An Overview of Peri-Implant Diseases
Early detection, treatment, and management are essential to prevent implant loss
Ramzi V. Abou-Arraj, DDS, MS | Hussein S. Basma, DDS
Dental implants have been extremely successful substitutes for missing teeth and have demonstrated high survival and success rates in multiple, long-term studies.1 Nonetheless, dental implants are not immune to inflammatory processes and are subject to numerous risk factors that may lead to the development of infectious peri-implant diseases. Peri-implant mucositis is defined as a mucosal inflammation around implants without the loss of supporting bone, whereas peri-implantitis is an inflammatory process that includes both soft-tissue inflammation and the progressive loss of supporting bone.2 The literature presents controversial findings about the frequency of peri-implant disease. Prevalence ranges of 19% to 65% and 1% to 47% have been reported for peri-implant mucositis and peri-implantitis, respectively.3 This wide variability may be explained by the use of different parameters to evaluate and diagnose peri-implant diseases.
Etiology of Peri-Implant Diseases
A direct relationship exists between bacterial plaque accumulation and the development of inflammatory changes in the soft tissues surrounding implants.4 Bacterial colonization of implant surfaces and peri-implant mucosal surfaces occurs rapidly, leading to local inflammation, which subsequently leads to a complex host response that might result in considerable tissue damage. This host response, which is provoked by inflammatory cells that secrete cytokines and matrix metalloproteinases, causes the degradation of both connective tissue and bone. Peri-implant diseases have been associated predominantly with gram-negative anaerobic bacteria. Predictably, the most virulent periodontal pathogens are found at higher frequencies in peri-implantitis sites, which harbor a more complex microbiota than both healthy tissues and those affected by periodontal disease. The identification of other bacteria, such as Staphylococcus aureus, and viruses, such as human cytomegalovirus and Epstein-Barr virus, in subgingival plaque suggests a pathogenic role in peri-implant diseases. Nevertheless, these microbial profiles are not currently used to aid in the diagnosis of peri-implant disease in clinical settings.
Predisposing Factors for Peri-Implant Diseases
Peri-implant diseases have multifactorial etiologies that can be related to implant factors (eg, material, design, surface properties), clinician factors (eg, surgical technique, restorative treatment), or patient factors (eg, systemic diseases and conditions, medications, smoking, bone quality and quantity).
Poor Oral Hygiene
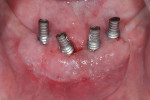
Given its infectious origin, it is not surprising that poor plaque control is a major risk factor for peri-implant disease (Figure 1). Implant sites that are less accessible to oral hygiene measures exhibit greater rates of plaque accumulation leading to peri-implant defects (48%) when compared with more accessible sites (4%).5
Local factors, such as exposed roughened implant surfaces and poor prosthetic design, contribute to greater retention and accumulation of plaque. In the scientific literature, uncertainty exists regarding the role of roughened implant surfaces in peri-implant disease. In the presence of peri-implantitis, roughened surfaces seem to facilitate a more advanced progression of the disease than smooth surfaces.
Lack of Keratinized Mucosa
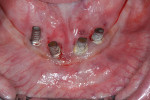
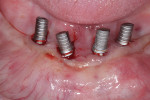
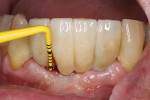
It is widely accepted that the presence of an adequate amount of keratinized mucosa (ie, 2 mm or greater) helps maintain healthy soft and hard tissues around implants (Figure 2 and Figure 3). Although more research is needed, the literature about this topic seems to indicate that, in the presence of good plaque control, the amount of keratinized mucosa has little influence on soft-tissue inflammation. In situations where proper hygiene measures are hindered by difficult access, the risk of inflammatory signs and destruction is greater when the keratinized mucosa is less than 2 mm.6
Smoking
Studies indicate that smoking is the greatest identifiable and most often cited risk factor for peri-implant disease followed by a history of periodontitis. The negative effects of smoking on the host response are well-established. Smoking before or after implant placement leads to a 35% to 70% higher risk of implant failure when compared with not smoking.7 Despite these numbers, prospective studies have failed to establish a significant connection between smoking and the incidence of peri-implantitis.
