Two dental industry magazines published by Conexiant were honored recently with MarCom Awards. Inside Dental Technology received a Platinum Award for Creativity and Design in the Magazine Interior category for the article “Implant Fundamentals,” published in its May 2024 issue; and Inside Dental Hygiene received a Gold Award for Creativity and Design in the Magazine Interior category for the article “Oral Cancer Screening,” published in its March/April 2024 issue.
Held annually, the MarCom Awards honor excellence in marketing and communication while recognizing the creativity, hard work, and generosity of industry professionals. MarCom Awards are administered by the Association of Marketing and Communication Professionals (AMCP). The international organization, founded in 1995, consists of several thousand marketing, communication, advertising, public relations, digital, and web professionals.
Implant Fundamentals Article

Inside Dental Technology is the complete information resource for dental laboratory decision makers, supporting their commitment to provide optimal patient care and to remain competitive by advancing education, artistry, scientific knowledge, productivity, and profitability. Included in the Conexiant dental portfolio, IDT is committed to presenting a broad perspective through print publications, online resources, and educational events that enhance collaboration among all types of dental professionals to lead the industry into the future.
To read the “Implant Fundamentals” article in IDT, click here.
Oral Cancer Screening Article
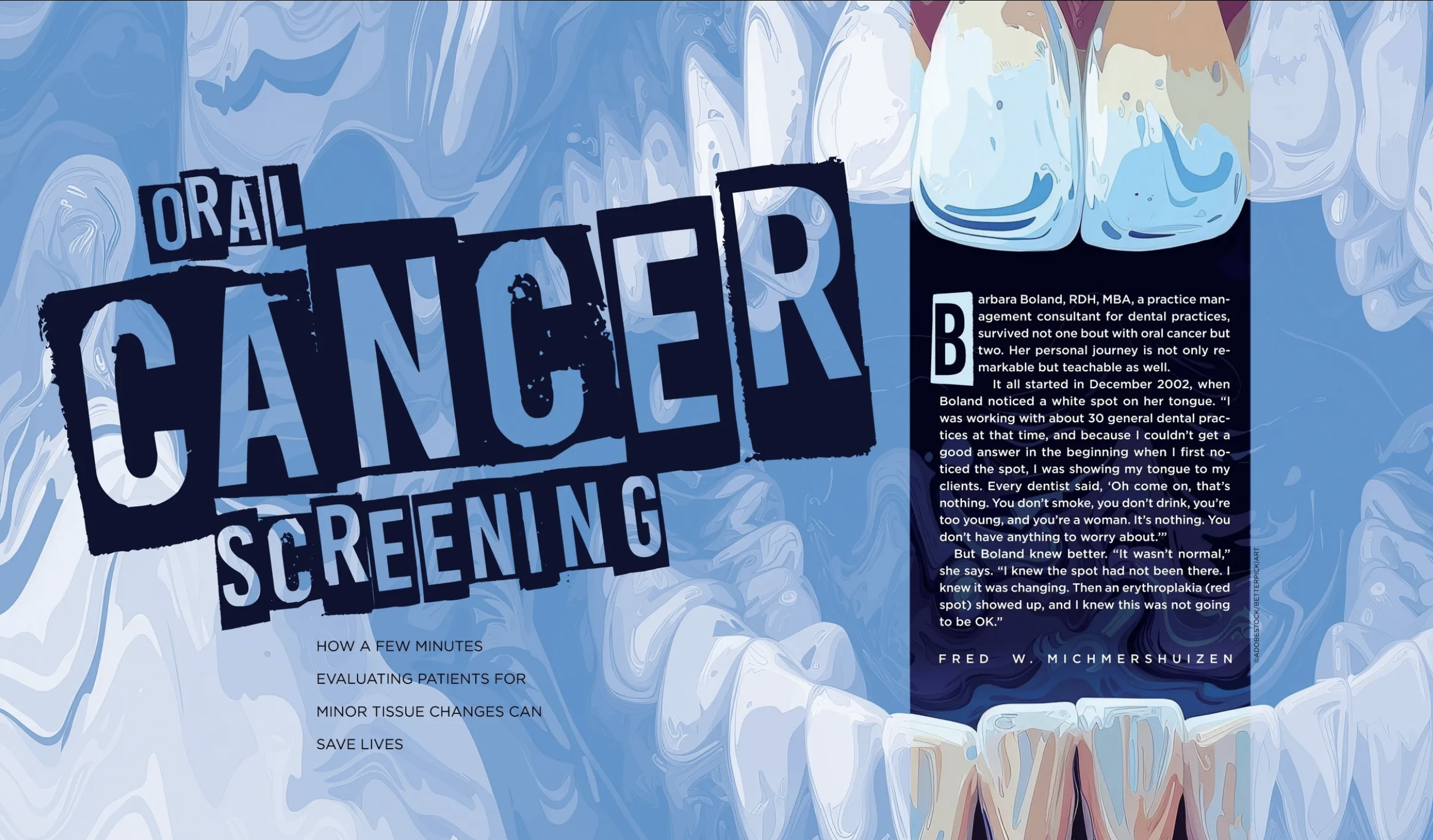
Inside Dental Hygiene is the complete information resource for the dental hygiene profession, supporting dental hygienists’ commitment to optimal patient care, education, regulatory compliance, positive career choices, and a healthy lifestyle.
