 Dr. Nigel W. Bunnett, an internationally known expert on the mechanisms of pain, itch, and neurogenic inflammation, has been named chair of theDepartment of Basic Science and Craniofacial Biology at the New York University College of Dentistry (NYU Dentistry), effective October 1, 2019.
Dr. Nigel W. Bunnett, an internationally known expert on the mechanisms of pain, itch, and neurogenic inflammation, has been named chair of theDepartment of Basic Science and Craniofacial Biology at the New York University College of Dentistry (NYU Dentistry), effective October 1, 2019.
Dr. Bunnett’s research focuses on primary sensory neurons, the first cells in the pathways to pain, itch, and neurogenic inflammation and the cells that they innervate. These include second order spinal neurons, which integrate information about pain and itch and transmit signals centrally, and cells in the periphery that mediate neurogenic inflammation.
Pain, itch, and neurogenic inflammation are evolutionarily-conserved and protective mechanisms that are essential for survival. Pain allows awareness and avoidance of danger, itch evokes scratching that removes noxious substances from the skin, and inflammation fights infection. Normally, pain, itch and neurogenic inflammation are tightly controlled and short-lived. However, during disease they can become dysregulated, chronic, and debilitating. Chronic pain afflicts 20 percent of the population at some point in their lives; chronic itch accompanies metabolic diseases and is often intractable; and chronic inflammation underlies diseases of global relevance, including atherosclerosis, diabetes, arthritis, and inflammatory bowel disease. Unfortunately, our understanding of acute and chronic pain, itch, and inflammation is incomplete, and current therapies are inadequate or have unacceptable side effects. Dr. Bunnett seeks to understand the normal mechanisms of pain, itch, and inflammation to determine why they can become dysregulated during disease and to discover new approaches for therapeutic intervention.
Dr. Bunnett studies two families of proteins at the surface of nerve cells that control pain, itch, and inflammation: G protein-coupled receptors and transient receptor potential ion channels. G-protein receptors are the largest class of signaling proteins, participate in all physiological and patho-physiological processes and are the target of one third of clinically used drugs. The small family of transient receptor potential ion channels also control important biological processes and are an emerging therapeutic target. G protein-coupled receptors and transient receptor potential ion channels allow nerves to sense noxious, irritating, and inflammatory stimuli. Their activation leads to the sensations of pain and itch, and controls the process of inflammation.
“I seek to understand the role of receptors and channels in the normal mechanisms of pain, itch, and inflammation, and to determine how their functions become dysregulated during disease,” says Dr. Bunnett. “By identifying the major mechanisms of chronic pain, itch, and inflammation, we aim to develop more effective and selective treatments for diseases that are a major cause of human suffering.”
At NYU, Dr. Bunnett will be joining his longtime research collaborator, Dr. Brian Schmidt, director of the NYU Oral Cancer Center, with whom he is currently collaborating on three grants that seek to develop strategies to improve the lives of patients suffering from chronic pain, including oral cancer pain: a $2.4 million, 3-year Department of Defense grant to determine how receptors inside nerve cells generate chronic pain; a $2.7 million, 3.5-year National Institute of Diabetes and Digestive and Kidney Diseases grant to understand the role of Protease-Activated Receptor 2 in chronic pain associated with irritable bowel syndrome and inflammatory bowel disease; and a $3.7 million, 5-year National Institute for Dental and Craniofacial Research grant to understand oral cancer pain, and ultimately develop new therapies. Dr. Bunnett and Dr. Schmidt collaboratively investigated the role of proteases in oral cancer pain in 2009 when they were both faculty members at the University of California, San Francisco (UCSF).
Dr. Bunnett has served since 2016 as the Gerald and Janet Carrus Professor of Surgical Sciences, vice chair of research in surgery; and professor of surgery and pharmacology at the Columbia University College of Physicians and Surgeons. Educated at Cambridge University, where he received his PhD degree in 1981, Dr. Bunnett went on to a postdoctoral fellowship at the University of California, Los Angeles, followed by an assistant professorship at the University of Washington Seattle. In 1987, he joined UCSF, where he became professor of surgery and physiology, vice chair of surgery, and director of the UCSF Center for the Neurobiology of Digestive Diseases. In 2011, Dr. Bunnett moved to Monash University, Melbourne, as NHMRC Australia Fellow, professor of pharmacology and medicine, and deputy director of the Monash Institute of Pharmaceutical Science.
