A Topical Anesthetic for Periodontal Procedures
Howard Golan, DDS
Dentists and hygienists have always searched for the least invasive, least painful techniques to provide their patients with the highest-quality care under the most comfortable circumstances. However, though pain-reducing materials have existed for many years, methods to deliver those materials have been limited.
Over the past 5 to 10 years, dentistry has increased its number of no-injection procedures. With the profession increasing its minimally invasive techniques, dissemination of information via the Internet, and increased use of lasers, no-shot procedures can be executed, and are being performed throughout the world.
The number of soft-tissue and periodontal procedures performed without injections has increased significantly. Many procedures, including traditional scaling, laser debridement, gingivectomy, and troughing for crown-and-bridge preparations, no longer require an injection. The advantages are numerous. Patients can be comfortable without the fear and discomfort associated with injections, they can go to work or social outings without concern of drooping lips or biting cheeks and, of course, there is the marketing advantage of patients associating the office with “painless, shotless” dental procedures.
For the procedures described previously, many topical formulations can be used that penetrate deeper, last longer, and are more effective overall. However, because these formulations are drugs and penetrate deeper into the mucosal membranes, the pharmacology and manufacturing of these anesthetics must be understood.
Topical anesthetics are available from two major sources, compounding pharmacies and dental product companies. Compounding pharmacies produce topicals by mixing together ingredients and shipping them directly to the purchaser. These anesthetics, when used properly, are effective, inexpensive, and easy to apply. However, concerns arise from their use. Drugs can have adverse effects and, if and when that happens, the practitioner has the potential to be questioned if the drug used is not a formulation regulated by the government. Furthermore, there is no confirmation of consistency in the manufacture of these products, thus the product one practitioner receives may not be the same as another clinician. Finally, many topicals contain a vasoconstrictor that can cause systemic effects, including tachycardia.
Cetacaine® (Cetylite Industries, Inc, Pennsauken, NJ), is a topical anesthetic that has been in existence since the 1960s. It comes in spray, liquid, and gel forms. For many years, practitioners have used the spray to anesthetize the soft palate in patients with a sensitive gag reflex. However, the liquid and gel can work well for soft-tissue procedures. Its formulation is unique in that its three active ingredients each have a specific purpose. Benzocaine is fast acting and shallow penetrating; butamben is intermediate penetrating and intermediate acting; and tetracaine is long acting and deep penetrating. In addition, Cetacaine is produced in a US Food and Drug Administration (FDA)-inspected facility and manufacturing process, and it is available from dental dealers. Thus, it is an alternative that is FDA regulated.
Topical anesthetics are effective on oral tissues because of the ability of mucosal membranes for absorption. Therefore, topical anesthetics are effective only on the mucous membranes to which they are applied. For periodontal procedures, the physical properties of the drug and an easy-to-use, cost-effective delivery system are required. If the anesthetic is too viscous or not viscous enough, the drug will not get to the target tissue. Unfortunately, some of the delivery systems on the market today are unnecessarily complicated and too expensive for consistent use in the dental office.
Cetacaine liquid has the perfect viscosity for periodontal procedures. This liquid has enough resistance to flow into the target tissue, but is viscous enough to not run all over the oral cavity. Furthermore, the manufacturer has developed an efficient, cost-effective delivery system that limits the potential for cross-contamination. An innovative cap design allows the practitioner to place any luer lock syringe onto the Cetacaine vial and draw into it the required amount for the periodontal procedure (the maximum dosage is 0.4 mL per office visit). This limits waste and makes for a mess-free delivery. A flexible, small tip is placed on the syringe and the Cetacaine is delivered into the pocket. The practitioner should wait between 30 and 60 seconds for maximum effectiveness, and then proceed with the procedure.
Cetacaine liquid also has been used effectively for laser frenectomies, laser curettage, troughing for crown-and-bridge, stage 2 implant uncovering, and other soft-tissue procedures.
Case example
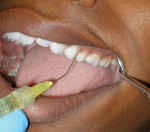
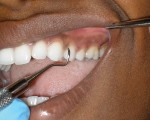
A 22-year-old woman was treatment planned for four quadrants of nonsurgical periodontal therapy. The patient had not been to the dentist in 3 years because of her fear of needles. Therefore, the periodontal treatment was performed with Cetacaine topical anesthetic instead of traditional injection. Cetacaine was drawn into a 1.2-mL syringe and attached to a unique and innovative luer lock cap on the bottle (Figure 1). A flexible tip was attached to the syringe (Figure 2) and delivered into each of the pockets in the quadrant (Figure 3). After 1 minute, the effective and fast-acting anesthetic took effect, and the patient was comfortable for the curettage, scaling, and laser procedures (Figure 4).
Conclusion
When used as directed, Cetacaine provides a safe, effective, and cost-effective alternative to other topical anesthetics on the market. Its pharmacology and history of use in the dental and medical professions allow practitioners to provide no-injection procedures with a drug they know is safe and effective. Cetacaine can enhance every dental practice and provide most patients with safe, comfortable soft-tissue procedures that are free of pain and the postoperative effects of injections.
Reference
1. Kravitz ND. The use of compound topical anesthetics. J Am Dent Assoc. 2007;138(10): 1333-1339.
For more information, contact:
Cetylite Industries, Inc
Phone: 800-257-7740
Web: www.cetylite.com
Disclaimer
The preceding material was provided by the manufacturer. The statements and opinions contained therein are solely those of the manufacturer and not of the editors, publisher, or the Editorial Board of Inside Dentistry. The preceding is not a warranty, endorsement, or approval for the aforementioned products or services or their effectiveness, quality, or safety on the part of Inside Dentistry or AEGIS Communications. The publisher disclaims responsibility for any injury to persons or property resulting from any ideas or products referred to in the preceding material.
About the Author
Howard Golan, DDS
Consultant
Cetylite Industries
Pennsauken, New Jersey