An Algorithmic Approach to Evaluation of an Ailing Implant
Marco Bergamini, DDS; Juliana Gomez, DDS; Huzefa S. Talib, BDS; Robert A. Horowitz, DDS; and Thomas G. Wiedemann, MD, DDS, PhD
Abstract: With the incidence of failed and ailing dental implants increasing, the authors conducted research to evaluate and characterize all known criteria used in the assessment of implant health in the clinical setting. A review of articles found in electronic databases was performed. Once all parameters for implant assessment as supported by current literature were selected, implant assessment documents were created: a data acquisition form and a quantitative comprehensive evaluation. These documents provide the clinician an algorithm that yields a prognosis of survival for each implant. Diagnostic criteria were organized to offer a comprehensive assessment of risk factors related to implant health. Data acquisition prior to establishing the prognosis is necessary in accordance with the staging system developed. Parameters for diagnosis and staging include a thorough medical/social history of the patient, implant history, and clinical evaluation. The authors concluded that the establishment of parameters for comprehensive implant evaluation in the clinical setting is feasible. This assessment process enables an effective clinical approach to evaluate and treat ailing implants while facilitating a clinical diagnostic algorithm.
Dental implants are a viable and predictable option to allow for restoration of form and function in patients who present with partial or full edentulism. Despite advancements in implant placement techniques and designs, an increasing incidence of failed and ailing implants has been reported.1-3 This incidence is controversial and has been historically underreported, with convenience samples often selected and a paucity of negative results presented.4,5 Nevertheless, recent meta-analysis assessed patient-based estimates of mean prevalence of peri-implantitis and peri-implant mucositis, yielding ranges of 1% to 47% and 19% to 50%, and prevalence of 22% and 43%, respectively.6 With more than 5 million dental implants placed each year in the United States alone according to the American Dental Association,7 these estimates imply that over 50% of implants will display some degree of peri-implant disease. Thus, peri-implant disease and implant failure are becoming principal health concerns that must be addressed and ultimately treated in a standardized manner.
As defined by the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions conducted by the American Academy of Periodontology, peri-implant mucositis is characterized by inflammation of soft tissue surrounding a dental implant without additional bone loss, excluding the physiologic bone loss that occurs immediately after implant placement.8,9 Peri-implantitis is defined as the inflammation of mucosa surrounding the implant with progressive loss of supporting peri-implant bone-thus, loss of osseointegration exceeding that of initial bone remodeling.2-4,8,10 Determining whether an implant is affected by peri-implant mucositis or peri-implantitis is done clinically and through radiologic examination of the surrounding bone level. The extent of peri-implant disease and etiologic or prognostic factors involved are more challenging to ascertain.
Various authors, notably Albrektsson et al,9 Misch et al,11 and Padial-Molina et al,12 have previously defined degrees of peri-implant disease and clinical assessment methods. These assessment methods, however, did not take into consideration the history of the implant, specifically placement conditions and soft/hard-tissue quality, or medical/social risk factors of the patient with regard to potential prognosis and treatment planning.9,12,13 These studies, among others, incorporate known risk factors and methods for clinical assessment that are supported by evidence-based systematic reviews. They allow for a distinction between a completely failed implant and degrees of an ailing and/or failing implant that may be restored to health.8,9,13-18
By definition, a failed dental implant is characterized as a non-salvageable implanted device that needs to be removed due to a significant amount of bone loss and/or the presence of mobility.12,14,17 Conversely, an ailing implant is a restored and primarily osseointegrated implant that begins to present with specific pathognomonic clinical,radiological, and microbiological patterns denoting peri-implant mucositis or peri-implantitis.8,15,16 If assessed and properly treated in the stages of failure prior to the compromise of implant osseointegration, an ailing implant may remain osseointegrated and have an acceptable long-term prognosis.9,12
Known mechanical causes of an ailing implant include but are not limited to improper surgical and prosthetic execution, low quality of surrounding bone and soft tissue, pathologic bone loss around the implant, and biological complications resulting in mucositis and peri-implant disease.9,12,13,16,18 Risk factors in an individual's medical, dental, and social history have been identified as playing a role in the progression from a healthy implant to an unsuccessful one. These include the presence of poor oral hygiene or pre-existing periodontitis, genetic predisposition for certain intraoral bacterial flora, uncontrolled diabetes, smoking history, alcohol consumption, systemic conditions, and functional habits leading to trauma.10,12,18-22
The aim of this study is to evaluate and characterize clinical and radiological criteria as well as historical data to assess implant health in the clinical setting. It establishes an algorithmic assessment system incorporating evidence-based diagnostic criteria that consistently and accurately differentiates between an ailing implant and a failed implant. The acquisition of the included criteria via review of high-quality evidence will allow for development of an objective scoring system to systematically determine an evidence-based prognosis for every implant assessed.
