Layered Bone Grafting in Preparation for Dental Implants
Management of a maxillary defect with a calcium apatite material and particulate allograft
Timothy Kosinski, DDS
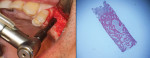
There are many biomaterials used in the bone grafting of socket sites following tooth extraction. If one considers all of the subcategories, there are hundreds of products on the market. Hydroxyapatite, tricalcium phosphate, and other calcium phosphate materials are promoted, and autografts and allografts represent other material options. The homogenous mixture of bioactive resorbable calcium apatite graft crystals and a bovine Achilles tendon collagen matrix (OsteoGen® Plug, Impladent Ltd.) that is used in this case report represents a significant advancement in synthetic grafting materials. This material utilizes a calcium deficient apatite similar to the mineral in human bone allograft. It is bioactive in that it controls soft-tissue invagination and forms a strong bond as it resorbs and is replaced by natural bone. In addition to containing the calcium apatite graft, the plug-shaped material also controls any connective tissue invagination through both a physical and chemical barrier. The physical barrier is created as the plug is firmly compressed into the defect, which causes epithelial cells to grow over the top rather than into the condensed material. As bleeding occurs, the hydrophilic matrix of intertwined graft crystals hydrate with blood and the resorption process begins with the release of calcium ions and the formation of bone.
Case Report
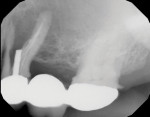
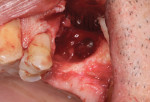
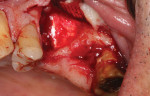
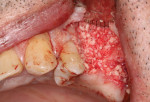
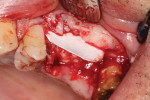
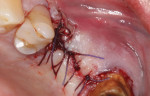
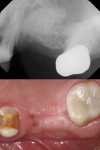
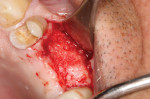
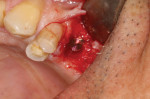
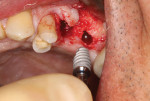
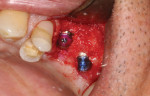
A patient presented with a failing bridge in the left-side maxillary posterior region. A preoperative radiograph revealed a severe vertical fracture of the bicuspid abutment which had resulted in bone loss. Following extraction and thorough surgical debridement of the extraction site and apical lesion, the site was evaluated. It was clear that the Schneiderian membrane was intact yet visible. To develop adequate bone for dental implant placement at the site, a large piece of the alloplastic calcium apatite graft material was carefully, yet firmly, packed into the defect, lifting the membrane. This material provided a new floor for the rest of the grafting procedure. The defect was then completely filled with a particulate allograft material (Newport Biologics™ Mineralized Cortico/Cancellous Allograft Blend, Glidewell Dental). This material was packed firmly into the defect but not crushed, extending to the facial contours of the adjacent teeth. Next, a resorbable collagen membrane (Newport Biologics™ Resorbable Collagen Membrane 3-4, Glidewell Dental) was passively positioned to serve as a barrier against epithelial ingrowth during the healing and remodeling phase. The membrane was trimmed to extend approximately 2 mm beyond the borders of the defect, and a sling Vicryl suture was used to reposition the envelope reflection.
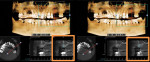
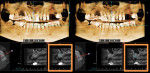
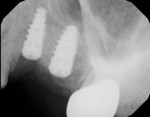
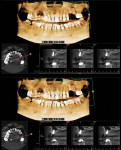
Following a 4-month period of integration, the tissue appeared to be healing well, and a cone-beam computed tomography (CBCT) analysis demonstrated new bone formation. The site was again reflected, and two dental implants (Hahn™ Tapered Implant System, Glidewell Dental) were strategically placed. These implants will be allowed to integrate for an additional 4 months, after which implant level impressions will be made and individual screw-retained, implant-supported crowns will be fabricated and seated. In this manner, the edentulous space will be permanently restored, providing the patient with both improved function and esthetics.
About the Author
Timothy Kosinski, DDS
Diplomate
International Congress of Oral Implantologists
Master
Academy of General Dentistry
Affiliated Adjunct Clinical Professor
School of Dentistry
University of Detroit Mercy
Detroit, Michigan
Private Practice
Bingham Farms, Michigan