Verifiable Outcomes Using Salivary Diagnostics in Periodontal Medicine
Test and retest for clinical success.
Salivary diagnostics and the ability to use the saliva as a diagnostic medium are creating tremendous opportunities in dentistry.1,2 A well-defined periodontal policy incorporating salivary diagnostics in the diagnosis, treatment, and maintenance of periodontal disease creates opportunities for healthier patients.
Key features of a periodontal policy are standardized criteria for disease identification, the use of salivary diagnostics to determine the causative pathogens, evaluation of innate and environmental risk factors that may modify the host response, and the development of a personalized plan to control the bacterial load and manage the disease. Once the disease is stabilized, it is suggested that salivary diagnostics be used to measure bacteria profiles against the original profile of the active disease. In other words, the activity of the infection should be assessed over time.
Studies conservatively estimate that 50% of American adults have some form of periodontal disease.3 Healthcare professionals are learning that recognizing and treating the periodontal infection early, identifying the causative pathogens, evaluating the immune response, and using a total-mouth disinfection strategy has dramatic effects on the gingival health of patients. In addition to reducing the risk the disease poses to the teeth, reducing oral inflammation may provide general health benefits, as there is abundant research that supports concern for the effects of periodontal disease on overall systemic health.4
Several studies suggest that bacteria specific to periodontal disease may be a link to certain forms of cardiovascular disease, and actually play a role in management of diabetic glycemic control.5-12 This article will demonstrate the use of salivary diagnostics in disease diagnosis and in disease reassessment to measure bacterial load control.
The Opportunity
In medical science, health professionals learned 150 years ago from Louis Pasteur that to manage disease one needs to identify causative pathogens in the disease process and target them with appropriate therapy.13 In dentistry, however, it is common to treat periodontal disease without knowing the pathogenic profile of the patient. This approach is similar to the empirical use of broad-spectrum antibiotics used by physicians for conditions such as sinusitis. However, directed therapy against known organisms with known antibiotic sensitivity produces better disease management outcomes.14
Salivary diagnostics creates a tremendous opportunity for earlier diagnosis, better treatment outcomes, and better health management. In other words, salivary diagnostics provides an opportunity for personalized periodontal medicine.
Disease Diagnosis
Current diagnostic criteria includes recording of six-point probing, notation of bleeding on probing, recession, clinical attachment loss, tooth mobility, anatomic liabilities, and radiographic analysis of bone levels.15 In addition to clinical observations, both innate and environmental risk factors should also be considered in the disease diagnosis.
When diagnosing active disease, dentists rarely look at the microbes responsible for the initiation of the disease. Currently, there are several identification methods in existence: anaerobic culturing, phase-contrast microscopy (for identifying morphology), enzyme testing (BANA), and a laboratory process called polymerase chain reaction (PCR) to detect bacterial DNA. Even though these methods lend clarity about the bacterial makeup, these processes have presented both implementation and clinical challenges.
Difficulty in culturing what comes from the periodontal pocket, false negatives, shipping obstacles, expense, and the general belief that traditional culturing would usually result in the recommendation of amoxicillin and metronidazole have kept these tests from mainstream use.
Periodontal disease management is getting easier. Now, with OralDNA® Labs (www.oraldna.com), and specifically the MyPerioPath® and MyPerioID PST® tests, dentists can use saliva that contains gingival crevicular fluid and bacteria to learn the microbial and genetic information specific to each individual to further personalize the disease and treatment. This testing process uses PCR DNA analysis. A 30-second swish and spit test provides information that produces many benefits. The test results provide:
Confirmation of the presence of and the concentration of high-, moderate-, and low-risk periodontal pathogens (the bacterial makeup of the disease) so that preferred antibiotic(s) to treat the infection can be targeted to specific pathogens.
A reference to understand the correlation between clinical observation and objective data in disease identification and reevaluation.
A systematic way to make earlier and more targeted disease management decisions.
The patient’s genetic susceptibility to periodontal infections and, potentially, to other inflammatory conditions (this genetic test is offered separately).
Identification of preferred antibiotics is one of the most valuable benefits of the test. Without pathogen information, dentists commonly use the amoxicillin/metronidazole combination except where the patient is allergic. Unpublished data from private practice using DNA PCR testing for periodontal disease demonstrates that the combination of amoxicillin and metronidazole is suggested approximately 26% of the time on the MyPerioPath® report. This limits patients’ exposure to excessive antibiotics and prevents the unnecessary use of antibiotics that are not ideal for the given situation.