Materials and Methods
Two independent authors (MB, JG)conducted an electronic literature search between June 2017 and January 2018 of all articles evaluating risk factors for peri-implant disease in the following databases: PubMed/Medline, EMBASE, Scopus, and the Cochrane Library. The following search terms were used: "diagnosis" (OR) "prognosis" (AND) keywords "ailing implant," "peri implantitis," "peri implant mucositis," "peri implant osteitis," "failing implant," "peri implant disease."All articles, which were published before December 2018, were screened for inclusion and exclusion criteria by abstract and study title. Initially, 282 articles were found, and further analysis of article content allowed for the selection of 35 relevant articles for review. Further literature was added with a manual search external to the electronic one that enhanced high quality of basic implant evidence to sustain the present study.
Inclusion criteria were: assessment of risk factors for peri-implant disease and prognosis of ailing and failing implants as outcomes; systematic reviews, meta-analyses, and randomized clinical trials in humans and review articles; and publication date from November 2008 to November 2018. Exclusion criteria were: studies conducted as in vitro studies, in vivo; case reports; and articles that addressed only management of peri-implantitis.
All of the present authors compiled the necessary information for the assessment criteria via the aforementioned literature review and clinical experience. The assessment criteria are listed in Figure 1, Intake Form for Ailing Implant, and Figure 2, Diagnostic Form to Evaluate Implant Prognosis Based on Information Acquired on Intake.
Results and Discussion
Thirty-five articles were identified through the established inclusion and exclusion criteria. Evidence-based literature recognizes several parameters that are considered diagnostic of an ailing implant. These parameters include measurement of clinical and radiographic changes. Furthermore, a series of factors assessed in medical/social history contribute to the determination of an implant prognosis.
As a result of this literature review, three of the present authors (two oral and maxillofacial surgeons [TGW, HST] and one periodontist [RAH]) determined that classification of the diagnostic criteria falls into two categories: a routine comprehensive evaluation and an advanced problem-targeted evaluation that requires more specific and sensitive instruments to assess the health status of an implant. All of the authors further established an implant data acquisition form (Figure 1) that allows for objective implant staging in the comprehensive and advanced implant evaluation (Figure 2).
Comprehensive Implant Evaluation
The comprehensive implant evaluation consists of an assessment that is feasible for any clinician to perform when evaluating a potentially ailing implant. This evaluation begins with a full medical and social history of the patient and continues with further appraisal of the history of the implant being assessed. The medical and social history may include potential risk factors that contribute to the prognosis of the implant being evaluated. Select systemic medical conditions or a smoking history, for example, can negatively affect and perhaps compromise intraoral structures containing prostheses.17
The implant history is pertinent in that deficient treatment planning and subpar placement could compromise osseointegration, maintenance, and longevity of the implant. Furthermore, clinical and radiological characteristics of unsuccessful implants vary depending on the pathological process occurring. The relevant clinical and radiological features identified for assessment include a series of clinically measurable parameters that can be indicative of peri-implant disease according to current evidence-based literature.
Therefore, the comprehensive implant evaluation is separated into three sections: Part I: Medical and Social History, Part II: Clinical Implant Risk Assessment, and Part III: Implant History.
Part I: Medical and Social History
To ultimately classify an individual's medical risk, the following information needs to be completed on the data acquisition form (Figure 1): medical problems requiring ongoing care and their level of control; medications being taken by the patient; any substance use (alcohol, illicit drugs, tobacco); and surgical and dental history (periodontal health, oral hygiene, parafunctional habits, etc). Based on review of the literature and internal author discussion, the authors concur that patients may be classified according to medical and social history in three groups: low risk, moderate risk, and high risk.