To read the “Oral Cancer Screening” article, click here.
Other Awards for Conexiant Publications
The MarCom Awards for Inside Dental Technology and Inside Dental Hygiene are not the first design awards bestowed upon Conexiant’s dental publications. Earlier this year, Inside Dentistry and Inside Dental Hygiene magazines were recognized in the 2024 Azbee Awards of Excellence, and Inside Dental Technology was recognized in the 2024 Hermes Awards. To read about these previous awards, click here.
Conexiant is the premier global destination for healthcare professionals looking for trusted clinical content, breaking news, and a wealth of educational resources designed to support professional growth. The company utilizes state-of-the-art technology to tailor information that fits the unique needs of various medical and dental specialties.
View all of the Conexiant news, topics, and publications by clicking here.




 VITA has announced the addition of the VITA VACUMAT® 6100 M to its line of tried and tested furnaces. The new VITA VACUMAT 6100 M is a fully-automatic, microprocessor-controlled firing unit that is ideal for all dental firing ceramic requirements.
VITA has announced the addition of the VITA VACUMAT® 6100 M to its line of tried and tested furnaces. The new VITA VACUMAT 6100 M is a fully-automatic, microprocessor-controlled firing unit that is ideal for all dental firing ceramic requirements.
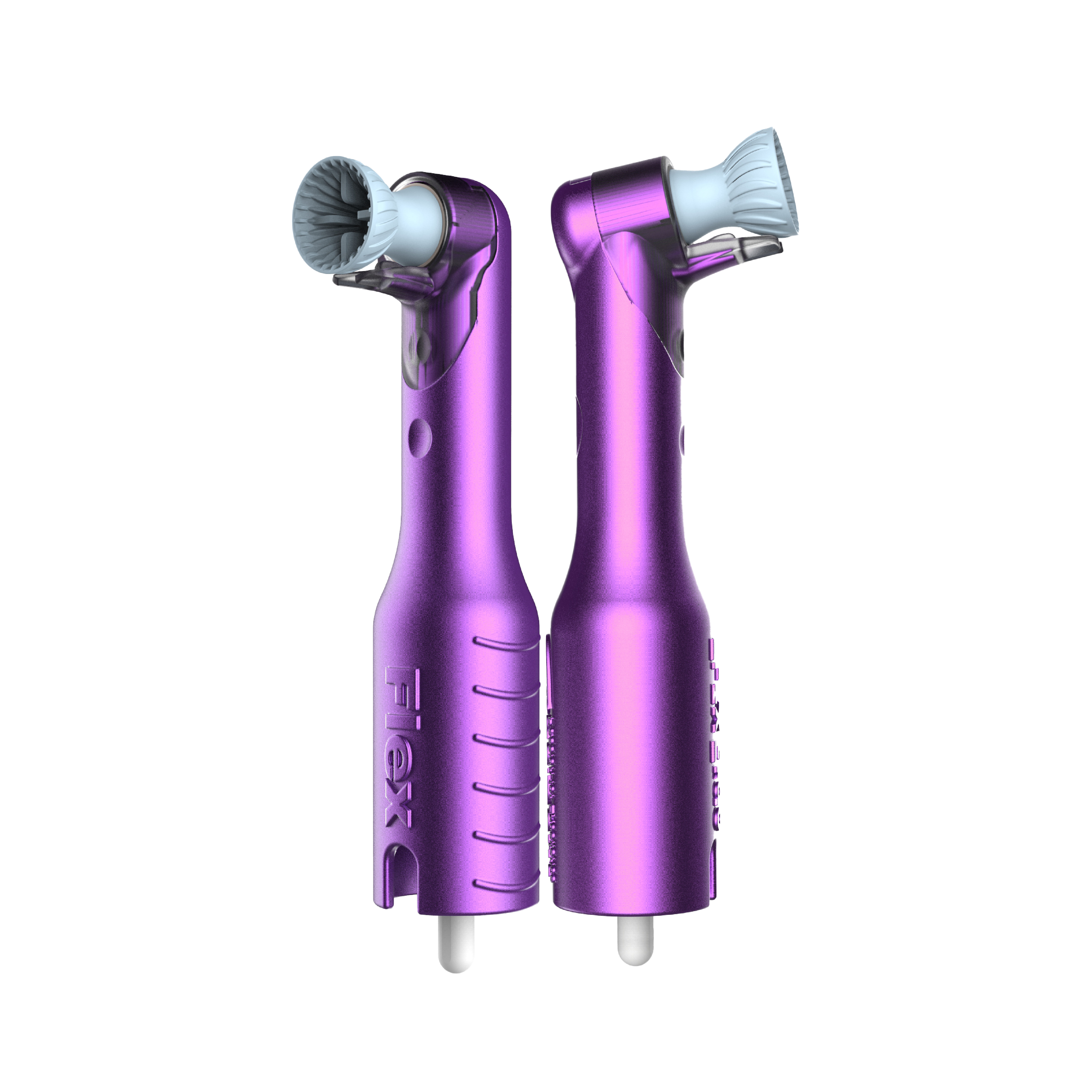 Pac-Dent, Inc., is thrilled to announce the launch of the AntiSplatr® Flex Slim and AntiSplatr® Flex Ergo Slim disposable prophy angles. The AntiSplatr® Flex Slim and AntiSplatr® Flex Ergo Slim expand Pac-Dent’s growing preventive portfolio, solidifying Pac-Dent as a leading innovative force in the dental industry.