History of Periodontitis
It is well-documented that individuals with periodontitis are more susceptible to developing peri-implant disease. Patients with a history of chronic periodontitis have a higher prevalence of peri-implantitis (28.6%) than healthy patients (5.8%).8 Unfortunately, periodontal pockets can become infectious reservoirs, allowing the transfer of microorganisms from a residual pocket to an adjacent peri-implant site.
Restorative Factors
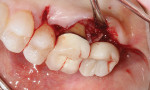
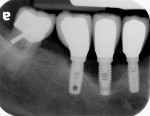
Considering that many of the commonly used cements are not detectable on radiographs, the incomplete removal of excess cement is a growing concern, especially when located on buccal and/or lingual surfaces (Figure 4 and Figure 5). Bacteria are easily retained on the rough surfaces of cement remnants. The prevalence of cement-induced peri-implant disease was reported to be higher when less than 4 weeks of soft-tissue healing was allowed after the second stage of implant surgery and before cementation.9 In addition, periodontally susceptible individuals with residual excess cement are more prone to develop peri-implant disease than periodontally healthy patients.
Because it should facilitate proper oral hygiene access and implant maintenance, the design of a prosthesis plays an important role. Non- or ill-fitting restorations (eg, those exhibiting microleakage at the implant/abutment or abutment/prosthesis interfaces) may favor bacterial infiltration in gaps that can lead to peri-implant disease.
Parafunctional Habits
The clenching or grinding of teeth is considered a risk factor for implant success. These parafunctional habits are capable of producing lateral forces that are detrimental to the wound healing process, especially in the early phase. Ultimately, uncontrolled parafunctional habits can result in implant fracture or loss. It has been theorized that excessive non-axial forces create a resorptive remodeling bone response around the implant collar that results from microdamage of the bone adjacent to the implant surface.10 It is important to remember that dental implants lack proprioception, which is a feature of the periodontal ligament. The presence of periodontal ligament around teeth offers a high degree of protection from excessive parafunctional forces, both through the properties of proprioception and by allowing the teeth to migrate away from occlusal forces. Therefore, prosthetic and occlusal principles must be respected during the treatment planning and restorative phases to avoid implant overloading. The importance of performing a risk assessment and providing patient education on the risks of parafunctional habits with regards to implant therapy cannot be overstressed.
Systemic Diseases and Medications
Uncontrolled systemic diseases and conditions, such as diabetes mellitus, osteoporosis, and hormonal changes, cause metabolic and host response alterations that could influence peri-implant health. Patients who take medications that affect bone turnover, such as antiresorptive medications (eg, oral and IV bisphosphonates, RANKL inhibitors) and antiangiogenic medications, should be carefully evaluated when considering implant therapy. Among these patients, the risk of developing osteonecrosis of the jaw following implant placement is unknown. However, a study with limited data recently showed that implant insertion is a potential risk factor for osteonecrosis of the jaw, especially among patients who have been using IV bisposphonates in combination with corticosteroids for longer than 3 years.11
Diagnosis of Peri-Implant Diseases
The early detection of peri-implant disease may prevent the progression of reversible peri-implant mucositis into peri-implantitis and implant loss. Peri-implant mucositis has similar features to gingivitis and is characterized by swelling, redness, and bleeding on probing. In addition to bleeding on probing, peri-implantitis presents with deepened probing depths and bone loss. The presence of suppuration alone fails to distinguish between the two conditions.
