The Nonretentive Ceramic Overlay
A biomimetic alternative to the full coverage crown
Steven Schiffenhaus, DMD
In the last 40 years, dentistry has benefited from many advances in adhesives, composites, and ceramics that have allowed the profession to segue from a mechanical restorative goal to a biologic one. These materials, especially lithium disilicate, provide clinicians with the ability to restore teeth in a minimally invasive manner and close to their original biomechanical function. Unfortunately, these materials are not often used to their full potential and are employed with only slight variations to more invasive traditional preparation designs. In fact, the conventional full coverage crown preparation still dominates as the most frequently used indirect restorative procedure when cusp coverage is indicated. A full coverage crown preparation removes approximately 70% to 75% of the tooth structure by weight, whereas preparations for overlays and onlays remove only 32% to 47%.1 The nonretentive overlay presents many advantages as a biomimetic alternative to the traditional crown preparation in situations that require full occlusal coverage. Why choose to do nonretentive partial coverage ceramics? These restorations conserve more tooth structure, work in harmony with the natural biomechanics of the tooth, help extend the restorative life cycle of the tooth, and maximize the fracture resistance of ceramic materials. The preparation design for nonretentive overlays is a highly fracture-resistant design for posterior glass ceramics, and the technique of immediate dentin sealing not only provides excellent retention for these restorations but also reinforces their fracture resistance.
Fracture Resistance
In many studies, the leading cause of ceramic failure is attributed to partial or complete fractures.2 There are many ways to influence the fracture resistance of a ceramic restoration. Increasing material thickness is one of the most obvious, but ultra-thick ceramics call for the removal of excess tooth structure, and their use is not always feasible given the clinical scenario. One way to increase the strength of a ceramic restoration that is independent of its thickness is through modifications to the preparation design.3
In general, conventional ceramic preparation designs are more focused on retention than fracture resistance. These preparation designs are based on parameters that were developed for metal materials from an age when true adhesion was not possible. Preparation designs from the "metal era" required the removal of healthy tooth structure to achieve retention and resistance form. Unfortunately, these traditional retentive design features also result in an increase in both ceramic and bonding (eg, C-factor) stresses as well as increased stress on the supporting tooth structure that can lead to less favorable failure modes.3,4 Complex geometries, sudden transitions, steep walls, edges, and corners all concentrate stress and decrease the fracture resistance of brittle glass materials.2-4Indeed, research has shown that increasing the geometric complexity of the preparation design from an occlusal veneer to a full coverage crown decreases fracture resistance.5,6
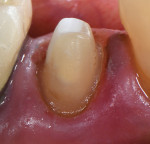
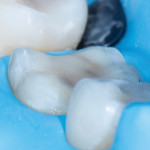
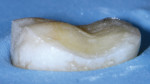
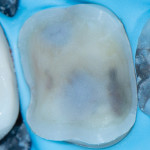
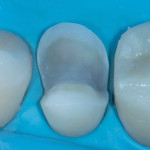
These are not abstract concepts. Regarding the two preparations depicted in Figure 1 and Figure 2, imagine form fitting a piece of glass over each of these structures and then striking down on them with blunt force. Which design seems less likely to fracture? Retentive features such as edges, corners, and boxes not only decrease the fracture resistance of ceramics but also produce higher stresses on the underlying tooth that can contribute to unrestorable failures.4 In general, nonretentive designs decrease ceramic and bonding stresses as well as internal stress on the tooth itself (Figure 3). Polymerization shrinkage stress increases with closely approximated bonded surfaces, such as in boxes and corners, which are typical of preparations with closed geometries.7
Structural Stress Mitigation
Important concepts to consider in ceramic design and maintaining the long-term health of teeth from both a structural and biologic standpoint are those of the "compression dome" and the "bio-rim."8 The compression dome refers to the enamel tooth structure that is above the height of contour that receives mainly compressive forces under load. Ceramics and glasses are very strong regarding compression and very weak regarding tension. In biomimetic dentistry, the tooth structure from the height of contour to the cementoenamel junction that supports the compression dome is referred to as the bio-rim. This region of the tooth sustains the highest amount of tensile stress from function aside from the central groove region.9-11 The dentinoenamel complex (DEC) and the mantle dentin in this region are important structures that have been shown to buffer and mitigate damaging tensile forces.12,13 According to Moiré fringe analysis, stress is highest in the cervical region and outer border of the tooth under load, and this stress is mitigated and dissipated through the DEC.14 The enamel in this region is better able to survive due to its complex microstructure, the presence of thick resilient mantle dentin, and its fiber-reinforced graded interaction with the DEC.12-16
