Current Implant and Abutment Design: A Team Approach
Surgical and restorative teams evaluate benefits for hard and soft tissues
Just as a root and its position affect the bone levels and gingival contours in the natural dentition, factors of abutment shape, subgingival restoration contour, and restorative materials have a significant impact on gingival tissue form and color surrounding implant restorations. With this in mind, remember that a successful restorative outcome begins in the surgical treatment phase. An ideal outcome can be achieved only through harmony of the tissue and the restoration.
Where it is not possible to idealize the tissue framework due to preoperative anatomic limitations, the restorative dentist and technician are under greater pressure to compensate for these deficiencies. This emphasizes the importance of comprehensive treatment planning by the surgical–restorative team and underscores the fact that surgical decisions made by the periodontist as well as the skill, methodology, and materials used will create the foundation for prosthetic success or failure.
Similarly, the prosthetic handling, including restoration and abutment design, cementation, and occlusion, can have an important impact on long-term peri-implant tissue health and stability.1,2 With high implant success rates reported in the literature, the issue of integration has become secondary, with numerous metals exhibiting favorable responses in bone.3 Current focus has shifted to exploring strategies to enhance primary implant stability, the speed at which osseointegration occurs, enhanced bone-to-implant contacts, strategies to reduce crestal bone remodeling, and designs and treatment protocols aimed at idealizing soft tissue volume, color, and contour.
Impact of Implant Design on Primary Stability
Surgical decisions regarding implant size (ie, length/width) and bone density–based site preparation protocols (ie, press-fit or undersizing the osteotomy) are important for the goal of optimizing initial implant stability; however, implant design also influences the short- and long-term successes of implants.4 Although no one optimal implant design exists, macrogeometry features that allow for superior initial stability open opportunities for implant placement at the time of extraction into less favorable bone quality and early or immediate restoration protocols. Implant design can influence initial or primary stability; tapered implant designs significantly improve initial or primary stability as assessed by insertion torque over traditional parallel-walled implants, by mechanically enhancing implant stability through a wedging effect. Also, thread design, pitch, angle, depth, and width, as well as the crestal module (microthreads), can influence the stability of an implant by affecting the distribution of stress forces around it. Decreased thread pitches have been shown to influence primary stability, and deeper threads have been positively linked to enhanced stability in poorer bone quality. Introduction of progressive thread designs (ie, graduated from the collar to the apex) can address multiple requirements; those at the crest are designed to engage cortical bone, while those at the apex are designed to cut or guide the implant during insertion.5 These design features, coupled with surgical technique and implant size, can facilitate implant placement in sites with poorer bone quality, thus improving primary stability, potentially increasing the surface area for osseointegration, and possibly enabling the clinician to consider early placement and restoration protocols.
Impact of Surface Treatments
Due to efforts to improve integration or bone-to-implant contacts, newer surface treatments have been developed. The introduction of moderately rough implant surfaces, generally random patterns, increase the implant surface area, thus increasing the potential for enhanced bone-to-implant contacts.6,7 Most of these surfaces are generated through some type of acid-etching or various blast treatments, whereas others are created using additive processes (TiUnite®, Nobel Biocare, www.nobelbiocare.com) and historically with rougher surfaces (eg, titanium spheres, titanium plasma-sprayed, hydroxylapatite). Moderately rough surfaces are particularly important in sites with inferior bone quality (type III to type IV), when placed in previously grafted sites, and when placing short implants (reduced surface or contact area). The osseointegration rate might be influenced by surface chemistry (addition of specific ions) or activity (hydrophilicity). At the time of this writing, no clear scientific evidence suggests a superior specific surface treatment. Some implant designs have eliminated the smooth surface at the collar in favor of the bone-retentive features of moderately rough surfaces, although some clinicians still favor the smooth collar when placed at or coronal to the ridge crest; this is due to concerns over potential microbial contamination of the rough surface, and the possible impact on peri-implant health, despite literature that indicates clinical and microbial outcomes do not differ between minimally and moderately rough implants.8,9
Impact of Microthreads
Marginal bone change occurs as a result of multiple etiologic factors, including remodeling due to the position of the implant platform relative to the ridge crest, the inflammatory cell infiltrate at the microgap, stability of the connection, and repetitive prosthetic manipulations. Important for long-term and predictable esthetic outcomes, features to enhance crestal bone stability and gingival tissue architecture have been incorporated into implant collar designs, including the addition of bone-retentive collar microgrooves. These have resulted in reduced crestal bone remodeling compared with implants that have standard threads. Microgrooves may mitigate the formation of the biologic width by increasing the surface area at the implant collar. This is particularly important in situations where adjacent implant placement is necessary and the advantages of teeth in supporting supracrestal soft tissues are lost.
