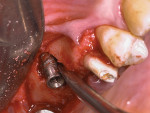
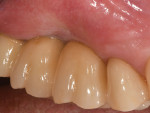
A 52-year-old, female patient with an insignificant medical history presented to the clinic with a detached implant-supported prosthesis. Visual inspection of the prosthesis and site indicated that the restoration had been cement-retained to a titanium abutment (Figure 1 and Figure 2). Clinical examination also revealed the presence of a misplaced dental implant with a cemented post, a high frenum attachment, poor mucosal quality due to a lack of keratinized mucosa, and bleeding on probing at the adjacent dentition due to biologic width violation (Figure 2). Furthermore, the whole prosthesis had also been cemented to a natural tooth (ie, the maxillary second premolar). The patient was unable to provide any information on what implants she had received.
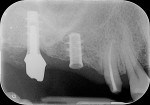
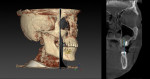
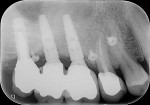
The periapical radiograph showed that the misplaced implant was in an incorrect mesiodistal and buccolingual position (Figure 3), which had required the prosthesis to be fabricated with an extra artificial premolar to fill the restorative space between the distal implant and the natural tooth. A 3-dimensional analysis using the sagittal cuts as well as a 3D reconstruction (NobelClinician, Nobel Biocare) revealed peri-implant buccal bone loss associated with the implant platform (Figure 4).
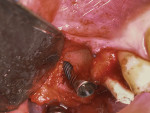
Osseous resective surgery, implant remo-val, sinus elevation, implant placement, and simultaneous GBR to correct the buccal plate loss were performed during one surgical phase. Following the administration of local anesthesia (ie, articaine 4%, 1:100.000), a full-thickness flap was elevated to expose the failing implant. A pronounced dehiscence on the buccal aspect with exposed implant threads was clearly visible. This was most likely related to incorrect implant placement (ie, too buccal) as well as subsequent buccal plate loss. Following implant removal using a retrieval kit (Figure 5), sinus elevation was achieved with a vertical approach, compressing the bone through the osteotomy site with the use of osteotomes. Spongious collagen was inserted into the sinus, and then demineralized bovine bone was packed through the osteotomy.
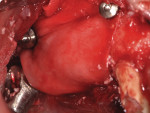
Subsequently, two endosseous dental implants (NobelReplace Conical Connection 4.3 x 13 mm, Nobel Biocare) were placed using a prosthetically-driven protocol. A surgical template was fabricated based on a wax-up, and the desired emergence profile guided the depth of insertion. For the GBR procedure to correct for buccal plate loss, autologous bone was harvested with a micro elevator (Tabanella 2, Hu-Friedy) from the non-supporting buttressing bone and then packed on the implant surface (Figure 6). Next, bovine bone mineral matrix (creos xenogain, Nobel Biocare) was layered around the autologous bone. The goal of performing this layering technique was to obtain greater bone-to-implant contact. Finally, a resorbable non-cross-linked collagen membrane (creos xenoprotect, Nobel Biocare) was fixed over the graft with titanium pins (Master-Pin-Control, Meisinger) (Figure 7). The pins were utilized to fixate the membrane, create a self-contained defect, and prevent the displacement of the graft particles as well as soft-tissue invasion.
After the membrane was fixated, pressure was applied to it in order to evaluate bone chip mobility. The flap was also moved mesiodistally to assess the stability of the graft. A provisional restoration was immediately delivered to improve the patient's comfort and facilitate protection of the bone graft for the duration of the healing time. The provisional connected the distal implant placed in the position of tooth No. 1 to the anterior premolar.
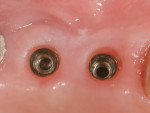
Following 6 months of healing, the implant was uncovered to evaluate the osteointegration and increase the thickness of the peri-implant mucosa with a minimally invasive approach.9 This second phase of surgery allowed the frenum and the mucogingival junction, which had been previously moved coronally at the time of GBR, to be shifted apically. After a final impression was taken with a closed tray, CAD/CAM titanium abutments (NobelProcera, Nobel Biocare) were connected to the implant platforms, and an all-ceramic, 3-crown implant bridge (NobelProcera, Nobel Biocare) was definitively cemented. The implant previously placed in the No. 1 position was left in situ because it was undamaged and too close to the greater palatine artery. In addition, a single, all-ceramic restoration (NobelProcera, Nobel Biocare) was cemented onto the natural premolar, thus generating an overall stable and esthetic result (Figure 8 through Figure 10). Prior to cementation, retraction cord was inserted inside the peri-implant sulcus to avoid excess cement around the implant platforms.10 The same polycarboxylate cement (Durelon™, 3M ESPE) was used for all parts of the restoration. Because the titanium pins were neither visible nor associated with any other esthetic concerns, they were left in place to avoid additional surgery.