Cracks in enamel that result from compressive forces are usually quickly mitigated through enamel rod decussation; however, cracks that manage to pass all the way through enamel typically start from the cervical region and the DEC, where the tensile forces are highest, and propagate upward toward the occlusal region.17 In fact, enamel tufts, which are anatomically "precracked" areas of enamel at the DEC that are filled with elastic collagen, are hypothesized to aid in stress management because they are more numerous in species that consume hard diets, such as otters that eat sea shells.12,17 These flexible protein-filled clefts that aid in crack blunting are a great example of how nature engineers faliures away from the dentin. Many studies of ceramic fractures indicate that they begin from damaging tensile forces at the internal cement-ceramic interface, not from the occlusal surface.18,19 It is well understood that the mismatch in modulus of elasticity exacerbates stresses and that the DEC provides a graded interface to manage stress and modulus mismatch between the dentin and enamel.12,16,19
Restorative Failure
Research on post and core systems has shown that under stress/strain, both luted and bonded crowns fail at the cervical margin first, which is followed by the occurrence of leakage and then failure of the rest of the system.20,21 The cervical margins of crowns are some of the weakest interfaces, especially when they are luted and not bonded, where there is nothing to stop the margin from microscopically flexing open and closed and pumping oral fluids into the "spacer" region during function. The cervical margin is under the highest tensile load during function, and the Poisson effect results in enhanced lateral tensile load in the cervical region.8,10 Oftentimes, this is why patients with seemingly retentive preparations present with crowns in hand saying that they "popped off," and this includes bonded crowns when the debonding does not result in fracture.
Clinically, in situations involving traumatic occlusion in which higher than normal occlusal forces are overwhelming the tensile mitigating forces of the cervical DEC, the tensile forces can result in failures of the natural tooth structure known as abfractions.22 Although ceramic failures typically occur in the central fissure area, it is not uncommon to observe abfraction-like failures on luted ceramic crowns.
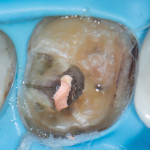
The bio-rim helps to mitigate the deformation of the tooth under load because it is the widest portion of the tooth. Applying beam theory demonstrates that the load bearing capacity of the tooth lies in the outer walls. The stiff and strong outer enamel helps resist deformation, while the DEC buffers damaging tensile forces as well as enhances the brittle enamel's fracture resistance through the fiber-reinforced interface of the two materials.12,15 Under a luted crown, with the outer enamel and DEC no longer bracing and stabilizing the tooth to deforming loads, the much narrower and weaker dentin is less able to prevent cracks from propagating down into the root, which can result in the need for root canal treatment and a guarded future prognosis (Figure 4).
Above the Height of Contour
Preservation of the bio-rim also has implications for the long-term health of the pulp. Preparations and margins in the bio-rim are much closer in proximity to the pulp. As a posterior tooth is reduced occlusally, there is generally 4 mm to 6 mm of tooth structure that can be removed before encountering the pulp, whereas below the height of contour, that distance is often less than 2 mm, and the remaining dentin thickness of cervical ceramic margins is only 0.5 mm to 1.5 mm. Long-term pulp vitality following a restoration involves an important interplay between remaining dentin thickness and microleakage.23-25 The thicker the amount of remaining dentin, the better the pulp is able to withstand the damaging effects of microleakage if it occurs.23,24 In the absence of microleakage, as long as proper burs and coolant are used, preparations can extend until the remaining dentin is 0.5 mm in thickness before irreparable trauma to the pulp occurs.24
In situations where the thickness of the remaining dentin is equal, pulp vitality is directly proportional to how well the restorative method is at minimizing microleakage.23-25 Margins below the height of contour are more prone to damage and microleakage due to the tensile forces concentrated in the cervical tooth structure, which have been described in fatigue loading studies.10,20,21 Not only do margins above the height of contour experience much less tensile flexion, they also are easier to isolate for bonding, contain thicker amounts of enamel for bonding, are easier to clean, and can facilitate much earlier detection of marginal deterioration, improving the long-term prognosis of intervention.