Risk factors determined to be significant based on evidence-based literature and the authors' clinical experience are given point values with respect to severity (Figure 2). When an individual possesses any of the characteristics assigned a numerical value, these values are annotated on the comprehensive implant evaluation sheet (Figure 2), and the values are added together. Similarly, the combined implant history values yield a diagnosis of low, moderate, and high risk based on patient-reported history. It is important to note that neither medical history risk nor implant history are indicative of the current status of peri-implant health but are incorporated as a measure for prognosis.
Low risk (+1): Low-risk patients are considered healthy (American Society of Anesthesiologists [ASA]
Moderate risk (+2): Moderate-risk individuals are those who present with mild or severe systemic disease (ASA ≥II), poor oral hygiene, the presence of calculus or significant accumulations of plaque, and/or a history of periodontitis. Individuals in this category are assigned a +2 score per factor.
High risk (+3): Patients classified in the high-risk category are considered more likely to present implant complications due to having severe medical conditions that have been shown to affect implant osseointegration/bone healing. Patients included in this group have uncontrolled diabetes (HbA1C >10), use tobacco or areca nuts, have had previous head and neck radiation and/or bone pathology, are receiving ongoing intravenous antiresorptive medications, and/or are immunosuppressed individuals. Patients in this group are assigned +3 points per factor identified.
Part II: Clinical Implant Risk Assessment
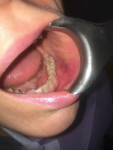
Clinical appearance of implant/soft tissue (swelling, visible threads, erythema): In early stages, peri-implant lesions are typically asymptomatic. Peri-implant soft tissues present with signs of enlargement, erythema, and inflammation, and these symptoms can be reversed if treated early.1,17,23-25
Probing depth: Probing depth, a highly sensitive test and a key criterion in assessing the severity of ailing implants, is determined by using a periodontal probe to detect the greatest distance between the gingival margin and the base of the peri-implant pocket. This assessment should be performed with light pressure (0.25 Ncm) and done routinely after crown placement to compare progression of peri-implant disease versus initiation of healing in the site.14,19,23,26 Increased probing depth is associated with the loss of epithelial attachment and the presence of inflammation.27 According to Passi et al in 2017, the proposed probing depth parameters used to assess peri-implant disease are as follows: <4 mm = healthy or minimal soft-tissue inflammation (+0); 4 mm to 6 mm = initial peri-implantitis (+1); >6 mm to 8 mm = moderate peri-implantitis (+2); >8 mm = severe peri-implantitis (+3).13
Bleeding on probing (+1): Bleeding on probing (BOP) in peri-implant tissues is a highly reliable method for assessing soft-tissue inflammation.19,26 In healthy peri-implant conditions, periodontal probing will not cause bleeding when the probe enters into the biological width. According to literature, BOP is present 67% of the time in peri-implant mucositis and 91% in peri-implantitis.12-15 Clinicians should be aware that a recent systematic review has quantified a high false-negative rate in BOP as an indicator of disease. The study established that only 24% of all implants with BOP are actually affected by peri-implant disease.28
Suppuration (+2): The presence of suppuration is related to an existing peri-implant infection or inflammatory lesion that indicates the possible compromise of the peri-implant marginal bone.1,12-14 The cause of the infection must be assessed carefully and include analysis of the oral hygiene of the patient and the possible presence of residual cement. Some researchers consider suppuration to be a higher sensitivity test compared to BOP because it is directly related to attachment loss.26
Presence of plaque/calculus: The presence of plaque and calculus on an implant-supported restoration is a risk factor that may compromise peri-implant health. Poor oral hygiene is associated with increased risk of peri-implant mucositis, which if not treated can lead to marginal bone loss over time.1,24 Patient control of oral hygiene is critical and should be emphasized at every recall visit. Patients should be given proper oral health instructions prior to implant placement, and clinicians should take steps to ensure that these patients maintain their oral health.12,14,15,19,25,29 As with other parameters, plaque index should be assessed at every follow-up visit.
