Dental Implant Hybrids for Maximum Esthetics
A next-generation option for implant-retained prostheses
Patients are generally aware of their dental problems when they present to their clinician's office. Part of the clinician's job is to keep up with the newest and best techniques and to educate patients about the benefits and risks of available procedures. Implant dentistry, which has become a routine and effective means for restoring edentulous spaces in patients, is a prime example. As engineering principles are better understood and surgical and prosthetic components are made more reliable, the resulting successes are astounding.
Fixed all-ceramic implant prosthetics are quickly becoming a viable option for restoration of edentulous patients. These esthetic, functional, and stable devices provide increased chewing ability, exceptional wear resistance, and better speech due to their minimal palatal coverage.1 As material availability and computer technology have improved, allowing dentists to mill precise and esthetic prostheses, the conventional hybrid has evolved into the solid zirconia bridge (eg, BruxZir®, Glidewell Laboratories, www.glidewelldental.com).2 The highly esthetic final result can be screw-retained or cemented over custom-aligned abutments and resembles a conventional bridge more than a conventional denture. In the author's experience, patient response has been extremely positive in every aspect.
Dentists' diagnostic treatment planning is limited by the viable anatomy presented. Large maxillary sinuses may inhibit placement of dental implants in the posterior maxilla without more involved surgical intervention such as sinus augmentation procedures. Proper engineering of the final prosthesis requires well-thought-out implant position and number. Arch form is considered to maintain long-term stability.3 The advent of cone-beam computed tomography (CBCT) diagnosis has aided the practitioner in determining the type, number, position, and angulation of potential implants.
Case Presentation
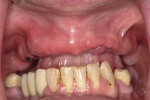
A 48-year-old white woman presented to the author's office with some maxillary teeth that were functionally compromised with poor long-term prognoses. Her transitional maxillary removable appliance was difficult to wear and did not provide a positive quality of life.
The plan was to stage her restoration and the remaining non-restorable maxillary teeth were atraumatically removed and a conventional maxillary complete denture was fabricated. After a 4-month period, the realization was made that the denture was not acceptable long term, as the patient preferred a fixed option. We then diagnosed and planned for a six-implant retained fixed prosthesis. The maxillary sinuses were rather large in the posterior, so the implants were digitally planned for ideal function and esthetics.
Planning and Implant Placement
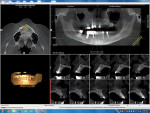
A preoperative CBCT scan (i-CAT, Imaging Sciences International, LLC, www.i-cat.com) was taken to help determine the potential positioning of dental implants and evaluate the significant anatomy, such as the maxillary sinus, as well as the horizontal and vertical bone available (Figure 1). Per the diagnosis, the remaining maxillary teeth were scheduled to be surgically and atraumatically removed, and immediate implants were to be placed. Immediately following extraction of the remaining maxillary teeth, an immediate complete denture was fabricated and seated (Figure 2).
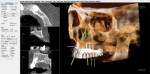
Figure 3 illustrates the CT diagnosis and proper positioning of six maxillary dental implants within the viability of the existing anatomy using software (Invivo 5, Anatomage Dental, www.anatomage.com). Implants (3.7-mm diameter Glidewell Inclusive tapered implants) were strategically placed essentially parallel and away from the sinus areas in the edentulous maxilla as determined by CT planning. A surgical guide was created to allow for initial depth determination. Subsequent increase in diameter of the surgical burs created the osteotomy site for the implants. For this patient, implants were positioned in the edentulous areas of position Nos. 4 through 6 and 11 through 13. They were torqued to 25 Ncm, flat cover screws were placed, and the existing maxillary conventional complete denture was seated. A Mucopren® Soft liner (Kettenbach, www.kettenbach.com) was used to condition the tissue during the 4-month integration period.
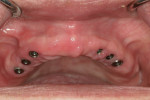
After the healing period, the arch is examined prior to exposing the flat implant-healing abutment. The healthy tissue responded well to the Mucopren Soft liner, which was functional during the entire 4-month period (Figure 4). After integration, the implants were exposed using the DEKA Smart CO2 laser (Implant Direct, www.implantdirect.com) to minimize tissue damage.
Impression and Seating
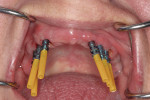
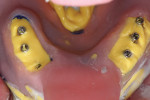
Open-tray impression copings are placed within the internal hex of the six implants (Figure 5). The long yellow sleeves Glidewell Laboratories provides with its open-tray impression copings prevent any material from engaging the inside of the seating screws. The initial impression was completed using a medium-body flowable material around the dental implants along with a heavy body putty material (Kettenbach) (Figure 6). This firm polyvinylsiloxane material provides a reliable tool to create the initial implant cast.
Conventional denture techniques using bite rims establish jaw relations including vertical dimension of occlusion and centric relation. Denture teeth were set to please the patient's desired shade and tooth arrangement for esthetics and speech function. The clinician and patient approved the wax try-in.
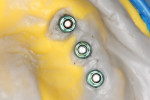
An important next step in this process is the fabrication of a verification jig. This tool is created by the technician from the initial master cast and then numbered and sectioned and ensures a passive fit of the final prosthesis.
The acrylic pieces of the section jig (Figure 7) were seated using titanium cylinders, which passively rest into the implant body intraorally. Pieces not in contact can be luted together with an acrylic resin (eg, PATTERN RESIN™, GC America, www.gcamerica.com) or similar material.
