Zirconia: The Material, Its Evolution, and Composition
John O. Burgess, DDS, MS
Full-contour zirconia restorations are now the most prescribed ceramic material for posterior crowns.1 The rapid transition to zirconia monolithic restorations was fueled by the clinical success of monolithic zirconia posterior ceramic restorations, accurate milling, and rising metal costs. Zirconia continues to evolve, ranging from opaque and strong to more translucent but weaker materials. With this evolution, clinicians must be aware of the properties and differences between zirconia materials, which will affect the success or failure of their final restoration. This article describes the material, its continuum, and how zirconia has evolved.
The Material
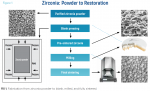
Zirconium is a soft, silver-colored metal mined from large deposits in Australia and Africa as a silicate mineral called zircon (ZrSiO4). Berzelius first isolated the metal in an impure form in 1824.2 The zircon mineral is purified to produce zirconia powder with controlled particle size and composition (Figure 1). Metal oxides are added to the powder: yttrium to stabilize the tetragonal phase of zirconium, aluminum to prevent water corrosion, and hafnium oxide to reduce pore development and coloring components. The original, opaque white-frame zirconia was made from one powder pressed into the blank. As with paint, which has tints added to create color in a base shade, various powders are added to achieve the final shade.
Both composition and powder particle size affect the ceramic microstructure, which dictates the mechanical and physical properties of the zirconia. The starting powders contain a binder and additives which, with the zirconia powder, are pressed into flexible molds to provide cohesiveness. Pressing is done with the isostatic method:the material is placed in a flexible mold, the mold is put into a liquid, and pressure is applied to the solution, which applies pressure to the mold containing the powder from all sides (uniaxial pressing applies pressure from the ends only and is not effective in forming zirconia with uniform density). Compressing from all directions reduces porosity in the finished product. Some still use uniaxial pressing only, others isostatic pressing, whereas a few use both to obtain uniform shrinkage and less stress in the final restoration. The binder and other additives are removed by heating the formed zirconia to a "green" or pre-sintered state, which is sintered enough to maintain its shape but soft enough to be easily machined. The partially sintered zirconia puck or block is milled into the final restoration shape and then fully sintered, producing approximately 20% shrinkage in the finished restoration. The mill reads the density of the blank and enlarges the final restoration to compensate for sintering shrinkage. Dentistry uses 3, 4, or 5 mole % yttria-stabilized tetragonal zirconia polycrystalline material (3, 4, or 5 Y-TZP).
Zirconia is a polymorphic material existing in three phases, depending on temperature applied to the powder. The first phase is monoclinic (stable at room temperature up to 1,170 ºC); with additional heat, the tetragonal phase occurs (stable at 1,170 ºC to 2,370 ºC); and with further heating, the cubic phase is obtained (stable over 2,370 ºC to 2,716 ºC). At room temperature, zirconia is present in its most stable phase, monoclinic, but when the monoclinic phase transforms into the tetragonal phase, it is accompanied by a volume shrinkage of approximately 4% to 5%. The tetragonal phase converts to the cubic phase at 2,370 °C, with minimal changes in volume.3,4
Dental Zirconia Evolution
Current zirconia can be classified into three basic groups based on the yttria content. The first group is strong, 3 mole % Y-TZP (mainly tetragonal) (IPS e.max® ZirCad LT and MO, Ivoclar Vivadent, ivoclarvivadent.com; Lava™ Plus, 3M, 3m.com; BruxZir®, Glidewell Laboratories, glidewelldental.com; and KATANA™ HT, Kuraray Noritake, KATANAzirconia.com). The second group is more translucent, 4 mole % Y-TZP (Zpex® 4, Kraun, kraun.eu; IPS e.max ZirCAD MT; and KATANA™ ST/STML). The third group is most translucent, 5 mole % Y-TZP (eg, Lava Esthetic; Cercon® XT, Dentsply Sirona, dentsplysirona.com; BruxZir Anterior; KATANA™ UT/UTML; and Zpex Smile), with reduced mechanical properties.5
The mole percent of yttria generally defines the mechanical and physical properties of the zirconia and is provided as round numbers (3, 4, or 5). Mole percentages are actually in tenths mole percent, which explains the variation in mechanical and physical properties in each category. Opaque zirconia containing about 3 mole % yttria is strongest (85% to 90% tetragonal phase); zirconia containing approximately 5 mole % yttria produces a more translucent material, with approximately 50% cubic phase (Table 1).6 Yttria increases the zirconia grain size and lowers the coefficient of thermal expansion. Mole % and weight % define the amount of yttria in zirconia: 3 Y-TZP is zirconia with approximately 3 mole % or 5 weight % yttria; 4 Y-TZP is zirconia containing 4 mole % or 7.1 weight % yttria; and 5 Y-TZP zirconia contains 5 mole % or 8.8 weight % yttria.
