Predictable Soft-Tissue Grafting for Extreme Cases
Combining SCTGs with ADM to simultaneously treat multiple gingival recession defects
Edgard El Chaar, DDS, MS | Gretchen Stern, DDS
Gingival recession, defined as the apical shift of the gingival margin with respect to the cementoenamel junction, is a common finding among adult patients.1,2 In addition to attachment loss, recession-type defects are associated with exposure of the root surface to the oral environment, which renders the tooth susceptible to both carious and noncarious lesions, hypersensitivity, and poor esthetics.3
There are a variety of treatment modalities that have been proposed for the plastic surgical correction of these defects, including the free gingival graft, subepithelial connective tissue graft (SCTG), lateral or sliding pedicle graft, and coronally advanced flap, with or without the use of extracellular matrix-based scaffolding technologies.4 The fundamental principles that support the successful surgical treatment of mucogingival deformities remain consistent regardless of the elected treatment modality. These principles include flap management and integrity, graft stabilization, and an understanding of wound healing.
Mucogingival Surgical Techniques
One of the main sources of blood supply to a graft is the overlying flap. Consequently, meticulous incision design and flap management are critical for the preservation of the vasculature, which ultimately leads to graft revascularization and successful and predictable surgical outcomes. Although gingival recession is more often generalized in distribution, the majority of studies report on the treatment of localized defects.5 In 1975, Bernimoulin and colleagues described the first technique for the treatment of multiple adjacent gingival recession defects. This two-stage approach involved the placement of a free gingival graft that was followed by a second surgery 2 months later to coronally position the flap.6 Twenty-five years later, Zucchelli and De Sanctis presented a novel technique for flap management of multiple teeth in the esthetic zone, using a modification to the envelope flap first described by Bruno in 1994.7,8 The authors proposed the creation of a surgical papilla to facilitate coronal advancement along with the use of a split-full-split-thickness flap design. Deep periosteal incisions were utilized to achieve tension-free flap advancement.7 The tunnel technique has also been utilized for the treatment of multiple adjacent gingival recession defects; however, the majority of the data comparing use of a coronally advanced flap to that of the tunnel technique demonstrate greater success with a coronally advanced flap.9
The main metric used to assess surgical success is complete root coverage, which is the complete coverage of the exposed root surface to a point at the cementoenamel junction. Despite the advances in mucogingival surgical techniques, cases in which complete root coverage cannot be expected due to rotation, extrusion, or loss of interproximal clinical attachment are associated with greater complexity and less predictable outcomes. In such cases, it may be more pragmatic for the clinician and patient to anticipate achieving maximum root coverage rather than complete root coverage.
Differences in root coverage outcomes have been observed between the dental arches with greater success rates correlated with the maxillary arch with respect to complete and maximum root coverage.5,9,10 Some have attributed the lower success rates associated with the mandible to the reduced papilla size, greater muscle pull, and the shallow vestibular depth characteristic of the mandibular arch.10 In such cases, treating the full mandibular arch in one surgical attempt may facilitate better flap mobilization and decrease patient discomfort.
Increasing Predictability
Lack of predictability is also associated with cases involving malpositioned teeth, prominent roots, and loss of interproximal attachment. During the initial days of wound healing, a graft survives by plasmatic diffusion prior to initiating revascularization. The blood supply to the graft is derived from the periodontal ligament, the supraperiosteal plexus, and the overlaying flap.11 Teeth with prominent roots that protrude beyond the limits of the alveolar housing naturally develop adjacent concavities localized on the buccal aspect of the alveolar bone between adjacent roots. If a soft-tissue graft is placed over a prominent root and the adjacent area, this can lead to a dead space between the alveolar bone and the grafted tissue. The uneven recipient bed created by the prominent root promotes a lack of contact between the soft-tissue graft and the underlying bone with its associated vasculature, which can compromise the blood supply to the graft during the initial phases of healing.12 A reduced blood supply may negatively impact the successful revascularization of the graft and, consequently, the amount of root coverage that can be achieved.13
To eliminate the dead space created from excessive tooth proclination and/or root prominence, some have proposed a subtractive technique involving odontoplasty of the root surface.14,15,16 However, flattening of the root surface requires the elimination of sound tooth structure. This can weaken the tooth and expose the dentinal tubules, potentially resulting in hypersensitivity, particularly in the event that complete root coverage is not achieved, which is common in these more complex cases. As such, an additive technique in which bone graft material is placed on either side of the prominent root is preferred to create a level recipient bed for the soft-tissue graft. This technique can be employed utilizing autogenous and/or extracellular matrix-based scaffolding technologies and eliminates the need for unnecessary removal of viable tooth structure.12
Multiple adjacent gingival recession defects often require a large quantity of tissue to cover the affected area. The use of SCTGs, which are considered the gold standard for root coverage, may be limited by an insufficient quantity of autogenous tissue available to cover all of the affected sites, necessitating the use of an additional soft-tissue graft substitute. Various extracellular matrix-based scaffolding technologies, such as acellular dermal matrix (ADM), have been incorporated into the periodontal plastic surgical armamentarium for this purpose.