Discussion
The key to successful implant dentistry is the clinician's ability to provide adequate volume and quality of bone and mucosa at the recipient site. This requires a precise diagnostic evaluation11,12 based on cone-beam computed tomography (CBCT), 3D reconstruction, and surgical knowledge. Once the volume of the deficiency has been properly registered, the most appropriate bone grafting technique can be selected. The objective is to utilize the least invasive13and most conservative techniques possible to achieve the desired results. Methodology, including the periodontal aspect of treating these advanced clinical cases; preservation strategies for soft- and hard-tissue structures; and the ability to provide function, esthetics, and long-lasting results14 are all dependent on accurate diagnosis. The combination of periodontal and implant protocols to alter the soft-tissue profile around natural teeth and implants will be an important component to the future of "biomimetics in dentistry."
During the last 3 decades, regenerative techniques have improved to the point that resorbable collagen membranes can be utilized in all types of bone atrophies (with the exception of esthetic cases in need of vertical ridge augmentation). The type of GBR approach proposed in this article uses titanium pins to fixate a tear-resistant membrane, thus stabilizing the underlying bone particles. The advantage to using pins as a fixation system rather than periosteal sutures is mainly related to the reduced risk of graft particles shifting during the healing phase, which facilitates a more predictable and desirable augmentation of bone volume. For horizontal augmentation, this type of approach is very predictable, reducing the risk of wound opening associated with the use of titanium mesh or titanium-reinforced membranes.
Dental implants have also evolved dramatically, particularly during the last decade, and so have expectations regarding function and esthetics. In addition, the augmentation of soft tissue around dental implants to enhance the architecture of inter-implant papilla has emerged as an area of considerable concern and focus, changing clinicians' ambitions regarding the reestablishment of a normal scalloped contour. A minimally invasive approach to augment the peri-implant mucosa using a buccal pedicle flap can also help to increase the thickness of the keratinized mucosa.9 This facilitates a mucosal seal around the neck of each implant without the need for several connective tissue grafts. Performing a buccal pedicle flap also helps to increase the height of the mucosa. This improves the esthetics on the buccal aspect while leaving the soft tissue level more apical on the lingual aspect to ensure that the cleaning procedure is effective.
Conclusion
This case report demonstrates how simplified GBR protocols using resorbable non-cross-linked collagen membranes can be applied to treat even complex cases, including those characterized by a misplaced, ailing implant and a large buccal dehiscence. The main advantage to using a non-cross-linked collagen membrane is that it causes less inflammation when compared with cross-linked membranes. In addition, a non-cross-linked collagen membrane is rich in elastin fibers; therefore, it can be gently stretched before fixation, when needed. The use of resorbable non-cross-linked collagen membranes should be considered the standard of care for horizontal ridge augmentation, particularly given that these membranes have been previously shown to reduce the risk of wound opening during the healing phase7 and eliminate the need for further surgical intervention.
About the Author
Giorgio Tabanella, DDS, MS
Private Practice
Rome, Italy
Founder
Oral Reconstruction and Education Center
Rome, Italy
References
1. Lim G, Lin GH, Monje A, et al. Wound healing complications following guided bone regeneration for ridge augmentation: a systematic review and meta-analysis. Int J Oral Maxillofac Implants. 2018;33(1):41-50.
2. Machtei EE. The effect of membrane exposure on the outcome of regenerative procedures in humans: a meta-analysis. J Periodontol. 2001;72(4):512-516.
3. Mellonig JT, Triplett RG. Guided tissue regeneration and endosseous dental implants. Int J Periodontics Restorative Dent. 1993;13(2):108-119.
4. Shanaman RH. A retrospective study of 237 sites treated consecutively with guided tissue regeneration. Int J Periodontics Restorative Dent. 1994;14(4):292-301.
5. Wang HL, Carroll MJ. Guided bone regeneration using bone grafts and collagen membranes. Quintessence Int. 2001;32(7):504-515.
6. Hitti RA, Kerns DG. Guided bone regeneration in the oral cavity: a review. Open Pathol J. 2011;5:33-45.
7. Charulatha V, Rajaram A. Influence of different crosslinking treatments on the physical properties of collagen membranes. Biomaterials. 2003;24(5):759-767.
8. Funato A, Salama H, Ishikawa T, et al. Timing, positioning, and sequential staging in esthetic implant therapy: a four-dimensional perspective. Int J Periodontics Restorative Dent. 2007;27(4):313-323.
9. Tabanella G. The "buccal pedicle flap technique" for peri-implant soft tissue boosting. Int J Esthet Dent. In press.
10. Ferreira CF, Shafter MA, Jain V, et al. Evaluation of effectiveness of cement removal from implant-retained crowns using a proposed circular crisscross flossing technique. J Oral Implantol. 2018 Feb 13. doi: 10.1563/aaid-joi-D-17-00265.
11. Tripodakis AP, Gousias H, Mastoris M, et al. Five-year volumetric evaluation of periodontally compromised sites restored by immediate implant restorations. Int J Periodontics Restorative Dent. 2016;36
(5):645-653.
12. Steigmann M, Monje A, Chan HL, et al. Emergence profile design based on implant position in the esthetic zone. Int J Periodontics Restorative Dent. 2014;34 (4):559-563.
13. Gargiulo AW, Wentz FM, Orban B. Dimensions and relations of the dentogingival junction in humans. J Periodontol. 1961;32(3):261-267.
14. Tabanella G, Nowzari H, Slots J. Clinical and microbiological determinants of ailing dental implants. Clin Implant Dent Relat Res. 2009;11(1):24-36.