Opaque zirconia contained 3 mole % yttria to partially stabilize the tetragonal phase.7 This composition had the highest fracture toughness (3.5 to 4.5 MPa·m1/2) and flexural strength of 1,200 to 1,500 MPa (Lava, BruxZir, Cercon, and IPS e.max ZirCad), and 0.25 weight % alumina. Alumina (0.025%) stabilizes grain boundaries, is a sintering aid decreasing pore formation, and prevents water corrosion. Restorations made from 3 Y-TZP had a frame veneered with feldspathic porcelain because it was feared that the zirconia core would wear opposing enamel. With further testing, little opposing enamel wear was produced, but clinical fractures of the veneering material were common in early clinical trials. Three important changes reduced chipping of the veneering material: (1) matching the coefficient of thermal expansion of the veneering and zirconia core materials, (2) designing an anatomically correct zirconia core, and (3) slow cooling of the restorations.8
Even though 3 Y-TZP had significant strength, its opacity limited its use to posterior applications. An improved translucent 3 Y-TZP zirconia reduced alumina content from 0.25 weight % to 0.05 weight % (Lava Plus and Cercon), which maintained the zirconia's mechanical properties but with slightly more translucency. More recent versions of approximately 4 and 5 mole % Y-TZP retained the alumina at 0.05 weight % and increased yttria from 3 mole % to 4 to 5 mole %.7 These compositions (BruxZir Anterior, Lava Esthetic, KATANA™ HT, and Cercon XT) have 25% to 50% cubic polycrystals and lower fracture toughness and flexural strength compared with opaque 3 Y-TZP, but light transmission increased by 43% to 45% due to the increased cubic phase, less porosity, and increased size of the cubic grains (Figure 2).
Zirconia with approximately 4 mole % yttria has a flexural strength of 600 to 900 MPa and fracture toughness of 2.5 to 3.5, whereas 5 Y-TZP zirconia has a flexural strength of 700 to 800 MPa and fracture toughness of 2.2 to 4 MPa·m1/2, compared with opaque zirconia's flexural strength of 1,000 to 1,400 MPa and fracture toughness of 3.5 to 4.5 MPa·m1/2. The 5 Y-TZP translucent zirconia has similar but higher flexural strength and fracture toughness compared with e.max; this material could be considered a stronger but less-translucent lithium disilicate. Translucent zirconia contains approximately 50% cubic crystals, which are weaker and do not transform. Zirconia with 8 mole % yttria will completely stabilize the cubic phase,6 whereas the most translucent zirconia with5 mole % yttria is partially stabilized with approximately 50% cubic zirconia.6 With translucent zirconia, greater reduction is required for the posterior tooth preparation: 1-mm occlusal reduction with 3 Y-TZP posterior restorations, whereas 4 and 5 Y-TZP requires 1.2 to 1.5-mm occlusal reduction. Axial reduction should range from 0.3 to 0.8 mm.9
Ceramic Flexural Strength vs Fracture Toughness
Flexural strength specimens are highly polished. Fracture toughness testing uses a notched specimen and describes the ability of the material to resist crack propagation. Cracks originate from flaws (stress risers) in a material created by finishing, machining, or porosity and act as an initiation point for crack growth. For example, an auto windshield can be hit with a rock that chips the glass, and the crack can spread from the chip across the windshield. The energy required to grow the crack is measured by fracture toughness. Materials with low toughness have increased chipping and bulk fracture compared with materials with high fracture toughness. Fracture toughness values for feldspathic porcelain range from 1 to 1.3, translucent (5 Y-TZP) zirconia are 2 to 4, and opaque (3 Y-TZP) zirconia vary from 3.5 to 4.5 MPa·m1/2. For comparison, metals such as titanium alloys have significantly higher fracture-toughness values (44 to 107 MPa·m1/2) compared with ceramics and are highly resistant to cracking. Chipping occurs more frequently in the less-tough veneering materials compared with the zirconia frame.10,11
Low-Temperature Degradation
Partially stabilized tetragonal crystals can spontaneously revert to monoclinic crystal with a 3% to 5% expansion by low-temperature degradation (LTD). 5 Y-TZP containing more than 50% cubic phase has less LTD and lower fracture toughness, and because the material has little residual stress, water corrosion is limited. High cubic-containing zirconia (5 Y-TZP) is weaker, but its strength may not degrade further from LTD. Low-temperature transformation of the tetragonal to the monoclinic phase begins at the restoration surface and produces a rougher occluding surface.4