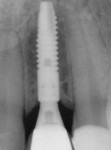
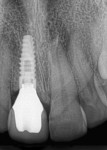
Examples of unique microgrooves such as Astra Tech Implant (DENTSPLY Implants, www.dentsplyimplants.com) with Micro Threads™ and their impact on crestal bone stability are illustrated by the following radiograph in Figure 1 and micro/nanostructure surface treatments such as Laser-Lok®, laser-ablated microgrooves at the implant-abutment junction (BioHorizons, www.biohorizons.com) in Figure 2. Laser-ablated surfaces are intended to provide a substrate that functionally guides both bone and connective tissue, by influencing both fibroblast and osteoblast proliferation partly through unique contact guidance.10 Several radiographic-outcome reports illustrate crestal bone-retention on implants with Laser-Lok microchannels.11,12 Histologic evidence suggests that laser-etched grooves at the implant collar result in a functionally orientated connective tissue and attachment to the implant collar. Clinically, this may result in superior soft tissue anatomy and tissue levels and a more effective mucosal seal. This should be advantageous over the soft tissue cuff that typically forms around an implant abutment, where connective tissue fibers are aligned more parallel to the implant and restorative interface.13
Impact of Platform Switching or a Horizontal Abutment Offset
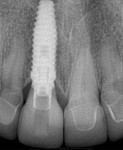
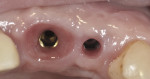
Equally important in the effort to reduce crestal bone changes are improved implant-abutment connections that are hermetic and reduce the effect of micromotion on bone. Some implant connections potentially create bacterial reservoirs that will negatively influence tissue health and stability. In addition to internal connection designs that have been shown to be advantageous over external connections, significant interest has been generated in the concept of horizontal offsetting of the abutment–implant interface, commonly referred to as platform switching (Figures 3 and Figure 4). Support for reduced crestal bone remodeling by medializing the microgap or distancing this junction from the bone surface is largely based on animal studies, with limited human trials.14,15 What is not emphasized about the platform-switch design is the benefit of increased space for soft tissue development at the implant collar. A horizontal offset potentially allows for a soft tissue volume gain at the crest early in the healing process. Again, animal histologic studies support this, although little to no evidence from human clinical trials corroborates it. Figure 5 contrasts the tissue volume around two size-matched implants, one with and one without a horizontal offset. Periodontists trained in a variety of grafting procedures to treat recession recognize that tissue volume and quality can affect tissue stability. Where bone is thin or deficient, enhancing soft tissue volume becomes an important strategy for shielding or reducing hard and soft tissue changes. Extrapolating from this, the reader can presume the soft tissue volume gain may have a protective effect on bone in the case of implants with a horizontal offset.
Setting the Framework for Success with the Provisional Restoration
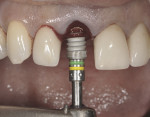
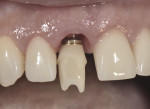
Bone and soft tissue stability can also be influenced by subtle alterations in restorative treatment protocols. Zirconia and, to some extent, lithium disilicate and gold-hued titanium abutments are routinely used in the esthetic zone to better control soft tissue color and to position abutment margins more coronally for ease of cementation. Coupled with this, carefully planned abutment designs initiated in the provisional phase of treatment and duplicated in the definitive restoration design can enhance marginal tissue health and esthetics; concave subgingival restoration contours increase the soft tissue volume adjacent to the implant collar and bone crest, while positioning the abutment margin as coronally as possible in the case of a cemented restoration, reduces the risk for retained subgingival cement. Figure 6 illustrates an ideal provisional restoration contour that helps develop the soft tissue profile (as seen in Figure 7)and will act as a template for the definitive restorative subgingival prosthetic contour.