In addition to the above benefits, this test is easy to administer and the resulting report is a visual, non-biased scientific document validating the disease process for the patient. This is a huge benefit for treatment acceptance over other in-office analytic modalities.
Periodontal Policy
Currently, there are no profession-wide standardized active disease criteria other than ongoing attachment loss. This has created confusion regarding when disease should be diagnosed. Because clinical appearance is not enough to diagnose disease, it is advised that office teams develop or adopt a set of criteria to identify when active disease is imminent. Bleeding on probing is clearly not the gold standard for disease identification, but until something more scientific and reliable comes along, it is used as an indication of inflammation.
As part of a preferred periodontal policy, when four or more bleeding areas are discovered, show patients the bleeding and enter into a brief discussion about inflammation and the potential risk to the teeth and to systemic health. This begins the patient education process. Language such as, “Joe, you are bleeding more than we are comfortable with and it is not normal,” is usually enough to get the conversation started. In addition, dentists may include, “Bleeding is usually caused by your system’s response to bacteria living around the teeth under the gums. We have a simple test to determine what type of bacteria are present and their concentrations. In addition, we discover the ideal antibiotic regimen to assist in bacteria control. Depending on the results of this test, perhaps treatment is needed to stabilize this infection.” Many patients will simply respond with a comment, “Then take the test.”
Having a written periodontal policy enhances the ability to detect disease earlier and implement effective treatment. The following case report demonstrates the clinical use of salivary diagnostics in the identification and treatment of periodontal disease.
Historical Data
The patient, a 56-year-old white man, had a history of localized scaling and root planing (SRP) around teeth Nos. 11, 14, and 15 in 2005 and 2007. Periodontal pockets measured 2 mm to 4 mm and, considering prior recession, maximum clinical attachment loss was 5 mm. There was no significant bleeding at the patient’s re-care visits.
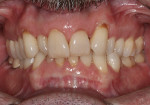
The patient’s medical history was remarkable. Periodontal risk modifiers were prior bone loss (< 20%), and smoking (1/2 pack daily for 30 years). In late 2008, active disease was diagnosed, represented by nine bleeding sites. However, the clinical presentation (Figure 1) did not show obvious inflammation. The patient’s radiographs represented bone loss (< 30%), Class I furcation involvement, and generalized moderate to clinically severe attachment loss, considering pocket depths and prior recession.
Upon the discovery of inflammation represented by bleeding, the office initiated a salivary diagnostic test from OralDNA, shown in Figure 2. Clearly, the test showed the presence of pathogenic bacteria with a large pathogen load. Four quadrants of SRP were recommended in a conventional manner using a combination of ultrasonic and hand scaling plus chlorhexidine as an irrigant. No other treatment modalities were used.
At the 7-week disease reevaluation, periodontal charting showed improvement in pocket depths and reduction in bleeding sites to five. Another test using MyPerioPath® was administered. The results after conventional SRP produced improvement in bleeding and pocket depth but affected the pathogen profile very little (Figure 3). This provoked the need to take a more comprehensive approach to disease treatment and employ a total-mouth disinfection methodology. This method addresses the following areas:
1. Occlusal analysis to identify and remove any fremitus in chewing.
2. Attack the bacteria in the pockets mechanically (using SRP).
3. Attack the bacteria in the pockets chemically by irrigating with CariFree Treatment Rinse (Oral BioTech, www.carifree.com), iodine, or chlorhexidine and depositing Arestin® (OraPharma, Inc., www.orapharma.com) a locally applied time-release antibiotic, in all pockets equal to or greater than 5 mm.
4. Attack the bacteria in the bloodstream with the antibiotic regimen suggested on the OralDNA report.
5. Attack the bacteria in the rest of the oral cavity with CariFree Treatment Rinse, ProFresh (ProFresh International Corp., www.profresh.com) or another rinse to control microbial load used for 1 minute twice daily.
6. Prescribe home care adjuncts such as a mechanical toothbrush, Waterpik® (Water Pik, Inc., www.waterpik.com) or Hydroflosser (Shazzam Tsunami™, Bling Dental Products, www.blingdentalproducts.com) and interproximal aids, etc., for enhanced bacteria load control.