After 2014, the powder particle size decreased to approximately 0.2 to 0.5 μm,12 and increased yttria was added to stabilize zirconia, which increased cubic phase, resulting in decreased LTD13 and increased hydrothermal stability14 of the zirconia. As long as the tetragonal phase is present in zirconia, transformation toughening creates a transformation zone around developing cracks, slowing crack growth and producing few chips and fractures in the frame material. An in vitro test measures the effects of aging on zirconia specimens. This accelerated aging test for measuring transformation from the tetragonal to monoclinic phase is conducted according to ISO 13356 by autoclaving the specimens under steam pressure for 5 hours (at 134 °C, two bars of pressure).15 The test demonstrates that grain size affects transformation, and within limits, small grains transform more slowly. An hour of autoclaving represents several years of intraoral wear, with longer autoclaving times producing more change; 5 hours of aging at 134 °C, corresponds to an in vivo aging of 2 years.16
Wear of Zirconia and the Opposing Enamel
Multiple in vitro and in vivo studies have reported the low wear of zirconia opposing enamel.17-19 The author's wear testing20,21 uses an unground enamel cusp sliding 2 mm across the ceramic. Results are summarized by the following:
• Polished zirconia wears opposing enamel 10 times less than veneering porcelain.
• Polished zirconia wears opposing enamel slightly less than gold.
• Wear of the opposing enamel versus zirconia does not increase with time.
• Glazed zirconia significantly increases wear of opposing enamel compared with polished zirconia.
• The articulating-surface opposing enamel should be polished zirconia.
Why Dental Zirconia Is Opaque
When the properties of a material are the same in all directions, the material is isotropic. For Y-TZP zirconia, the grains have irregular boundaries that limit light transmission (anisotropic), whereas cubic-containing zirconia is isotropic, which improves light transmission. Improved light transmission occurs with smaller grains.5,22 The different index of refraction of the materials compositing 3 Y-TZP zirconia (alumina, yttria, colorants) increases light reflection as incident light transmission is decreased. Furthermore, different index of refraction of enamel or dentin as well as pores in the zirconia produce light scattering and absorption, resulting in an opaque material (Figure 2), which is important for light-curing zirconia restorations. A 0.5-mm-thick zirconia layer produces a 50% decrease in light transmission; at 1 mm, the reduction is 75%. With moderate staining and 0.5-mm thickness, light transmission is reduced by 85%, and at 1 mm, there is a 95% reduction.23
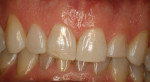
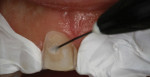
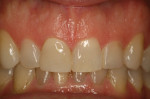
Opaque zirconia is especially beneficial in cases such as masking a single discolored tooth. Translucent zirconia (4 or 5 mole % yttria) is less translucent than lithium disilicate or enamel; if a highly translucent anterior restoration is needed, lithium disilicate or cutting back and layering the facial surface of the zirconia restoration are possible solutions. The improved translucent zirconia makes it a more viable option for less-demanding monolithic anterior restorations than previous versions of zirconia (Figure 3 through Figure 5).
The Future
Zirconia is supplied in disks, multi-layered disks, and machinable blocks. First introduced by Kuraray, the multi-layered disk reduces chairside staining and produces a more esthetic restoration. Ivoclar has also introduced a zirconia multi-layered puck, IPS e.max ZirCAD MT Multi. Kuraray has recently introduced rapid-firing zirconia blocks for chairside milling of zirconia restorations. Using the CEREC® SpeedFire oven (Dentsply Sirona) with a 30-minute sintering cycle, chairside zirconia milling is a clinical reality. The rapid sintering cycle does not affect the mechanical or physical properties of the zirconia. The future may reside in nano-zirconia with high-quality properties and improved translucency.