Impact of the Abutment: Treatment and Handling
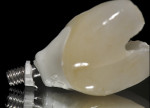
Recent studies similarly indicate that laser etching of abutments could potentially allow soft-tissue adhesion or connective tissue fiber in-growth to the abutment microchannels, thus further stabilizing the soft tissue (Figure 8 and Figure 9).16,17 This abutment surface treatment may improve crestal bone stability by supporting a more robust soft tissue barrier. Extrapolating from this, one might speculate that although peri-implant bone loss has a multifactorial etiology, microbial colonization of a roughened surface and the development of inflammatory cell infiltrate at the implant-abutment microgap could be reduced through a more effective soft tissue barrier. Limiting prosthetic manipulation of the implant–abutment interface has been suggested to minimize disruptive events that impact the epithelial and connective tissues at the abutment level and, in turn, affect the crestal bone levels, although the supporting evidence for this is weak.18
Abutment handling is rarely discussed in the literature but is an important consideration in establishing tissue health and fostering a stable relationship between the abutment and epithelium, and connective tissue in the case of abutments with lased components. Placing “clean” abutments at the time of surgery and upon insertion of the definitive restoration is important; how this is achieved is not clearly defined in current clinical protocols. It seems logical to consider steam cleaning prior to insertion of a restorative material, but how surface treatments change the biocompatibility of the restorative material has been poorly described. For instance, polished surfaces may have remnants of pastes that could interfere with the development of a soft tissue attachment at the abutment level. Some evidence suggests that polishing zirconia will negatively affect the soft tissue response and “as-milled” surfaces have superior biocompatibility concerning the adherence and colonization of fibroblasts.19
Abutment design considerations should focus not only on the transmucosal tissue material, but also on the connection material itself. The illustrative case indicates that either due to imprecision of milling or micromotion, internal deformation of the implant can occur as evidenced by metallic discoloration on retrieved abutments. More clinically impacting is the risk for abutment fracture at the level of the stem, resulting in catastrophic failure of the prosthesis. It seems rational to strongly advocate against the use of zirconia stems, as long-term survival of the restoration is at risk (Figure 10 and Figure 11).
Concluding Remarks
This brief review of current implant and abutment design features that are considered in today’s implant rehabilitations bears relevance to all types of restorations. Although one may focus on the benefits of hard and soft tissues from an esthetic perspective, enhanced marginal bone stability should also be viewed as an important goal for long-term implant and tissue health. Unique and challenging clinical situations such as poor bone quality, reduced ridge height, and grafted areas demand the use of new surfaces and implant shapes that improve both initial and secondary stability. Although many abutment materials are available, ranging from titanium, gold-anodized coatings to zirconia and lithium disilicate, their design and handling can significantly contribute to a healthy and stable soft tissue response. It remains important to emphasize that implants and abutments are merely the tools, and that the clinician’s treatment planning prowess and level of skill will have the most impact on the treatment outcome. When planning the team approach to treatment, the periodontist must have a clear vision as to the desired restorative outcome, including final materials selection, whereas the restorative dentist should have an understanding of the surgical decisions and limitations to ensure the best possible outcome. Never in the short history of highly predictable esthetic implant treatment for our patients has this collaboration been of more importance.
Acknowledgment
The author would like to thank and acknowledge the significant collaboration and contribution of prosthodontist Dr. Brahm Miller and the excellent technical work by technicians Nico Pienaar and Klaas van der Walt.
Disclosure
Dr. Sonia Leziy is a paid consultant for BioHorizons.
References
1. Leziy S, Miller B. Esthetics in implant therapy: a blueprint for success. In: Cohen M, ed. Interdisciplinary Treatment Planning: Principles, Design, Implementation. Hanover, IL: Quintessence Publishing Co, Inc. 2008.