Clinical Exam
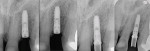
A thorough clinical exam is necessary to differentiate healthy tissues from those that are diseased. Evaluation of peri-implant tissues is performed, preferably with a plastic periodontal probe, to determine probing depth and evaluate for the presence of bleeding on probing and suppuration (Figure 6). It is worth noting that probing around implants does not hold the same diagnostic value as probing around natural teeth. Due to the lack of true soft-tissue attachment to dental implants or abutments, the probe tends to advance much closer to the bone crest level, even in healthy tissues. In addition, the implant, abutment, and prosthesis design; direction of probe insertion; and soft-tissue health status all have a significant influence on probing measurements. Despite these limitations, probing around implants should be performed regularly to allow for a comparison of probing depths over time. A working group from the Seventh European Workshop of Periodontology concluded that the presence of bleeding on probing characterizes peri-implant disease.12Although bleeding on probing is unlikely at healthy sites around natural teeth, it could still occur at healthy implant sites.13
Soft-tissue characteristics are inspected visually and by palpation for color, consistency, and contour. These parameters should be recorded at each maintenance visit and evaluated for progression.
Implant mobility should be routinely checked with two rigid instruments. When movement is detected, the clinician must determine whether it is caused by a loosening of the prosthetic component or true mobility of the implant fixture. Percussion of the implant restoration creates a ringing sound when healthy and a dull sound in cases of implant fixture failure or prosthetic instability. To eliminate untoward forces that may contribute to irreversible changes in the peri-implant tissues, implant occlusion should be verified at every visit. Pain and swelling are unusual signs, which if present, are typically associated with an acute infection at the implant site.
Radiographic Exam
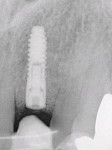
Similar to gingivitis, peri-implant mucositis does not present with bone loss. Conversely, peri-implantitis is characterized by radiographic evidence of crestal bone destruction (Figure 7). The defect is usually saucer-shaped and is associated with the formation of a peri-implant pocket, bleeding on probing, and suppuration.
Radiographs should be taken at the time of implant placement, the prerestorative visit, the final impression, the abutment seating, the restoration placement (baseline radiograph), every 6 months during the first year, and annually or biennially thereafter. The baseline radiograph is of particular importance because it represents the time that the implant enters into function and serves as a reference for the evaluation of future bone loss.
Because panoramic radiographs lack the detailed information that individual radiographs can capture, periapical and vertical bitewing radiographs are recommended for the evaluation of peri-implant bone levels and pathoses. These radiographs should be taken perpendicular to the implant body so that the threads can be seen independently. The use of cone-beam computed tomography should be reserved for implant treatment planning and cases with evidence of peri-implantitis that require 3-dimensional information for the consideration of treatment options.
Management of Peri-Implant Diseases
Non-Surgical Treatment
Non-surgical treatment, which includes patient and professional measures for plaque control around dental implants, is most successful for peri-implant mucositis and, to a lesser degree, early peri-implantitis. If the prosthesis hinders access for proper cleaning, it must be modified.
Mechanical debridement of plaque and calculus is typically performed during prophylactic and maintenance visits with carbon fiber or titanium-coated curettes, ultrasonic scalers with non-metallic tips, and rubber cups with polishing paste. Plastic instruments cause the least damage to implant surfaces, but are also the least effective. Due to implant geometry, the sub-mucosal area provides the most challenges, especially in the presence of bone loss.
When a site is persistently inflamed despite proper plaque control, locally-delivered antibiotics and chlorhexidine rinses can be used as adjuncts to mechanical debridement. There is some evidence suggesting that signs of inflammation in peri-implant disease may be reduced with a submucosal application of local antibiotics and glycine powder air-polishing.14
Surgical Treatment
When the signs of peri-implant disease fail to resolve after non-surgical therapy, advanced implant surface decontamination treatment is required, especially when radiographic bone loss is evident along with probing depths greater than 5 mm, bleeding on probing, and/or suppuration.