About the Author
John O. Burgess, DDS, MS
Adjunct Professor
University of Alabama at Birmingham -
School of Dentistry,
Birmingham, Alabama
References
1. Makhija SK, Lawson NC, Gilbert GH, et al. Dentist material selection for single-unit crowns: findings from the National Dental Practice-Based Research Network. J Dent. 2016;55:40-47.
2. Lide DR, ed. CRC Handbook of Chemistry and Physics. New York, NY: CRC Press; 2007:42.
3. Chen YW, Moussi J, Drury JL, Wataha JC. Zirconia in biomedical applications. Expert Rev Med Devices. 2016;13(10);945-963.
4. Kelly JR, Denry I. Stabilized zirconia as a structural ceramic: an overview. Dent Mater. 2008;24(3):289-298.
5. Zhang Y. Making yttria-stabilized tetragonal zirconia translucent. Dent Mater. 2014;30(10):1195-1203.
6. Zhang F, Inokoshi M, Batuk M, et al. Strength, toughness and aging stability of highly-translucent Y-TZP ceramics for dental restorations. Dent Mater. 2016;32(12):e327-337.
7. Zhang Y, Lawn BR. Novel zirconia materials in dentistry. J Dent Res. 2018;97(2):140-147.
8. Sailer I, Pjetursson BE, Zwahlen M, Hämmerle CH. A systematic review of the survival and complication rates of all-ceramic and metal-ceramic reconstructions after an observation period of at least 3 years. Part II: Fixed dental prosthesis. Clin Oral Implants Res. 2007;18(suppl 3):86-96.
9.Nakamura K, Harada A, Inagaki R, et al. Fracture resistance of monolithic zirconia molar crowns with reduced thickness. Acta Odontol Scand. 2015;73(8):602-608.
10. Nathanson D, Chiu S, Yamamoto H, Stappert C. Performance of zirconia based crowns and FPDs in prosthodontic practice [abstract]. J Dent Res. 2010;89(spec iss B). Abstract 2115.
11. Denry I, Kelly JR. Emerging ceramic-based materials for dentistry. J Dent Res. 93(12):1235-1242.
12. Lucas TJ, Lawson NC, Janowski GM, Burgess JO. Effect of grain size on the monoclinic transformation, hardness, roughness, and modulus of aged partially stabilized zirconia. Dent Mater. 2015;31(12):1487-1492.
13. Papanagiotou HP, Morgano SM, Giordano RA, Prober R. In vitro evaluation of low-temperature aging effects and finishing procedures on the flexural strength and structural stability of Y-TZP ceramics. J Prosthet Dent. 2006;96(3):154-164.
14. Morris G, Farr J, Flanigan PJ, et al. Crystal phase composition and hydrothermal stability of zirconia ceramics [abstract]. J Dent Res. 2017;96(spec iss A). Abstract 3206.
15. International Organization for Standardization. ISO 6872:2015 Dentistry-ceramic materials. ISO. https://www.iso.org/standard/59936.html. Accessed May 17, 2018.
16. Cattani-Lorente M, Durual S, Amez-Droz M, et al. Hydrothermal degradation of a 3Y-TZP translucent dental ceramic: a comparison of numerical predictions with experimental data after 2 years of aging. Dent Mater. 2016;32(3):394-402.
17. Lohbauer U, Reich S. Antagonist wear of monolithic zirconia crowns after 2 years. Clin Oral Investig. 2017;21(4):1165-1172.
18. Stober T, Heuschmid N, Zellweger G, et al. Comparability of clinical wear measurements by optical 3D laser scanning in two different centers. Dent Mater. 2014;30(5):499-506.
19. Passos SP, Torrealba Y, Major P, et al. In vitro wear behavior of zirconia opposing enamel: a systematic review. J Prosthodont. 2014;23
(8)593-601.
20. Burgess JO, Janyavula S, Lawson NC, et al. Enamel wear opposing polished and aged zirconia. Oper Dent. 2014;39(2):189-194.
21. Janyavula S, Lawson N, Cakir D, et al. The wear of polished and glazed zirconia against enamel. J Prosthet Dent. 2013;109(1):22-29.
22. Lucas TJ, Lawson NC, Janowski GM, Burgess JO. Effect of grain size on the monoclinic transformation, hardness, roughness, and modulus of aged partially stabilized zirconia. Dent Mater. 2015;31(12):1487-1492.
23. Kim MJ, Kim KH, Kim YK, Kwon TY. Degree of conversion of two dual-cured resin cements light-irradiated through zirconia ceramic disks. J Adv Prosthodont. 2013;5(4):464-470.