Nonretentive Preparations and Bonded Ceramics
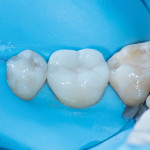
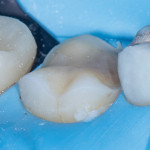
A preparation design that creates an ideal platform for minimizing ceramic stress and functions in harmony with the natural biomechanics of the tooth displays an open and simple geometry, has no edges or corners, and keeps the ceramic margins above the bio-rim (Figure 5 through Figure 8).3,8,26 Although nonretentive preparation designs are arguably more fracture resistant, some clinicians are concerned about the long-term retention of these types of restorations. The use of advanced adhesive bonding techniques, such as immediate dentin sealing, not only provides excellent retention for these restorations but also increases their fracture resistance.27-30
It is well known that bonding a resin to a ceramic will significantly increase the strength of the ceramic.31 In fact, bonded lithium disilicate can approach 75% of the load bearing capacity of 3Y zirconia, exceeding its raw flexural strength tests.18,32 It has also been shown that bonded lithium disilicate can match the strength of 4Y zirconia and exceed the strength of 5Y zirconia, even when bonded to a dentin-like substrate.18
Achieving a higher bond strength to the underlying tooth structure also significantly increases the fracture resistance of the ceramic and has a much greater impact on fracture resistance than the modulus of elasticity of the underlying tooth structure.33 Resin cement systems that achieve better adhesion have been shown to demonstrate higher fracture resistance,33,34 and when utilizing proper dentin bonding protocols, the fracture resistance of lithium disilicate bonded to dentin can match that of ceramic bonded to enamel.18,35
Considering this relationship between adhesion and fracture resistance, it is not surprising that multiple studies have demonstrated that immediate dentin sealing not only results in significantly higher bond strengths for indirect procedures36,37 but also results in a significant increase in ceramic fracture resistance when compared with conventional resin bonding protocols.27-30
Immediate Dentin Sealing
Immediately sealing the dentin with a dentin bonding agent at the time of preparation is a well-documented and well-studied technique that is used to maximize the strength and durability of bonds to dentin.38 This technique was first proposed during the early 1990s, and then, it was further developed into its current methodology.
Since 1984, it has been known that the setting stress of composites during polymerization can have negative effects on dentin bond strength.39 It was recognized that it can take several minutes for the development of the dentin bond to reach sufficient strength as to not be damaged by the polymerization shrinkage stress generated by overlying composite as it cures.39 Quite simply put, if the hybrid layer is not given sufficient time to mature, the forces "pulling" on it damage it and allow dentinal fluid to penetrate into the adhesive, which results in decreased bond strength, poor durability, and sensitivity/pain. Contrary to what many dentists believe, although there is only a small volume of resin in the spacer region, there is quite a high amount of C-factor shrinkage stress due to the lack of unbonded surfaces and closely approximated walls.
Copolymerization of the bonding agent and resin cement results in significantly lower dentin bond strengths.40 It is well known by manufacturers that precuring the bonding agent prior to the application of resin cement is a superior technique regarding bond strength and durability. This technique is not typically used by dentists due to concerns that pooling of the precured adhesive will result in film thicknesses significant enough to interfere with the seating of the final restoration. However, if immediate dentin sealing is considered a technique to precure the bonding agent prior to impression taking, a strong and intact hybrid layer can form that will not interfere with the seating of the final restoration.
Research regarding immediate dentin sealing generally shows that it results in a 400% to 600% increase in bond strengths to dentin and increased durability, including in CAD/CAM workflows.36-38,41 Oftentimes, the strength of these bonds approaches or even exceeds the cohesive strength of enamel, dentin, and the DEC, which is why the technique is sometimes referred to as "biomimetic."16,42 In summary, immediate dentin sealing increases the durability of the dentin bond, providing bond strengths similar to the strengths of the tooth's natural biologic structures, and increases the fracture resistance of ceramic restorations while minimizing sensitivity and leakage.36-38,42,43
Conclusion
Although modern dental ceramics such as lithium disilicate are much stronger than tooth enamel, the enamel on intact natural teeth has a much better survival rate. The conventional approach is to seek out stronger materials or to make existing ones thicker. However, by studying the natural tooth, it should be evident that long-term success in the oral cavity is not about the strongest single material—it's about systems. The tooth is a system. These weaker natural materials work synergistically in such a way as to maximize and exceed the strength of any one part. By approaching restorative design in a systematic way that mimics and supports the natural structure and function of the tooth, clinicians can achieve long-lasting results with minimally invasive techniques. The modifications to preparation designs and adhesive techniques outlined in this article provide a systematic way to maximize the efficacy and longevity of modern ceramics in harmony with the natural tooth.