Pac-Dent, Inc., is thrilled to announce the launch of the AntiSplatr® Flex Slim and AntiSplatr® Flex Ergo Slim disposable prophy angles. The AntiSplatr® Flex Slim and AntiSplatr® Flex Ergo Slim expand Pac-Dent’s growing preventive portfolio, solidifying Pac-Dent as a leading innovative force in the dental industry.

 exocad, an Align Technology, Inc. company and a leading dental CAD/CAM software provider, today announced its participation at the 100th Annual Greater New York Dental Meeting from December 1–4, 2024. exocad will exhibit at booth #2003 and host educational sessions featuring Thanh Nguyen, Senior Manager of Product Requirements at exocad, award-winning cosmetic dentist Dr. Diana Tadros and digital expert Dr. Michael Scherer.
exocad, an Align Technology, Inc. company and a leading dental CAD/CAM software provider, today announced its participation at the 100th Annual Greater New York Dental Meeting from December 1–4, 2024. exocad will exhibit at booth #2003 and host educational sessions featuring Thanh Nguyen, Senior Manager of Product Requirements at exocad, award-winning cosmetic dentist Dr. Diana Tadros and digital expert Dr. Michael Scherer.
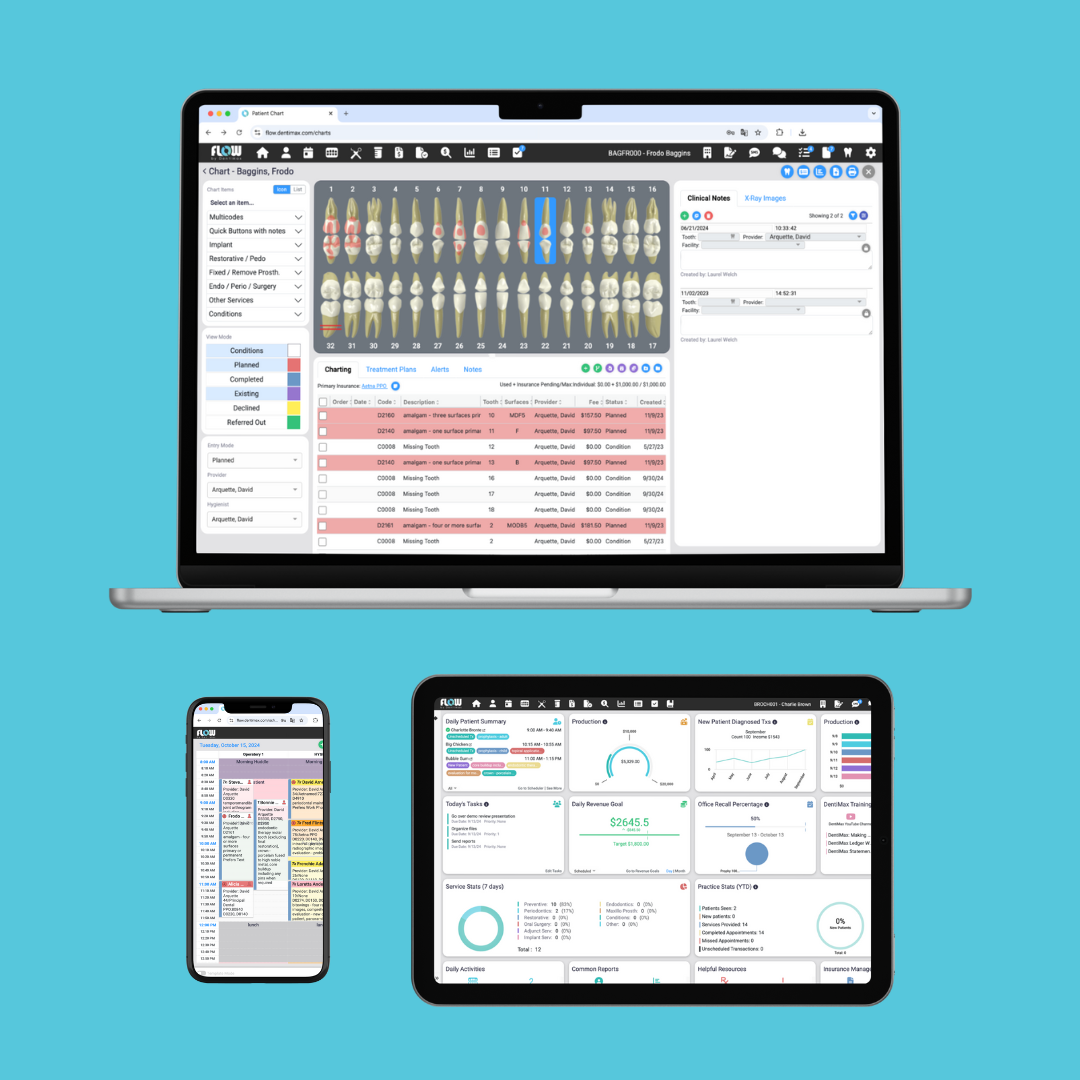 DentiMax, a leader in dental technology solutions, is thrilled to announce the launch of DentiMax Flow, innovative browser-based practice management software designed to transform the way dental practices operate. Built to meet the dynamic needs of modern dental offices, DentiMax Flow gives dental professionals access to their entire software suite, including appointments, patient records, billing, reports, SMS, documents and more from any device that has an internet connection.