Over the past decade, Dr. Bunnett has been awarded more than $20 million in federal and foundation support. He has published more than 350 research articles, reviews, and chapters, which have been cited over 31,000 times. His contributions have been recognized by numerous awards, including the Australia Fellowship, the NIH MERIT Award, the Novartis Neurogastroenterology Award, the Jansen Award for Basic Research in Gastroenterology, and the Victor Mutt Award for Research in Regulatory Peptides.
“Dr. Bunnett’s path-breaking work in uncovering the way in which chronic pain is transmitted through nerve cells, along with his consistent research funding, scholarship, and collaborative approach, make him uniquely well-qualified to lead the Department of Basic Science and Craniofacial Biology in advancing the basic science enterprise as the foundation for innovative therapeutic applications,” said Dr. Charles N. Bertolami, Herman Robert Fox Dean of NYU Dentistry. “It is predictable,” he added, “that these talents will enable him to develop partnerships across departments and centers throughout the university as a whole, as well as at the College of Dentistry.”
About NYU College of Dentistry
Founded in 1865, NYU Dentistry is the third oldest and the largest dental school in the US, educating more than 9 percent of all dentists. NYU Dentistry has a significant global reach with a highly diverse student body. Visit https://dental.nyu.edu for more.





 Zero to 4 mm in 3 seconds
Zero to 4 mm in 3 seconds

 Palmero Healthcare, a Hu-Friedy subsidiary, is pleased to introduce a tension relief band that works to enhance the physical and mental well-being of dental practitioners. CaRing Tension Relief Bands are a flexible and convenient way to soothe the sore, achy fingers that plague many professionals. The company hopes to eliminate finger fatigue and promote hand care maintenance, which can improve overall stress management and well-being.
Palmero Healthcare, a Hu-Friedy subsidiary, is pleased to introduce a tension relief band that works to enhance the physical and mental well-being of dental practitioners. CaRing Tension Relief Bands are a flexible and convenient way to soothe the sore, achy fingers that plague many professionals. The company hopes to eliminate finger fatigue and promote hand care maintenance, which can improve overall stress management and well-being.
 Dr. Nigel W. Bunnett, an internationally known expert on the mechanisms of pain, itch, and neurogenic inflammation, has been named chair of theDepartment of Basic Science and Craniofacial Biology at the New York University College of Dentistry (NYU Dentistry), effective October 1, 2019.
Dr. Nigel W. Bunnett, an internationally known expert on the mechanisms of pain, itch, and neurogenic inflammation, has been named chair of theDepartment of Basic Science and Craniofacial Biology at the New York University College of Dentistry (NYU Dentistry), effective October 1, 2019.
 Peter E. Dawson, D.D.S., founder of The Dawson Academy and dental industry pioneer, has died at the age of 89. Dr. Dawson is widely considered one of the most influential clinicians and teachers in dental history.
Peter E. Dawson, D.D.S., founder of The Dawson Academy and dental industry pioneer, has died at the age of 89. Dr. Dawson is widely considered one of the most influential clinicians and teachers in dental history.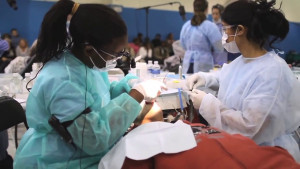 On Saturday, September 14, 2019, A Night for Smiles will host its second annual Black Tie Gala to benefit The Kenneth D. Owen North Carolina Missions of Mercy (NCMOM) – Charlotte Chapter (
On Saturday, September 14, 2019, A Night for Smiles will host its second annual Black Tie Gala to benefit The Kenneth D. Owen North Carolina Missions of Mercy (NCMOM) – Charlotte Chapter (