7. Maintain 2-month re-evaluation appointments until stability (zero bleeding on periodontal chart) has been achieved.
After developing a method to be much more comprehensive (ie, a total mouth disinfection approach), treatment following the above protocol was reinitiated. The patient was kept on a tight 2-month re-evaluation schedule until zero bleeding was established. The patient was then retested using MyPerioPath® (Figure 4). With the use of a total-mouth disinfection approach, the microbial profile was altered through eradication, suppression, or alteration of the bacterial load. It is important to note this profile was measured when there was zero bleeding, suggesting gingival stability.
To summarize, incorporation of the MyPerioPath® test is easy. It provides pathogen information and the ability to measure the effectiveness of different approaches to treatment. These benefits greatly enhance oral health management.
Conclusion
It is understood that periodontal disease is initiated by bacteria.15,16 Knowing the bacterial profile of an infection is important for a number of reasons. To successfully stabilize the disease and realize health, certain pathogens in the periodontal biofilm need to be eradicated, suppressed, or altered. When dentists identify the causative bacteria in the biofilm, treatment can be personalized to target the root cause of the disease. Many dentists are using this information to help their patients become and stay well.
With a defined periodontal policy and the tools to provide an agreed-upon structured approach, the protocols outlined will guide dental professionals to identify, discuss and treat periodontal disease with success.
Information gained from the saliva positively influences the ability to create disease awareness, provide a platform for disease discussion, and improve treatment and disease control outcomes..
Disclosure
Dr. Thompson is a consultant for OralDNA® Labs.
References
1. Giannobile W, Beikler T, Wong T, et al. Saliva as a diagnostic tool for periodontal disease: Current state and future directions. Periodontology 2000. 2009;50:52-64.
2. Zimmerman, Par, Wong. Oral-based diagnostics. Annals of the New York Academy of Sciences. 2007;1098.
3. Eke PI, Thornton-Evans G, Dye B, Genco R. Advances in Surveillance of Periodontitis: The Centers for Disease Control and Prevention Periodontal Disease Surveillance Project. J Periodontol. 2012 Feb 11. [Epub ahead of print.]
4. Rose LF, Steinberg BJ, Minsk L. The relationship between periodontal disease and systemic conditions. Compend Contin Educ Dent. 2000;21(10A):870-877.
5. Beck J, Garcia R, Heiss G, et al. Periodontal disease and cardiovascular disease. J Periodontol. 1996;67(suppl 10):1123-1137.
6. Beck JD, Pankow J, Tyroler HA, et al. Dental infections and atherosclerosis. Am Heart J. 1999;138(5 pt 2):S528-S533.
7. Danesh J, Collins R, Peto R. Chronic infections and coronary heart disease: is there a link? Lancet. 1997;350:430-436.
8. Hujoel PP, Drangsholt M, Spiekerman C, et al. Periodontal disease and coronary heart disease risk. JAMA. 2000;284:1406-1410.
9. Mealey BL, Oates TW for the American Academy of Periodontology. Diabetes mellitus and periodontal diseases. J Periodontol. 2006;77:1289-1303.
10. Nelson RG, Shlossman M, Budding LM, et al. Periodontal disease and NIDDM in Pima Indians. Diabetes Care. 1990;13:836-840.
11. Loe H. Periodontal disease: the sixth complication of diabetes mellitus. Diabetes Care. 1993;16:329-334.
12. Committee on Research, Science and Therapy. American Academy of Periodontology. Diabetes and periodontal diseases [position paper]. J Periodontol. 2000;71:664-678.
13. Schwartz M. The life and works of Louis Pasteur. J Appl Microbiol. 2001;91:597-601.
14. Leszczynska A, Buczko P, Buczko W, Pietruska M. Periodontal pharmacotherapy—an updated review. Adv Med Sci. 2011;56(2):123-131.
15. Armitage GC, Research, Science and Therapy Committee of the American Academy of Periodontology. J Periodontol. 2003;74(8):1237-1247.
16. Socransky SS, Haffajee AD. Periodontal microbial ecology. Periodontol 2000. 2005;38:135-187.
About the Author
Douglas G. Thompson, DDS
Private Practice
Bloomfield Hills, Michigan