Therefore, a simultaneous hard- and soft-tissue grafting technique utilizing particulate bone graft to support the use of both an autogenous SCTG and an ADM can improve success when treating multiple adjacent gingival recessions. The following case report demonstrates how this technique can be implemented in complex cases where root coverage is considered less predictable.
Case Report
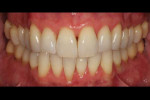
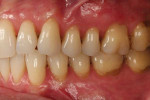
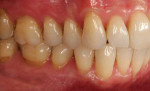
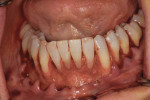
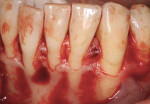
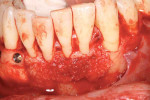
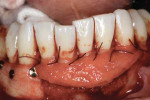
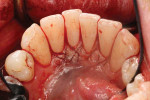
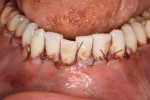
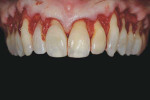
A patient presented with generalized gingival recession with interproximal clinical attachment loss that was consistent with Cairo recession type 2 (RT2) (Figure 1 through Figure 3).17 The recession defect associated with tooth No. 25 resulted in a lack of keratinized tissue. The thin gingival phenotype and root prominence were particularly apparent through the blanching of the gingiva, which was most clearly visible on tooth Nos. 22 and 21.
Preoperative Considerations
Preoperatively, the patient was prescribed a 1-week course of antibiotics to begin taking on the day of surgery. In addition, the patient was prescribed an antimicrobial rinse with instructions to begin use the day after surgery. Peri-oral disinfection was accomplished utilizing a povidone-iodine solution. A gauze soaked in antimicrobial solution was wiped intraorally, and then all of the teeth were debrided and ultrasonically scaled.
Incision Design and Flap Management
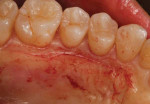
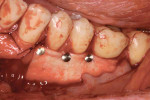
An intrasulcular, papilla-sparing incision was made from the mesial aspect of the mandibular second molar to the mesial aspect of the opposing mandibular second molar (Figure 4). Next, a full-thickness envelope flap was reflected without the use of vertical releasing incisions. The exposed roots were scaled and treated with a neutral root conditioner (PrefGel®, Straumann) to aid in the removal of all debris. In addition, the alveolar bone was thoroughly debrided.
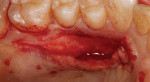
The full-thickness flap elevation revealed prominent roots with thin and dehisced buccal bone (Figure 5). Blood can be seen pooling adjacent to the root surfaces, occupying the space of the bony concavities that result from root prominence. If left untreated, these concavities will result in dead space.
SCTG Harvesting
One or two SCTGs can be harvested from either side of the palate. Using a single incision approach and depending upon the depth of the palate, the SCTGs may be harvested from the area mesial to the canine extending to the area mesial or mid-palatal to the second molar (Figure 6). To avoid damaging the palatal gingiva of the maxillary teeth, the incision should respect a 3- to 4-mm distance from the free gingival margin. Presurgical evaluation of the palatal anatomy is critical in order to prevent intraoperative complications, such as excessive bleeding, which may occur due to violation of important palatal anatomical landmarks.