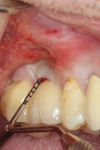
Restoration (fixed vs. removable, cemented vs. screw-retained, occlusion, contour, etc): The type of restoration placed needs to be carefully assessed. An improper restoration is a common iatrogenic cause of implant compromise.12 Improper positioning of the implant, overcontouring of the superstructure (+1), and incorrect seating of the abutment (+3) lead to difficulty in cleaning the peri-implant area (Figure 3).24 Difficulty in implant cleansability is a major risk factor for peri-implantitis. The design of the prosthesis for effective oral hygiene accessibility is a crucial factor in the algorithm. A poorly designed implant-supported restoration may increase plaque retention.12,14,29,30 These factors in conjunction with occlusal overloading may cause extraneous forces on the implant structure that lead to potential fracture and failure. Occlusal overloading (+1) is a key factor that could be associated with iatrogenic force on the implant complex stimulating bone resorption around the implant. Extensive overload may result in complete disintegration and implant failure.31 Loosening of screws, fracture of the restoration, and detachment of the crown are signs indicative of occlusal overloading.12 Parafunction and bruxism (+2) also are known risk factors that transmit pathological forces to the implant surface.
Cement-retained restorations are particularly useful for correcting improper position and angulation of an implant. Conversely, cement must be placed carefully and removed thoroughly. Residual cement (+2) in the sulcus is a principal cause of early peri-implant disease.19,32 A recent review revealed that excess cement could lead to 85% of iatrogenic bone loss and titanium leakage. The incidence increases when this risk factor is associated with previous history of periodontitis.32
Percussion: Another tool to detect implant osseointegration is percussion. An integrated implant presents a high-pitched sound while a non-integrated implant exhibits a low, dull sound (+3).23 The percussion test has to be administered laterally and apically with a force up to 500 g with the inactive parts of two opposing instruments or by Periotest® (Medizintechnik Gulden, med-gulden.com). The occurrence of concomitant pain upon percussion implies the presence of actively inflamed peri-implant tissue. If pain on percussion occurs during implant function, the implant is considered failed (+X).14,23
Mobility (+X): Assessment of implant mobility is straightforward. Mobility is tested using pressure with opposing instruments on the supragingival portion of the implant or on the restoration that has been placed. It is important to differentiate between implant mobility and mobility of the superstructure alone, which could be related to overloading. A highly sensitive implant stability quotient (ISQ) system (Osstell®, Osstell, osstell.com) may be used to diagnose implant osseointegration, measuring the implant oscillation frequency on the bone thereby providing a stability measurement.33 Literature maintains that implant mobility indicates the terminal stage of peri-implant disease characterized by complete loss of bone-to-implant contact and absence of osseointegration.17,34 Thus, the presence of mobility is a pathognomonic sign directly associated with implant failure.12,14,23,34
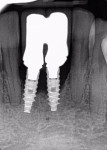
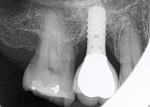
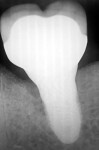
Percent and region of bone loss around implant via 2-dimensional radiologic assessment: Bone loss is conventionally detected radiologically by bitewing, periapical (PA), or panoramic (PAN) x-rays. The physiologic bone loss ratio of an implant is 0.1 mm to 0.2 mm yearly for the first 5 years (up to a total of 1.5 mm).14,23 To correctly visualize interproximal marginal bone loss around the implant, a PA is the most useful radiograph.12,14,35 The PA provides high-quality images with the necessary detail to allow detection of early defects.35 A PAN gives an excellent view of the upper and lower jaws but shows less detail and is not currently the standard of care in early stages.1,12,15 A limitation of conventional radiological assessment is that the buccal-lingual plane of the implant cannot be evaluated (Figure 4 through Figure 8). Nevertheless, through a proper variation of the angulation, it is possible to obtain additional information about the peri-implant bone loss (Figure 9 and Figure 10). Irrespective of the aforementioned limitation, the following staging system that addresses the severity of peri-implant disease may be used to characterize bone loss1,12,15:
early peri-implantitis/early ailing implant (≤2 mm or <20% bone loss) (+1)
moderate peri-implantitis/moderate ailing implant (>2 mm to ≤4 mm or 20% to 50% bone loss) (+2)
advanced peri-implantitis (>4 mm or >50% bone loss) (+3)
Part III: Implant History
Date of placement: Assessing the date of placement can aid in determining whether the identified clinical and radiological characteristics are physiological or pathological.
