Milled Implant Abutments
Creating custom restorations for the benefit of the patient
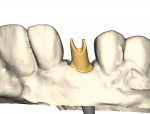
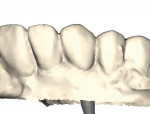
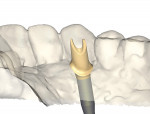
As analog labor-intensive fabrication processes give way to digitally driven virtual design and milling production, CAD/CAM technology continues to shape and reshape the dental technology industry. The same production efficiencies and accuracy that impact the virtual design and automated manufacturing processes for crown-and-bridge fabrication are expanding exponentially into other areas of product manufacture. One such area is CAD designed and CAM milled patient-specific implant abutments. With new materials and design capabilities rapidly being introduced to the market (Figure 1 through Figure 4), the industry as a whole is undergoing a paradigm shift, thus changing what we do and how we do it—all to the ultimate benefit of the patient.
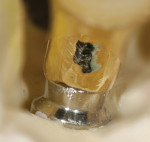
As the industry continues to adapt to digitally-driven processes, laboratories investing in scanning technology have discovered that the CAD software used for the design of substructures and full-contour restorations also enables them to provide their customers with custom-designed and milled implant abutments. Creating a custom abutment using conventional analog processes, whether modifying stock or pre-fabricated abutments, or creating custom-cast UCLA abutments, has always been a labor-intensive technique fraught with inconsistencies and work-arounds. These analog methods often resulted in poorly formed or even misformed interfaces (Figure 5). As medical devices, implant abutments are not merely a connection between the implant root and final crown, but also perform the important task of sealing the entire implant system against infection and bone loss. If the technician does not understand emergence profile, biologic width, and proper tissue support, as well as the importance of the abutment connection, a microgap can be created in the connection. These micrograps permit microscopic bacterial leakage when the patient applies a lateral load as low as 100 Newton centimeters (Ncm). A properly fitted implant abutment is a way to prevent microleakage prevention, and it provides one of the most important connections in implant dentistry. Microleakage is also behind the movement within the implant industry toward the conical interface implant systems such as Nobel Biocare’s NobelActive® (www.nobelbiocare.com), DENTSPLY Implants’ ANKYLOS® (www.dentsplyimplants.us), and Straumann’s Bone-Level implant (www.straumann.us), which all provide a secure biological seal.
CAD/CAM and Custom Abutments
CAD/CAM technology, in conjunction with other advanced digital technologies for diagnosing and treatment planning implant cases, has significantly improved patient care. On the laboratory side, CAD designed and machine-milled abutment components (Figure 6) no longer need technician intervention to grind, touch, polish, or sandblast the interface to obtain fit and function, which in the past was the origin of the microgap and subsequent infection and bone loss for the patient. Individual control of CAD designed abutments has resulted in proven benefits for the laboratory, including improved fit, faster turnaround time, and reduced production expenses. Although advances in design software have simplified the design process, technicians must take into consideration the multiple complications and risk factors that any abutment presents, such as the abutment design, the choice of material, and the method of manufacture. Among the questions technicians must answer are: What implant system is being used? Is there a conical interface? Does the material chosen provide enough strength? Are there wear and occlusal considerations that must be taken into account? Are there parafunctional issues? Again, custom abutments are medical devices and as such come with myriad considerations prior to design and manufacture.
CAD/CAM produced custom abutments are available in three material configurations: titanium, zirconia, and titanium-zirconia hybrids (Figure 7). Titanium abutments are a popular choice given their strength and some manufacturers offer optional special coatings like gold nitride to enhance the esthetics underneath the tissue of the final restoration.
All-zirconia has also become a popular choice for the manufacture of custom abutments, particularly among those milling in-house. Available in a wide variety of base shades, zirconia is a great material to obtain the patients desired esthetics while still demonstrating excellent strength. As a precautionary note, research has shown that zirconia abutments can be vulnerable to fracture if not designed correctly or if not handled properly by the laboratory after milling or in the practice. When creating a zirconia abutment, technicians need to keep in mind that they are not dealing with metal alloy, which has inherent give against masticatory forces.
Titanium-zirconia hybrid custom abutments have been the proposed solution to providing the mechanical reliability of a titanium-to-titanium interface with the esthetic advantage of a shaded zirconia abutment. Typically an option only for bone level implant platforms, the titanium-zirconia hybrid is uniquely configured with a titanium interface for the implant, plus a cylinder that is cemented to a custom-milled zirconia abutment. Again, laboratory technicians must have intimate knowledge regarding proper design and cementation protocols to create a biologically functional, long-term solution.
Factory vs. Factory Compatible
Every implant manufacturer offers stock implant abutments for their implant systems and many offer custom-milled solutions. For years, laboratories have been modifying stock abutments and casting their own UCLA abutments to create patient-specific solutions. Today a number of third-party companies offer stock abutments and parts compatible with factory implants, as well as custom-milled options (Figure 8). There is a question of warranty when utilizing the services of a third-party supplier for milled custom abutments versus those of the implant manufacturer, however, this has become an even grayer issue with the number of implant manufacturers that now offer custom abutments and parts that are compatible with other manufacturers’ implant systems.
Whether or not to use factory parts, third-party parts, or mill your own becomes the choice of the laboratory and clinician involved in the case.
Scan, Design, and Mill
For the many laboratories already invested in scanning technology, a digital workflow is a part of their daily routine. This workflow either involves scanning and designing the custom abutment case and electronically submitting that data to the manufacturer or third-party supplier or scanning the model and letting the supplier design and mill the case. For laboratories not yet invested in scanning technology there remains the option of shipping the model to the supplier for the scan, design, and mill process, although this business model involves more time and cost in shipping and turnaround. Several implant manufacturers have integrated digital impressioning to allow clinicians to scan the case in the practice and send it electronically to the laboratory for virtual custom abutment design and either outsourced or in-house milling of the abutment.
The business model for laboratories that have invested in complete CAD/CAM systems involves keeping the entire manufacturing operation in-house—from scan and design to milling the custom abutment. This business model places all the onus and responsibility on the laboratory to guarantee the integrity of the final product. This includes everything from the CAD abutment design to ensuring the mechanical operation and calibration of the milling machine.
For the majority of laboratories, a scan-only or scan and design business model is the norm. CAD abutment design modules are available for the most common dental CAD software packages. To aid in accurately identifying the position of the implant, scan locators or scan abutments are available that provide the CAD software with a flat surface to calculate the location and orientation of the analog in the model.
Many of the software CAD modules support concurrent design of the custom abutment and the substructure or full-contour restoration, speeding up case processing time and allowing the CAD designer to take all aspects of the case into consideration.
Conclusion
CAD/CAM technology has given dental technicians incredible tools to manufacture restorations that are more accurate, of the highest quality, and at a production level that surpasses any analog process. However, as the technology continues to develop in its complexity and ability to produce new components and products, it is the technician’s responsibility to understand at the deepest level the oral biomechanics of the end product, the properties of the materials being used, and the intricacies of the machinery milling the end product. As the technology becomes more intuitive, it will become even more imperative that technicians understand the dentistry behind their creations in order to best assess the quality and accuracy of the end product. Intuitiveness of the technology is not a substitute for the knowledge and skill possessed by an educated dental technician. It is our responsibility to use these tools to take control, and ensure that the products we manufacture offer the highest levels function and fit, so that we may provide our patients with best practices dentistry.
Lars Hansson, CDT, FICOI, is the head of Bay View Dental Laboratory’s implant
department in Chesapeake, VA.