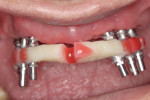
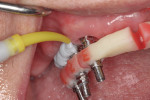
The entire luted verification jig was then picked up with a custom tray fabricated by the dental laboratory using an open-tray approach. The medium-body flowable material was injected around the verification jig, and heavy body putty was used to pick up the entire jig (Figure 8). This allows for fabrication of a passive and extremely accurate master cast from this final impression. Open-tray screws pass through the material, allowing for easy removal (Figure 9). Figure 10 illustrates the clean, crisp, accurate impression made using the medium-body (gray) and heavy-body putty (yellow material). The seating screws were loosened through the open tray, and the final impression was extremely accurate. As with all implant procedures, whenever metal-to-metal components touch, an accurate digital radiograph should be made to ensure a complete seat.
Provisional and Final Fabrication
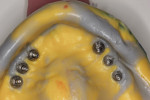
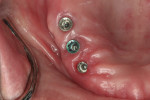
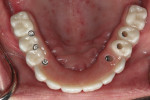
The dental laboratory next created a very precise working master cast. The final wax try-in setup was again tried in the mouth to correct any minor potential discrepancies in esthetics or occlusion (Figure 11 and Figure 12). Figure 13 documents the healing abutments in position prior to the placement of subsequent prostheses. Note the healthy tissue cuff in Figure 14, which was created by ideal implant placement, healing abutment positioning, and the use of a high-quality tissue conditioner. These steps are important prior to the fabrication of the CAD/CAM–created dental implant prosthesis.
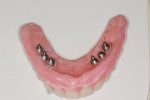
Glidewell creates a provisional appliance that will serve as a transitional appliance to allow the patient a trial period to evaluate the definitive prosthesis before the final BruxZir monolithic zirconia hybrid is fabricated (Figure 15). If no adjustments are made in the provisional, the laboratory can be asked to make the final zirconia hybrid, and the patient can wear the provisional. However, if any adjustments are needed or made, the provisional must be returned to the lab with the new bite relations and the healing abutments and the old temporary conventional denture is returned.
Final Seating
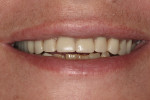
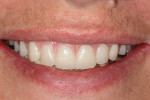
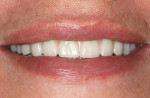
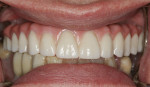
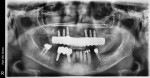
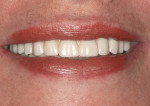
Once the dentist and the patient were satisfied, the final BruxZir hybrid was seated and the seating screws were torqued to 25 Ncm as per manufacturer's directions (Figure 16 and Figure 17). A small piece of cotton was placed into the screw hole and composite was placed. A final panoramic radiograph was taken to ensure a complete and proper fit (Figure 18). Figure 19 shows the beautiful, lifelike esthetics of the patient's new smile.
Conclusion
Patients have become very sophisticated in determining the type of dental therapy they require. Options may include a conventional denture, several implants, and a removable palate-less implant-retained overdenture. When a patient requests some type of fixed appliance, many implants are considered, abutments are prepared, and the bridge or bridges are created, either using conventional techniques or wax and cast, or they can be CAD/CAM designed. When the anatomy does not allow many implants to be placed properly to support a conventional implant-retained bridge, the techniques of hybrid fabrication can be considered.
In the past, hybrids could be maintained over a minimum of four implants as long as they were spaced along the arch properly to allow for engineered support of the distal cantilever. The use of implants that angle away from the posterior maxillary sinus position and angle abutments allowed this design to be achieved. This type of hybrid uses a CAD/CAM-designed bar, and the process is similar to that described above. High-quality denture teeth with an acrylic base over the CAD/CAM titanium bar are used.2
The next step up from the traditionally designed screw-retained hybrid is the CAD/CAM-created BruxZir monolithic zirconia bridge. This type of prosthesis allows for outstanding wear and elimination of chipping, breaking, or fracturing of any acrylic base or denture tooth.4 The BruxZir bridge is created from solid zirconia and is attached to the implants through titanium copings. This bridge design is an outstanding solution and provides a wonderful functional and esthetic solution to edentulous arch reconstruction.
About the Author
Timothy Kosinski, DDS
Adjunct Clinical Professor
University of Detroit Mercy School of Dentistry
Detroit, Michigan
Private Practice
Bingham Farms, Michigan
References
1. Gallucci GO, Doughtie CB, Hwang JW, et al. Five-year results of fixed implant-supported rehabilitations with distal cantilevers for the edentulous mandible. Clin Oral Implants Res. 2009;20(6):601-607.
2. Harris D, Höfer S, O'Boyle CA, et al. A comparison of implant-retained mandibular overdentures and conventional dentures on quality of life in edentulous patients: a randomized, prospective, within-subject controlled clinical trial. Clin Oral Implants Res. 2013;24(1):96-103.
3. Awad MA, Locker D, Korner-Bitensky N, Feine JS. Measuring the effect of intra-oral implant rehabilitation on health-related quality of life in a randomized controlled clinical trial. J Dent Res. 2000;79(9):1659-1663.
4. Limmer B, Sanders AE, Reside G, Cooper LF. Complications and patient-centered outcomes with an implant-supported monolithic zirconia fixed dental prosthesis: 1 year results. J Prosthodont. 2014;23(4):267-275.