Previous implant loss (+2): Previous implant loss is a risk factor for future implant failure. Causes of previous implant failure must be evaluated carefully.11 In the absence of a clear pathologic finding or cause, an advanced assessment should then be conducted (see "Advanced Evaluation of an Ailing Implant" section below).
Grafted site: This refers to soft versus hard tissue, socket preservation, guided bone regeneration (GBR), sinus lift, etc.
Any bone graft in site? (+1): A recent systematic review showed a slight difference between an augmented site and a pristine site in terms of peri-implant bone loss and implant failure, with augmented sites tending to be more susceptible to peri-implantitis.36
Immediate (+1), delayed, or late placement: Botticelli et al showed that immediate socket implants that are placed without grafting result in as much site collapse as non-grafted sockets.37 Immediate implant placement requires sufficient clinical experience and an abundance of soft tissue and bone in the treatment area. Immediate implants have been shown to have significant susceptibility to crestal bone loss and an incidence of peri-implantitis.38
Size and type of implant: The implant must be the proper size to correspond to the region of placement with consideration given to bone density, volume, and type. Potential mechanical occlusal forces must be assessed before implant placement. Posterior implants may require a wider diameter to support a greater occlusal force in comparison with premolars and incisors. Implants should be placed at a correct distance between each other and between teeth. Tarnow et al suggested a minimum of 1.5 mm proximally between implant and teeth and 3 mm of distance between two implants.39 Buser et al and Grunder et al both advised that to achieve an esthetic outcome 2 mm of bone was necessary on the facial and lingual aspects of implants.40,41
Implant surface: Evidence shows that rough surfaces tend to accumulate more plaque, allowing for biofilm formation and making cleansability more difficult.17 The implant surface could play an important role in both the development and treatment of ailing implants.12,42
Region of placement: esthetic, non-esthetic: The presence of a buccal concavity (+1), which most often occurs in the anterior region, may necessitate a prior or simultaneous osseous augmentation. This deficiency could lead to future bone or graft resorption and exposure of threads. In a study, Huynh-Ba et al showed that the majority of extraction sites in the premaxilla displayed less than 1 mm of buccal plate.43 Therefore, alveolar ridge preservation is suggested in esthetic areas to maintain the ideal 2 mm of thickness of bone.40,41
Soft-tissue conditions: gingival biotype, keratinized soft tissue, gingival recession: Thin gingival biotype (+1) has a higher risk of recession, and often a soft-tissue graft might be needed before surgery to prevent pathologic recession.23
A recent study showed that when <2 mm of keratinized soft tissue (+2) is present, there is a poor prognosis for implants with a lack of or a reduced amount of attached gingiva (<2 mm).27 Absence of attached gingiva is related to presence of plaque accumulation and peri-implant mucositis, especially in posterior areas.15 An adequate zone of keratinized tissue facilitates the stabilization of an implant and ultimately the longevity of the restoration.27,44
Advanced Evaluation of an Ailing Implant
Secondary to the routine examination, an advanced evaluation is conducted to determine the prognosis and stage of an ailing implant. In early stages and in more complex cases, two criteria, 3-dimensional (3D) assessment of bone loss and microbiological determinant analysis, may aid clinicians in the diagnosis of a problematic implant.
3D Assessment of Bone Loss
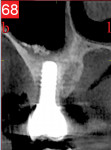
Cone-beam computed tomography (CBCT) is an advanced tool that can be used to characterize bone loss. It allows for 3D reconstruction of the region of interest and eliminates anatomical superposition seen in other adjunct radiologic techniques. This helps the clinician visualize buccal-lingual bone plates and the precise thickness of the alveolar ridge. Thus, CBCT is the current standard of care in decision-making and treatment planning prior to implant placement.45
Furthermore, CBCT yields an exceptionally high-quality image of every cut in the vertical, horizontal, and oblique planes for highly accurate detection of circumferential bone loss around implant threads. Although CBCT is extremely useful, high-density artifacts derived from amalgam restorations and metallic crowns can affect image interpretation and produce distortion.46 Fienitz et al postulated that the evaluation of peri-implant bone regeneration with CBCT is inaccurate.47 Also, the quality of results and their interpretation could be influenced by different manufacturers' CBCT units producing images of varying spatial resolution (voxel sizes), as well as different fields of view (FOV), patient positioning systems, or durations of scan.48 Furthermore, radiation dosages and costs are higher with CBCT compared to conventional bitewing, PA, or PAN radiographs.