About the Author
Steven Schiffenhaus, DMD
Private Practice
Gilbert, Arizona
References
1. Edelhoff D, Sorensen JA. Tooth structure removal associated with various preparation designs for posterior teeth. Int J Periodontics Restorative Dent. 2002;22(3):241-249.
2. Morimoto S, Rebello de Sampaio FB, Braga MM, et al. Survival rate of resin and ceramic inlays, onlays, and overlays: a systematic review and meta-analysis. J Dent Res. 2016;95(9):985-994.
3. Arnetzl GV, Arnetzl G. Biomechanical examination of inlay geometries--is there a basic biomechanical principle? Int J Comput Dent. 2009;12(2):119-130.
4. Vianna ALSV, Prado CJD, Bicalho AA, et al. Effect of cavity preparation design and ceramic type on the stress distribution, strain and fracture resistance of CAD/CAM onlays in molars. J Appl Oral Sci.2018;26:e20180004.
5. Huang X, Zou L, Yao R, et al. Effect of preparation design on the fracture behavior of ceramic occlusal veneers in maxillary premolars. J Dent. 2020;97:103346.
6. Ferraris F, Sammarco E, Romano G, et al. Comparison of posterior indirect adhesive restorations (PIAR) with different preparation designs according to the adhesthetics classification. Part 1: effects on the fracture resistance. Int J Esthet Dent. 2021;16(2):144-167.
7. Feilzer AJ, De Gee AJ, Davidson CL. Setting stress in composite resin in relation to configuration of the restoration. J Dent Res. 1987;66(11):1636-1639.
8. Milicich G. The compression dome concept: the restorative implications. Gen Dent. 2017;65(5):55-60.
9. Magne P, Belser UC. Rationalization of shape and related stress distribution in posterior teeth: a finite element study using nonlinear contact analysis. Int J Periodontics Restorative Dent. 2002;22(5):425-433.
10. Ford C, Bush MB, Lawn B. Effect of wear on stress distributions and potential fracture in teeth. J Mater Sci Mater Med. 2009;20(11):2243-2247.
11. Goel VK, Khera SC, Ralston JL, Chang KH. Stresses at the dentinoenamel junction of human teeth - a finite element investigation. J Prosthet Dent. 1991;66(4):451-459.
12. Thompson VP. The tooth: an analogue for biomimetic materials design and processing. Dent Mater. 2020;36(1):25-42.
13. Zaslansky P, Friesem AA, Weiner S. Structure and mechanical properties of the soft zone separating bulk dentin and enamel in crowns of human teeth: insight into tooth function. J Struct Biol. 2006;153(2):188-199.
14. Wang RZ, Weiner S. Strain-structure relations in human teeth using Moiré fringes. J Biomech. 1998;31(2):135-141.
15. Imbeni V, Kruzic JJ, Marshall GW, et al. The dentin-enamel junction and the fracture of human teeth. Nat Mater. 2005;4(3):229-232.
16. Bazos P, Magne P. Bio-emulation: biomimetically emulating nature utilizing a histo-anatomic approach; structural analysis. Eur J Esthet Dent. 2011;6(1):8-19.
17. Chai H, Lee JJ, Constantino PJ, et al. Remarkable resilience of teeth. Proc Natl Acad Sci USA. 2009;106(18):7289-7293.
18. Yan J, Kaizer MR, Zhang Y. Load-bearing capacity of lithium disilicate and ultra-translucent zirconias. J Mech Behav Biomed Mater. 2018;88:170-175.
19. Wang Z, Wang K, Xu W, et al. Mapping the mechanical gradient of human dentin-enamel-junction at different intratooth locations. Dent Mater. 2018;34(3):376-388.
20. Lazari PC, de Carvalho MA, Del Bel Cury AA, Magne P. Survival of extensively damaged endodontically treated incisors restored with different types of posts-and-core foundation restoration material. J Prosthet Dent. 2018;119(5):769-776.
21. Freeman MA, Nicholls JI, Kydd WL, Harrington GW. Leakage associated with load fatigue-induced preliminary failure of full crowns placed over three different post and core systems. J Endod. 1998;24(1):26-32.