DentiMax, a leader in dental technology solutions, is thrilled to announce the launch of DentiMax Flow, innovative browser-based practice management software designed to transform the way dental practices operate. Built to meet the dynamic needs of modern dental offices, DentiMax Flow gives dental professionals access to their entire software suite, including appointments, patient records, billing, reports, SMS, documents and more from any device that has an internet connection. In an open letter to the healthcare community released today, American Dental Hygienists’ Association (ADHA®) President Erin Haley-Hitz, RDH, issued a strong objection to recently passed American Dental Association (ADA) resolutions that would eliminate faculty-to-student ratios in dental hygiene programs and allow unlicensed practitioners to perform dental hygiene services.
In an open letter to the healthcare community released today, American Dental Hygienists’ Association (ADHA®) President Erin Haley-Hitz, RDH, issued a strong objection to recently passed American Dental Association (ADA) resolutions that would eliminate faculty-to-student ratios in dental hygiene programs and allow unlicensed practitioners to perform dental hygiene services. The NYU College of Dentistry will host its third AHEAD Symposium on Wednesday, November 13, 2024, from 9:00 AM to 12:00 PM ET. The virtual event, titled “Achieving Health Equity through Access for All with Disabilities,” will bring together policymakers, experts, and advocates to address the critical issue of oral health care access for people with disabilities.