Interestingly, Jacobs et al recently proposed that CBCT is not the gold standard in evaluating peri-implant osseous defects.49 However, a systematic review by Bornstein et al stated that CBCT is recommended in preoperative evaluation with regard to very large osseous defects, buccal wall loss when not visualized in PA (as demonstrated in Figure 5 and Figure 6), pathologies, or in cases of severe complications involving inferior alveolar nerve damage or sinus infection or perforation.50 Therefore, the present authors conclude that the PA radiograph is currently the most reliable and accurate imaging modality for detection of circumferential peri-implant bone defects, having been shown to perform significantly better than CBCT.49
Microbiological Determinant Analysis
The presence of pathological microbial flora may be the most important direct cause of implant failure. The interaction between the structure of the implant and colonizing microflora is a critical factor that can allow for either the success of implant placement and adequate stability or significant implant bone loss and failure to obtain stability. The correlation between microbial counts and presence of peri-implant mucositis and peri-implantitis is directly proportional. 24,25
Microbiological samples are typically taken with endodontic paper points in different locations and sent to a laboratory for analysis of the periodontal pathogens colonizing the region. A recent study evidenced that the microfloras that colonize 30% of an implant site are formed by the following bacteria: Tannerella forsythia, Porphyromonas gingivalis, Treponema socranskii, Staphylococcus aureus, Staphylococcus anaerobius, Staphylococcus intermedius,and Streptococcus mitis.25A systematic review and meta-analysis showed that gram-negative pathogens (P. gingivalis, Treponema denticola, Prevotella intermedia)are present more often than gram-positive pathogens in peri-implantitis cases and are responsible for increasing marginal bone loss and, therefore, lead to the progression of disease.24 Literature has revealed that bacteria related to peri-implantitis has shown a more elaborate composition than bacteria related to periodontitis.51,52 Another study, though, showed interesting findings on cross-infection between periodontal sites and implant sites, highlighting connections between the two pathologies. This finding is noted in patients with chronic periodontitis. Further periodontal debridement and other treatments need to be performed to resolve disease before implant placement, and these patients should be re-evaluated periodically.52
Quantitative Evaluation
The quantitative evaluation is an objective assessment and acquisition of data regarding the health status of existing implants. The parameters for a comprehensive implant health evaluation are represented in Figure 1 and Figure 2.If the final implant value (i) is less than 4, the prognosis is favorable. If the calculated implant value is between 4 and 8, the prognosis is guarded considering the greater number and severity of clinical risk factors. An implant value of more than 8 and less than 12 means the prognosis is poor, and any implant with an implant value of more than 12 or possessing one of the critical factors described below is considered hopeless.
Medical history and implant history must be considered in every evaluation, but these factors are not necessarily indicative of current status of peri-implant health. They are incorporated solely as a measure for prognosis as supported by the literature evaluated.
The importance of vitamin D supplements to systemic health and dental implants has been suggested in the literature.53 Vitamin D plays a significant role in the pathways controlling absorption of calcium and formation of new bone. Future studies should examine the possibility that a lack of vitamin D could affect osseointegration and the maintenance of dental implants.
Some authors have reported the existence of titanium intolerance or "foreign body" reaction in a small subset of individuals.54 When unexpected peri-implant bone loss or unexplained implant failure occurs, further analysis (eg, leukocyte transmigration test or memory lymphocyte immunostimulation assay [MELISA] test) can be conducted to determine the possibility of hypersensitivity to implanted dental materials. Any such assessment should be done with caution because currently there is not enough evidence to support these findings.
