Dentsply Sirona is announcing its latest upgrade for both Connect and CEREC Software giving users both new and improved functionalities, and even better performance. Patient communication is also improved with a new visualization step in the model phase. New firmware makes the intraoral scanner twice as fast, provides a higher-performing scanning experience and gives users access to new workflows and even better usability*.
 The latest upgrade of the Connect and CEREC Software, 5.2, and an improved firmware provide Primescan and CEREC users with additional options for making treatment more efficient, faster, and more convenient with digital workflows. Primescan intraoral scanner’s latest enhancements optimize performance and improve usability as described as follows; Through its enhanced firmware, Primescan captures up to 1.5 million 3D data points per second. With these enhancements, Primescan is significantly smoother and twice as fast, making it again easier to scan successfully even for relatively inexperienced users*.
The latest upgrade of the Connect and CEREC Software, 5.2, and an improved firmware provide Primescan and CEREC users with additional options for making treatment more efficient, faster, and more convenient with digital workflows. Primescan intraoral scanner’s latest enhancements optimize performance and improve usability as described as follows; Through its enhanced firmware, Primescan captures up to 1.5 million 3D data points per second. With these enhancements, Primescan is significantly smoother and twice as fast, making it again easier to scan successfully even for relatively inexperienced users*.
CEREC Software 5.2: New features and benefits
Important, user-friendly changes and enhancements have been introduced in CEREC Software 5.2. In addition to general quality improvements, CEREC Primemill users can benefit from the availability of Fast Milling mode for crowns made of IPS e.max® CAD (Ivoclar Vivadent). Additionally, posterior and anterior crowns, direct screw-retained crowns, inlays and veneers can now regularly be produced using the Extra Fine grinding mode to create very smooth and very detailed restorations.
On top of viewing the model directly without restoration selected to facilitate communication with patients, the entire CEREC Guide workflow has also undergone improvements. The mode "Full Range Dynamic Occlusion" was introduced for the articulation calculation. This includes a wider range of lower jaw movement in the calculations and thus helps to detect more functional occlusal interferences*. Chairside dentistry therefore takes another step forward with the CEREC Software 5.2.
Primescan: validated for Atlantis Suprastructures
The software enhancement also opens new possibilities for users around edentulous scanning. Based on the validation of the Atlantis IO FLO-S scanbody, Primescan is now the first and only intraoral scanner validated for a full digital workflow for full arch fixed implant restorations with Atlantis suprastructures– a workflow for implant-supported restorations that requires a high level of accuracy.
The suprastructures are ordered by the dental technician from Dentsply Sirona's Atlantis design and manufacturing sites around the globe. “Usually, Primescan for Atlantis Suprastructures not only reduces chair time and simplifies collaboration with my laboratory, but also increases the level of esthetic predictability and a very good fit,” said Dr. Carlos Repullo, a beta tester practicing in Spain.
Additionally, primary impression taking within the Digital Denture workflow to create an individual, customized impression tray can now be performed with Primescan. This regularly offers the patient a more comfortable start into the manufacturing process of their prosthesis.
Connect SW 5.2 now supports bridges on implants
This Connect software update brings new possibilities to the implant workflows. Due to Primescan’s very high level of accuracy, dentists can now perform long span implant supported bridges, for example “all on 4” or “all on 6” cases, depending on the individual case. This means a new indication for practitioners working with Connect Software.
“Primescan firmware enhancements make the digital workflow even faster and easier than before*,” explained Dr. Alexander Voelcker, Group Vice President of Global Digital Solutions Marketing at Dentsply Sirona. “In addition to significantly increased speed, the smoothness of the scan has also been improved*. For clinicians working with Primescan in their practice, these improvements regularly mean more efficient workflows, even greater reliability, and, at the same time, a very comfortable patient experience.”
Learn more about Primescan and connect with dental industry professionals at Dentsply Sirona World 2021
This year’s Dentsply Sirona World will be a three-day event in Las Vegas from September 23-25. The event comprises General Sessions, a live surgery, Breakout Sessions within 12 educational tracks, an exhibit hall, celebrity entertainment, health and wellness sessions, and numerous networking opportunities for the dental community. This year will also have a virtual registration option to access the event virtually, as well as an option to live stream the daily general sessions and live surgery for free to maximize the opportunities to reconnect.
Onsite participants will have the exciting opportunity to hear from inspirational speakers live, including Mel Robbins, whose popular TEDx talk has been viewed more than 25 million times, and be treated to three evenings of unforgettable entertainment, including a concert from this year’s headliners, the DJ duo The Chainsmokers, a Grammy-winning electronic music and production duo.
Clinical education is paramount to empowering dental professionals to provide the best possible care to their patients, and our extended program will combine product demonstrations and experiences from experts from all over the world. Dentsply Sirona World 2021 is the ultimate dental experience, offering engaging and dynamic speakers, educational sessions, networking opportunities, health and wellness sessions and educational webinars and more. Participants can even learn more about the various enhancements available with CEREC Software 5.2 by attending Dr. Mike Skramstad’s course at Dentsply Sirona World 2021.
More information on our groundbreaking program and registration can be found here: www.dentsplysironaworld.com .
More information about Primescan can be found here: www.dentsplysirona.com/primescan
*Unless stated otherwise, all comparative statements in this press release refer to a comparison between Dentsply Sirona products.
Due to the different approval and registration times, not all technologies and products are immediately available in all countries.







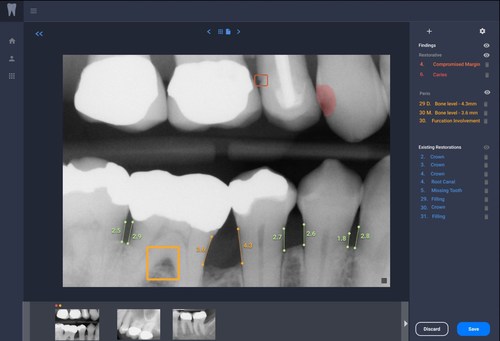


 The latest upgrade of the Connect and CEREC Software, 5.2, and an improved firmware provide Primescan and CEREC users with additional options for making treatment more efficient, faster, and more convenient with digital workflows. Primescan intraoral scanner’s latest enhancements optimize performance and improve usability as described as follows; Through its enhanced firmware, Primescan captures up to 1.5 million 3D data points per second. With these enhancements, Primescan is significantly smoother and twice as fast, making it again easier to scan successfully even for relatively inexperienced users*.
The latest upgrade of the Connect and CEREC Software, 5.2, and an improved firmware provide Primescan and CEREC users with additional options for making treatment more efficient, faster, and more convenient with digital workflows. Primescan intraoral scanner’s latest enhancements optimize performance and improve usability as described as follows; Through its enhanced firmware, Primescan captures up to 1.5 million 3D data points per second. With these enhancements, Primescan is significantly smoother and twice as fast, making it again easier to scan successfully even for relatively inexperienced users*. This National Tooth Fairy Day, Delta Dental reminds families and caregivers of the role the Tooth Fairy can play in teaching children about oral health. While 72% of parents
This National Tooth Fairy Day, Delta Dental reminds families and caregivers of the role the Tooth Fairy can play in teaching children about oral health. While 72% of parents 