Implant surface decontamination has been attempted by various methods. One such method, implantoplasty (ie, removing the implant threads) is supported by limited evidence that it improves clinical outcomes. Lasers, particularly CO2 and Er:YAG, have been used for decontamination treatment, but they yield similar outcomes to non-surgical or surgical therapies without lasers.15 The use of air-polishing or chemical agents, such as chlorhexidine or tetracycline, have also been shown to be effective, but fail to offer any clear advantage over the use of mechanical debridement and saline irrigation. Surgical therapeutic options include resective surgery, regenerative surgery, and explantation:
Resective surgery. The primary goal of resective surgery is to improve access for implant surface debridement and facilitate hygiene measures through hard- and soft-tissue reshaping. Resective surgery is preferred when the morphology of the bone defect is not conducive to grafting techniques. In this manner, the peri-implant bone is recontoured to eliminate the defects. A 2-mm reduction in probing depth has been reported after resective therapy.16 In this therapy, ostectomy, osteoplasty, and sometimes implantoplasty are performed to reduce or even stop the progression of peri-implantitis. Nevertheless, due to the increased postoperative recessions, this procedure is not suitable for every situation, especially in highly esthetic areas. In these cases, an open flap procedure may be the treatment of choice to achieve access for the debridement of implant surfaces with a minimal change in the final position of the mucosal margin.
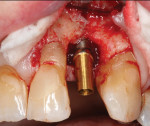
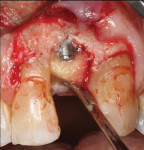
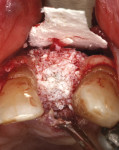
Regenerative surgery. Ailing implants with crater-shaped bone defects may benefit the most from regenerative therapies. Implants that show radiographic evidence of vertical (ie, angular) bone loss but lack residual buccal and lingual bony walls are not good candidates due to graft containment challenges. Various bone grafting materials (eg, autogenous grafts, allografts, xenografts), with or without the use of resorbable or non-resorbable barrier membranes, have been employed in the treatment of peri-implant defects (Figure 8 through Figure 14). Collectively, studies have demonstrated reductions in probing depth averaging 2.3 mm when bone grafts were used alone and 3.2 mm when they were used in combination with barrier membranes. The amount of radiographic bone fill has been found to be comparable when grafts were used alone or in combination with membranes (2.1 mm versus 2.2 mm).16 With respect to the choice of materials in regenerative procedures for peri-implantitis lesions, there is no conclusive evidence supporting the efficacy of one over another.
Explantation. In situations where advanced bone loss has led to a hopeless implant prognosis or when implant malposition renders maintenance practices too challenging, explantation becomes the most viable option. Failing implants are usually explanted using retrieval tools that are provided by the manufacturer, along with trephines and forceps, depending on the quantity of the residual bone. It should be noted, however, that the external diameters of trephines can be up to 1.5 mm greater than the diameter of implants, which may lead to significant bone destruction, including the buccal or lingual bone cortices, and damage to adjacent natural teeth where the interradicular space is limited. When there is less than 3 to 4 mm of residual bone support, implants may be extracted by forceps. Efforts to reconstruct the ridge after explantation are recommended and depend on the amount of residual bone and the future treatment plan to replace the lost implant(s).
To simplify the treatment of peri-implantitis, Lang and colleagues developed the cumulative interceptive supportive therapy (CIST) protocol.17 It provides guidance for which treatment actions to consider at various stages of peri-implant disease. The protocol is divided into four treatment sequences: A) mechanical debridement, B) antiseptic treatment, C) antibiotic treatment, and D) resective or regenerative treatment. In healthy implants, only CIST A (ie, mechanical debridement of the implant surface) is required as yearly maintenance. In cases of mild to moderate peri-implant disease, where pocket depth is 4 to 6 mm with bleeding on probing, suppuration, and bone loss of less than 2 mm, CIST A+B+C (ie, mechanical debridement, antiseptic therapy with chlorhexidine, and the use of local or systemic antibiotics) is indicated. In cases of severe disease with cratering bone loss, probing depths greater than 5 mm, suppuration, and/or bleeding on probing, treatment should include CIST A+B+C+D (ie, mechanical debridement, antiseptic therapy, antibiotic therapy, and surgical therapy) as well as maintenance treatment every 3 months.
Conclusion
Although peri-implant disease and periodontal disease share many common etiologies and treatment options, they are not entirely similar. Proper risk assessment at the time of treatment planning and following implant therapy remains essential for the prevention of implant complications. To avoid implant loss, dentists and hygienists must play a central role in the prevention, early detection, and management of peri-implant disease.