2. Leziy S, Miller B. The papilla between adjacent implants: treatment planning to optimize aesthetic outcomes. In: Cohen M. Interdisciplinary Treatment Planning, Volume II: Comprehensive Case Studies. Hanover, IL: Quintessence Publishing Co, Inc. 2011.
3. Roos J, Sennerby L, Lekholm U, et al. A qualitative and quantitative method for evaluating implant success: a 5-year retrospective analysis of the Brånemark implant. Int J Oral Maxillofac Implants. 1997;12(4):504-514.
4. Elias CN, Rocha FA, Nascimento AL, Coelho PG. Influence of implant shape, surface morphology, surgical technique and bone quality on the primary stability of dental implants. J Mech Behav Biomed Mater. 2012;16:169–180.
5. Abuhussein H, Pagni G, Rebaudi A. Wang HL. The effect of thread pattern upon implant osseointegration. Clin Oral Implants Res. 2010;21(12):129-136.
6. Valencia S, Gretzer C, Cooper LF. Surface nanofeature effects on titanium-adherent human mesenchymal stem cells. Int J Oral Maxillofac Implants. 2009;24(1):38-46.
7. Wennerberg A, Albrektsson T. Effects of titanium surface topography on bone integration: a systematic review. Clin Oral Implants Res. 2009;20(Suppl4):172-184.
8. Van Assche N, Coucke W, Teughels W, et al. RCT comparing minimally with moderately rough implants. Part I: clinical observations. Clin Oral Implants Res. 2012;23(5):617-624.
9. Quirynen M, Van Assche N. RCT comparing minimally with moderately rough implants. Part 2: microbial observations. Clin Oral Implants Res. 2012;23(5):625-634.
10. Pecora GE, Ceccarelli R, Bonelli M, et al. Clinical evaluation of laser microtexturing for soft tissue and bone attachment to dental implants. Implant Dent. 2009;18(1):57-66.
11. Shapoff CA, Lahey B, Wasserlauf PA, Kim DM. Radiographic analysis of crestal bone levels around Laser-Lok collar dental implants. Int J Periodontics Restorative Dent. 2010;30(2):129-137.
12. Botos S, Yousef H, Zweig B, et al. The effects of laser microtexturing of the dental implant collar on crestal bone levels and peri-implant health. Int J Oral Maxillofac Implants. 2011;26(3):492-498.
13. Nevins M, Nevins ML, Camelo M, et al. Human histologic evidence of a connective tissue attachment to a dental implant. Int J Periodontics Restorative Dentistry. 2008;28(2):111-121.
14. Becker J, Ferrari D, Herten M, et al. Influence of platform switching on crestal bone changes at non-submerged titanium implants: a histomorphometrical study in dogs. J Clin Periodontol. 2007;34(12):1089-1096.
15. Schwarz F, Alcoforado G, Nelson K, et al. Impact of implant-abutment connection, positioning of the machined collar/microgap, and platform switching on crestal bone level changes. Camlog Foundation Consensus Report. Clin Oral Implant Res. 2013:1-3.
16. Nevins M, Kim DM, Jun SH, et al. Histologic evidence of a connective tissue attachment to laser microgrooved abutments: a canine study. Int J Periodontics Restorative Dent. 2010;30(3):245-255.
17. Nevins M, Camelo M, Nevins ML, et al. Connective tissue attachment to laser-microgrooved abutments: a human histologic case report. Int J Periodontics Restorative Dent. 2012;32(4):385-392.
18. Berglundh T, Lindhe J, Ericsson I, et al.. The soft tissue barrier at implants and teeth. Clin Oral Implants Res. 1991;2(2):81-90.
19. Mustafa K, Wenerberg A, ArvidsonK, et al. Influence of modifying and veneering the surface of ceramic abutments on cellular attachment and proliferation. Clin Oral Implants Res. 2009(19):1178-1187.
About the Author
Sonia S. Leziy, DDS, Dipl Perio, FCDS(BC), FRCD(C)
Private Practice, Imperio Group Dental Health Specialists
North Vancouver, British Columbia, Canada
Clinical Associate Professor
University of British Columbia
Vancouver, British Columbia, Canada