22. Peumans M, Politano G, Van Meerbeek B. Treatment of noncarious cervical lesions: when, why, and how. Int J Esthet Dent. 2020;15(1):16-42.
23. Murray PE, Hafez AA, Smith AJ, Cox CF. Bacterial microleakage and pulp inflammation associated with various restorative materials. Dent Mater. 2002;18(6):470-478.
24. Murray PE, Smith AJ, Windsor LJ, Mjör IA. Remaining dentine thickness and human pulp responses. Int Endod J. 2003;36(1):33-43.
25. Kwang S, Aminoshariae A, Harding J, et al. The critical time-lapse between various restoration placements and subsequent endodontic intervention. J Endod. 2014;40(12):1922-1926.
26. Politano G, Van Meerbeek B, Peumans M. Nonretentive bonded ceramic partial crowns: concept and simplified protocol for long-lasting dental restorations. J Adhes Dent. 2018;20(6):495-510.
27. Hofsteenge JW, Hogeveen F, Cune MS, Gresnigt MMM. Effect of immediate dentine sealing on the aging and fracture strength of lithium disilicate inlays and overlays. J Mech Behav Biomed Mater. 2020;110:103906.
28. Gresnigt MMM, Cune MS, de Roos JG, Özcan M. Effect of immediate and delayed dentin sealing on the fracture strength, failure type and Weilbull characteristics of lithiumdisilicate laminate veneers. Dent Mater. 2016;32(4):e73-81.
29. van den Breemer CRG, Özcan M, Cune MS, et al. Effect of immediate dentine sealing on the fracture strength of lithium disilicate and multiphase resin composite inlay restorations. J Mech Behav Biomed Mater. 2017;72:102-109.
30. Yazigi C, Kern M, Chaar MS. Influence of various bonding techniques on the fracture strength of thin CAD/CAM-fabricated occlusal glass-ceramic veneers. J Mech Behav Biomed Mater. 2017;75:504-511.
31. Fleming GJP, Addison O. Adhesive cementation and the strengthening of all-ceramic dental restorations. J Adhes Sci Technol. 2009;23(7-8):945-959.
32. Ma L, Guess PC, Zhang Y. Load-bearing properties of minimal-invasive monolithic lithium disilicate and zirconia occlusal onlays: finite element and theoretical analyses. Dent Mater. 2013;29(7):742-751.
33. Piemjai M, Arksornnukit M. Compressive fracture resistance of porcelain laminates bonded to enamel or dentin with four adhesive systems. J Prosthodont. 2007;16(6):457-464.
34. Rojpaibool T, Leevailoj C. Fracture resistance of lithium disilicate ceramics bonded to enamel or dentin using different resin cement types and film thicknesses. J Prosthodont. 2017;26(2):141-149.
35. Clausen JO, Abou Tara M, Kern M. Dynamic fatigue and fracture resistance of non-retentive all-ceramic full-coverage molar restorations. Influence of ceramic material and preparation design. Dent Mater. 2010;26(6):533-538.
36. Magne P, Kim TH, Cascione D, Donovan TE. Immediate dentin sealing improves bond strength of indirect restorations. J Prosthet Dent. 2005;94(6):511-519.
37. Okuda M, Nikaido T, Maruoka R, et al. Microtensile bond strengths to cavity floor dentin in indirect composite restorations using resin coating. J Esthet Restor Dent. 2007;19(1):38-48.
38. Qanungo A, Aras MA, Chitre V, et al. Immediate dentin sealing for indirect bonded restorations. J Prosthodont Res. 2016;60(4):240-249.
39. Davidson CL, de Gee AJ, Feilzer A. The competition between the composite-dentin bond strength and the polymerization contraction stress. J Dent Res. 1984;63(12):1396-1399.
40. Lührs AK, Pongprueksa P, De Munck J, et al. Curing mode affects bond strength of adhesively luted composite CAD/CAM restorations to dentin. Dent Mater. 2014;30(3):281-291.
41. Murata T, Maseki T, Nara Y. Effect of immediate dentin sealing applications on bonding of CAD/CAM ceramic onlay restoration. Dent Mater J. 2018;37(6):928-939.
42. Urabe I, Nakajima M, Sano H, Tagami J. Physical properties of the dentin-enamel junction region. Am J Dent. 2000;13(3):129-135.
43. Giannini M, Soares CJ, de Carvalho RM. Ultimate tensile strength of tooth structures. Dent Mater. 2004;20(4):322-329.