About the Authors
Ramzi V. Abou-Arraj, DDS, MS
Associate Professor
Department of Periodontology
University of Alabama at Birmingham School of Dentistry
Birmingham, Alabama
Hussein S. Basma, DDS
Resident
Department of Periodontology
University of Alabama at Birmingham School of Dentistry
Birmingham, Alabama
References
1. Lang NP, Berglundh T, Heitz-Mayfield LJ, et al. Consensus statements and recommended clinical procedures regarding implant survival and complications. Int J Oral Maxillofac Implants.2004;19(suppl):150-154.
2. Lindhe J, Meyle J; Group D of European Workshop on Periodontology. Peri-implant diseases: consensus report of the sixth european workshop on periodontology. J Clin Periodontol. 2008;35(8 Suppl):282-285.
3. Derks J, Tomasi C. Peri-implant health and disease. A systematic review of current epidemiology. J Clin Periodontol. 2015;42(Suppl 16):S158–S171.
4. Pontoriero R, Tonelli MP, Carnevale G, et al. Experimentally induced peri-implant mucositis. A clinical study in humans. Clin Oral Implants Res. 1994;5(4):254-259.
5. Serino G, Ström C. Peri-implantitis in partially edentulous patients: association with inadequate plaque control. Clin Oral Implants Res. 2009;20(2):169-174.
6. Pranskunas M, Poskevicius L, Juodzbalys G, et al. Influence of peri-implant soft tissue condition and plaque accumulation on peri-implantitis: a systematic review. J Oral Maxillofac Res. 2016;7(3):e2.
7. Renvert S, Quirynen M. Risk indicators for peri-implantitis. A narrative review. Clin Oral Implants Res. 2015;26(Suppl 11):15-44.
8. Karoussis IK, Salvi GE, Heitz- Mayfield LJ, et al. Long-term implant prognosis in patients with and without a history of chronic periodontitis: a 10-year prospective cohort study of the ITI dental implant system. Clin Oral Implants Res. 2003;14(3):329-339.
9. Staubli N, Walter C, Schmidt JC, et al. Excess cement and the risk of peri-implant disease—a systematic review. Clin Oral Implants Res. 2017;28(10):1278-1290.
10. Brunski JB, Puleo DA, Nanci A: Biomaterials and biomechanics of oral and maxillofacial implants: current status and future developments. Int J Oral Maxillofac Implants. 2000;15(1):15-46.
11. Giovannacci I, Meleti M, Manfredi M, et al. Medication-related osteonecrosis of the jaw around dental implants: implant surgery-triggered or implant presence-triggered osteonecrosis? J Craniofac Surg. 2016;27(3):697-701.
12. Lang NP, Berglundh T, Working Group 4 of the Seventh European Workshop on Periodontology. Periimplant diseases: where are we now?—Consensus of the seventh european workshop on periodontology. J Clin Periodontol. 2011;38(Suppl 11)178-181.
13. Ericsson I, Lindhe J. Probing depth at implants and teeth. An experimental study in the dog. J Clin Periodontol. 1993;20(9): 623-627.
14. Muthukuru M, Zainvi A, Esplugues EO, et al. Non-surgical therapy for the management of peri-implantitis: a systematic review. Clin Oral Implants Res. 2012;
23(Suppl 6):77-83.
15. Mailoa J, Lin GH, Chan HL, et al. Clinical outcomes of using lasers for peri-implantitis surface detoxification: a systematic review and meta-analysis. J Periodontol. 2014;85(9):1194-1202.
16. Chan HL, Lin GH, Suarez F, et al. Surgical management of peri-implantitis: A systematic review and meta-analysis of treatment outcomes. J Periodontol. 2014;85(8):1027-1041.
17. Lang NP, Mombelli A, Tonetti MS, et al. Clinical trials on therapies for peri-implant infections. Ann Periodontol. 1997;2(1):343-356.