For this case, a single incision approach utilizing a full-thickness envelope flap was made on the hard palate (Figure 7). Once the donor tissue was harvested, the palatal tissue was sutured immediately, employing a modified sling technique that anchors on the facial aspect of the maxillary posterior teeth without engaging the maxillary papillae (Figure 8). The needle is passed without engaging the papilla to the buccal aspect of the maxillary teeth, where a knot is tied. This modified suture technique facilitates palatal hemostasis and provides stability to ensure closure. In this manner, the teeth are used as anchors, providing stability and assisting with closure of the donor site. As demonstrated in Figure 8, a collagen tape was used to support the palatal donor tissue after harvesting of the graft, and a tissue adhesive was placed for further reinforcement. This, however, is unnecessary, and superior healing has since been observed with suturing alone.
Bone Grafting and Soft-Tissue Stabilization
The span of the complete mandibular arch is often too large to be treated solely with autogenous soft tissue. Therefore, ADM may be used as a supplemental soft-tissue graft in less severely affected areas and where greater predictability of root coverage can be achieved (eg, posterior mandible). The SCTG should be reserved for areas of greater severity. In the mandible, this is frequently found in the anterior sextant.
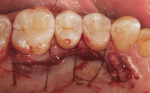
In this case, small particle mineralized cancellous bone allograft was used to graft the concavities and create a level recipient bed for the SCTG (Figure 9). The SCTG was placed over the root surface and particulate bone graft, and it was subsequently stabilized using interrupted sutures that were placed through the coronal portion of the graft and the sliced papilla, anchoring on the lingual gingiva (Figure 10 and Figure 11).
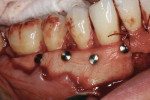
The ADM was prepared according to the manufacturer's protocol. Once it was positioned in the posterior, stabilization was accomplished with titanium alloy tacks, which would enhance graft adaptation to the alveolar bone and decrease the amount of inevitable micromovement produced during the normal healing response in which the flap undergoes physiologic dimensional changes (Figures 12 and Figure 13).
In order to avoid periosteal incisions that could compromise the blood supply to the flap, stretching of the flap was preferred to achieve tension-free coronal advancement. The flap was then secured in a coronal position utilizing sling sutures with tension-free closure.
Postoperative Follow-Up
The patient was seen on a weekly basis for the first 4 weeks after surgery. Because early suture removal has been shown to negatively affect complete root coverage, sutures should be removed no earlier than 10 days postoperatively.19 The patient was placed on a soft diet and instructed to avoid interproximal cleaning for the first 2 weeks and brushing for the first month. Additional appointments at 3, 6, 9, and 12 months were completed to monitor the healing and response to treatment.
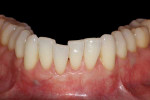
The patient experienced minimal discomfort postoperatively and healed uneventfully without infection. At the 24-month postoperative visit, the patient demonstrated a clinically appreciable reduction in gingival recession and significant root coverage (Figure 15). Although this case initially had a more unfavorable prognosis, 24 months later, the tissue is thick, clinically healthy, and stable. If visibility of the titanium alloy tacks through the gingiva becomes an esthetic concern for the patient, once healing is complete, they can be removed under local anesthesia without flap elevation.
Discussion
This technique for achieving root coverage in the mandibular arch in a single appointment has been documented by the authors in 13 cases with successful outcomes (See Table 1). The overall mean root coverage achieved was 97.6%, with a mean gain in keratinized tissue of 2.07 mm. This technique may also be utilized in the maxillary arch for the predictable treatment of multiple adjacent gingival recessions. However, when treating the maxilla, it is recommended that the incision design be modified to exclude the interdental papilla between the maxillary central incisors, leaving it intact for esthetic reasons. The incision design for the remainder of the arch is accomplished with sulcular, papilla sparing incisions in the same manner as the mandibular arch (Figure 16).
Conclusion
The presented technique for the treatment of complex cases of severe gingival recession utilizes the fundamental principles of wound healing to optimize graft revascularization and stability, which ultimately dictate treatment success. For dentists, the ultimate goal is to arrest disease and maintain the teeth in form and function when possible. This technique facilitates optimal treatment outcomes without compromising the integrity of the teeth and their surrounding periodontal tissues.
