The company unveiled the new monolithic full-arch restoration at the Chicago Dental Society (CDS) Midwinter Meeting.
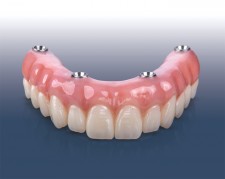
With the new addition to the extensive line of BruxZir restorative solutions, clinicians now have an option for edentulous implant patients with high esthetic expectations — without the risk of fracturing the veneering material used in most premium full-arch zirconia restorations. "The BruxZir Esthetic Implant Prosthesis is a superb monolithic restoration," said Darius Raudys, manager of the Implant department at Glidewell. "We use the latest CAD/CAM technology to mill the entire prosthesis from a block of BruxZir Esthetic Zirconia. And our technicians have spent years developing staining techniques that result in gingival anatomy that is remarkably lifelike. The result is a beautiful restoration that won't break — at nearly half the price you'll pay through boutique dental labs."
The original BruxZir Implant Prosthesis, fabricated from BruxZir Full-Strength Zirconia, remains a cornerstone of the company's dental lab implant offerings and is recommended for patients with parafunctional habits, a strong bite, or a history of damaging dental restorations. Whichever formulation is best-suited to the individual patient, the complete lab service includes a provisional implant prosthesis, which affords clinicians and patients an opportunity to confirm the esthetics, function and occlusion of the prosthetic design before the restoration is milled from solid zirconia.
The company also announced that the restorative protocol used to fabricate the BruxZir Implant Prosthesis has been overhauled to enhance clinical efficiency. The protocol, based on extensive feedback from dentists and clinical R&D conducted in the company's dental operatories, is expected to produce more accurate results. "Our new process will save dentists time by minimizing wax reset appointments," said Brian Volken, director of implant business development at Glidewell. "This frees up valuable chair time for the doctor and gives patients a better experience, as well as a lifelike restoration they'll be proud to show off."
To help clinicians adapt to the new, improved clinical workflow, cases are now packaged with appointment-specific step-by-step guides, as well as checklists for the items practices will need, receive and send back for each appointment. The company has also produced tutorial videos for key parts of the restorative process that are available online, along with additional educational resources to smooth the path to success in full-arch implant treatment.
Prosthodontist Dr. Taylor Manalili, a member of the clinical research team and director of prosthodontics at Glidewell, spearheaded the effort to streamline the clinical workflow. "Our goal is to simplify what has been a traditionally complex procedure, and I believe we've accomplished that. We're giving dentists the best of both worlds — the clinical tools and the laboratory support they need to better understand and execute the overall restorative process."
For more information on the BruxZir Implant Prosthesis and the BruxZir Esthetic Implant Prosthesis, including access to tutorial videos and a wide array of other clinical and educational resources, visit glidewelldental.com/bip or call 866-861-7955.
About Glidewell
Glidewell is among the world's largest providers of custom restorative services and is recognized as an industry-leading materials and devices manufacturer. Established in 1970 in Orange County, California, Glidewell continues to build on its storied history of technological innovation, continuing education and a commitment to making comprehensive, high-quality treatment more accessible to patients in the U.S. and internationally.