The NYU College of Dentistry will host its third AHEAD Symposium on Wednesday, November 13, 2024, from 9:00 AM to 12:00 PM ET. The virtual event, titled “Achieving Health Equity through Access for All with Disabilities,” will bring together policymakers, experts, and advocates to address the critical issue of oral health care access for people with disabilities.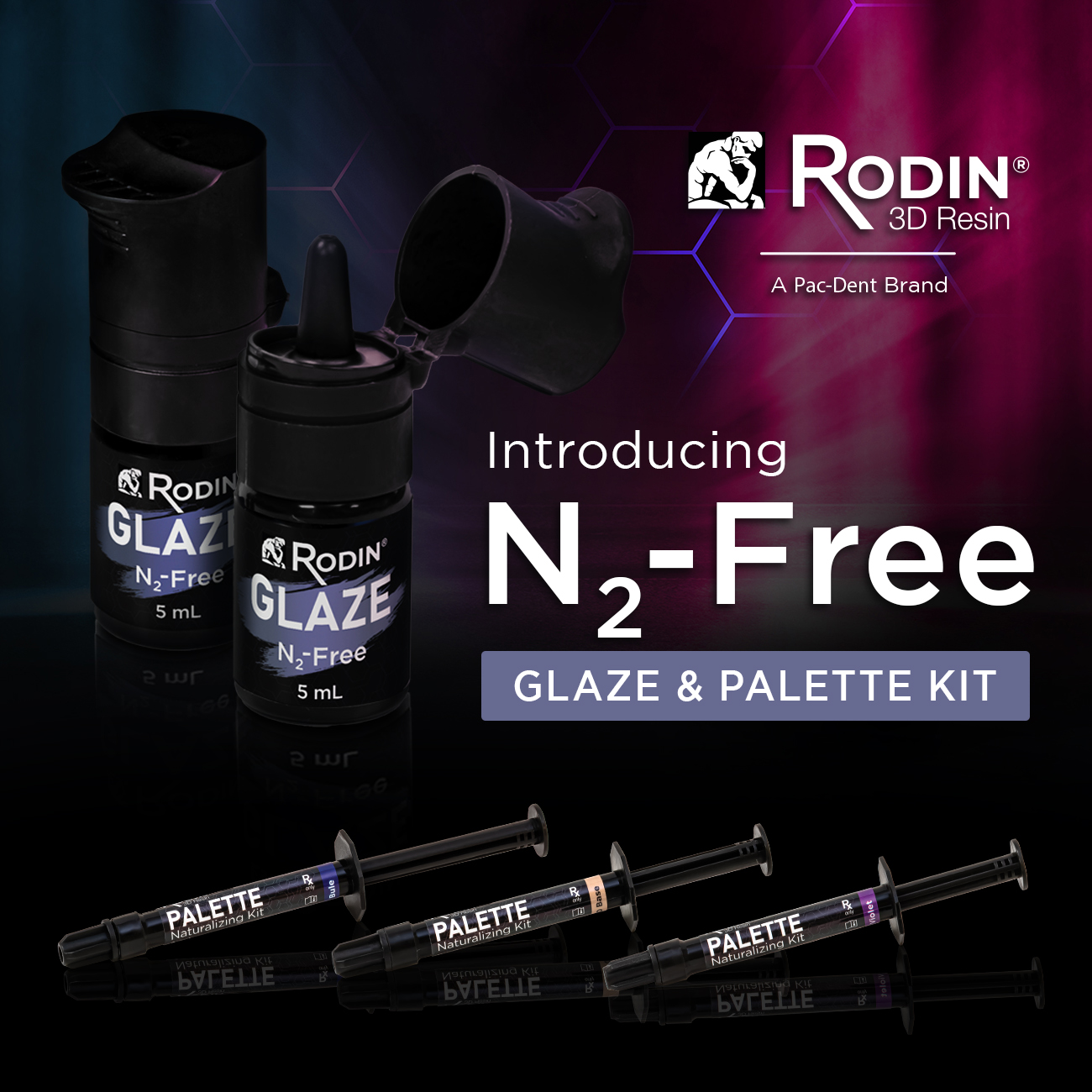 Pac-Dent, Inc. is excited to announce the launch of Rodin® Glaze N2-Free and Rodin® Palette N2-Free Naturalizing Kit, a groundbreaking resin characterization system set to redefine the staining and glazing procedure for printed dental restorations. This innovative system has been designed for surface detailing across a wide range of dental restorations, including hybrid ceramics, PMMA resins, and CAD/CAM composites, ensuring compatibility with various workflows.
Pac-Dent, Inc. is excited to announce the launch of Rodin® Glaze N2-Free and Rodin® Palette N2-Free Naturalizing Kit, a groundbreaking resin characterization system set to redefine the staining and glazing procedure for printed dental restorations. This innovative system has been designed for surface detailing across a wide range of dental restorations, including hybrid ceramics, PMMA resins, and CAD/CAM composites, ensuring compatibility with various workflows.