About the Authors
Edgard El Chaar, DDS, MS
Director and Clinical Associate Professor
Advanced Education Program in Periodontics
New York University
College of Dentistry
New York, New York
Diplomate
American Board of Periodontology
Private Practice
New York, New York
Gretchen Stern, DDS
Private Practice
New York, New York
References
1. Pini Prato GP. Mucogingival deformities. Ann Periodontol. 1999;4(1):98-101.
2. Kassab MM, Cohen RE. The etiology and prevalence of gingival recession. J Am dent Assoc. 2003;134(2):220-225.
3. Cortellini P, Bissada NF. Mucogingival conditions in the natural dentition: narrative review, case definitions, and diagnostic considerations. J Periodontol. 2018;89(Suppl 1):S204-S213.
4. Tavelli L, McGuire MK, Zucchelli G, et al. Extracellular matrix-based scaffolding technologies for periodontal and peri-implant soft tissue regeneration. J Periodontol. 2020;91(1):17-25.
5. Zucchelli G, Tavelli L, Barootchi S, et al. The influence of tooth location on the outcomes of multiple adjacent gingival recessions treated with coronally advanced flap: a multicenter re-analysis study. J Periodontol. 2019;90(11):1244-1251.
6. Bernimoulin JP, Luscher B, Muhlemann HR. Coronally repositioned periodontal flap. Clinical evaluation after one year. J Periodontol. 1975;2(1):1-13.
7. Zucchelli G, De Sanctis M. Treatment of multiple recession type defects in patients with esthetic demands. J Periodontol. 2000;71(9):1506-1514.
8. Bruno JF. Connective tissue graft technique assuring wide root coverage. Int J Periodontics Restorative Dent. 1994;14(12):126-137.
9. Tavelli L, Barootchi S, Nguyen TV, et al. Efficacy of tunnel technique in the treatment of localized and multiple gingival recessions: a systematic review and a meta-analysis. J Periodontal. 2018;89(9):1075-1090.
10. Aroca S, Barbieri A, Clementine M, et al. Treatment of class III multiple gingival recessions: prognostic factors for achieving a complete root coverage. J Clin Periodontol. 2018;45(75):861-868.
11. Guiha R, Khodeiry SE, Mota L, et al. Histological evaluation of healing and revascularization of the subepithelial connective tissue graft. J Periodontol. 2001;72(4):470-478.
12. El Chaar E, Oshman S, Danesh-Sani SA, et al. Increasing contact between soft tissue graft and blood supply. A technique for managing connective tissue grafting over prominent root clinical case. Current Opinion in Cosmetic Dentistry. 2016;32(3):52-66.
13. Mörmann W, Bernimoulin JP, Schmid M. Fluorescein angiography of free gingival autografts. J Clin Periodontol. 1975;2(4):177-189.
14. Miller PD. Root coverage using the free soft tissue autograft following citric acid application. III. A successful and predictable procedure in areas of deep-side recession. Int J Periodontics Restorative Dent. 1985;5(2):14-37.
15. Zadeh HH. Minimally invasive treatment of maxillary anterior gingival recession defects by vestibular incision subperiosteal tunnel access and platelet-derived growth factor BB. Int J Periodontics Restorative Dent. 2011;31(6):653-660.
16. Nunez J, Sanz-Esporrin J, Caffesse R, et al. A two-step surgical approach with flattening of the root surface to treat localized gingival recessions affecting mandibular incisors: A case series report. Int J Periodontics Restorative Dent. 2018;38(5):683-690.
17. Cairo F, Nieri M, Cincinelli S, et al. The interproximal clinical attachment level to classify gingival recessions and predict root coverage outcomes: an explorative and reliability study. J Clin Periodontol. 2011;38(7):661-666.
18. El Chaar E, Urtula B, Georgantza A, et al. Treatment of atrophic ridges with titanium mesh: a retrospective study using 100% mineralized allograft and comparing dental stone versus 3D-printed models. Int J Periodontics Restorative Dent. 2019;39(4):491-500.
19. Tatakis DN, Chambrone L. The effect of suturing protocols on coronally advanced flap root-coverage outcomes: A meta-analysis. J Periodontol. 2016;87(2):148-155.