Additionally, recent literature has considered a possible association between the host immune response and dental implant success. Metallic ions and degradation products released by the implanted structure have been demonstrated to induce proinflammatory cytokine activation that leads to an osteolytic cascade compromising implant osseointegration.55,56 Petkovic-Curcin et al demonstrated that the genotype tumor necrosis α-308 GA/AA is a predisposing factor to a more severe peri-implantitis.57 Other studies have also begun to evaluate a possible association between interleukin-1 (IL-1) and IL-1β genotypeand peri-implant bone loss.58,59Andreiotelli et al has affirmed that elevated IL-1 in crevicular fluid could increase the pathophysiology of peri-implantitis.59 Given the paucity of data on this topic, further investigation is indicated to give the subject more validity, especially because the study of genotyping individual cytokines could contribute to implant assessment and prognosis of outcome.
The findings of this study suggest that in order to assess an ailing implant, a profound clinical and comprehensive radiological evaluation must be performed. A periapical radiograph is the most recommended image for this purpose. Mobility and pain during mastication are features that are indicative of implant failure. In advanced cases of progressive failure or larger alveolar defects, 3D analysis is advised. These assessments should be completed to properly identify the stage of peri-implant disease and establish an ideal management plan.
Conclusion
To evaluate an ailing implant, a clinical and comprehensive evaluation that includes the medical and implant history must be performed. The prognosis of ailing implants is contingent upon early detection and management. The parameters for the proposed comprehensive implant evaluation are feasible in the clinical setting at any dental institution or private office. At a minimum, the authors advise evaluating implant and restoration design and performing periodontal probing, periapical radiographic evaluation, and mobility and percussion testing.
The implant assessment algorithm presented will enable a succinct clinical approach to evaluate and treat ailing implants and facilitate the establishment of a clinical management algorithm. Recent studies indicate that future assessments may include serologic testing (including biological markers, vitamin D, interleukins, and LDL), which may possess a predictive role for the prognosis of ailing implants.
Disclosure
The authors had no disclosures to report.
About the Authors
Marco Bergamini, DDS
Oral and Maxillofacial Surgery International Resident, Department of Oral and Maxillofacial Surgery, New York University College of Dentistry, New York, New York
Juliana Gomez, DDS
Post-graduate Year 1 Resident, Department of Oral and Maxillofacial Surgery, Ascension Macomb-Oakland Hospital, Detroit, Michigan
Huzefa S. Talib, BDS
Clinical Associate Professor, Department of Oral and Maxillofacial Surgery, New York University College of Dentistry, New York, New York
Robert A. Horowitz, DDS
Adjunct Clinical Assistant Professor, Department of Oral and Maxillofacial Surgery, New York University College of Dentistry, New York, New York
Thomas G. Wiedemann, MD, DDS, PhD
Clinical Assistant Professor, Department of Oral and Maxillofacial Surgery, New York University College of Dentistry, New York, New York
References
1. Atieh MA, Alsabeeha NHM, Faggion CM Jr, Duncan WJ. The frequency of peri-implant diseases: a systematic review and meta-analysis. J Periodontol. 2013;84(11):1586-1598.
2. Koldsland OC, Scheie AA, Aass AM. Prevalence of peri-implantitis related to severity of the disease with different degrees of bone loss. J Periodontol. 2010;81(2):231-238.
3. Lee CT, Huang YW, Zhu L, Weltman R. Prevalences of peri-implantitis and peri-implant mucositis: systematic review and meta-analysis. J Dent. 2017;62:1-12.
4. Salvi GE, Cosgarea R, Sculean A. Prevalence and mechanisms of peri-implant diseases. J Dent Res. 2017;96(1):31-37.
5. Tarnow DP. Increasing prevalence of peri-implantitis: How will we manage? J Dent Res. 2016;95(1):7-8.
6. Derks J, Tomasi C. Peri-implant health and disease: a systematic review of current epidemiology. J Clin Periodontol. 2015;42(suppl 16):158-171.
7. Dental Implants Market Size, Share & Trends Analysis Report By Product (Titanium Implants, Zirconium Implants), By Region (North America, Europe, Asia Pacific, Latin America, MEA), And Segment Forecasts, 2018-2024. Personalized Medicine Market Analysis By Product And Segment Forecasts To 2022. Report ID: GVR-1-68038-566-3. San Francisco, CA: Grand View Research, Inc;2018.
8. Renvert S, Persson GR, Pirih FQ, Camargo PM. Peri-implant health, peri-implant mucositis, and peri-implantitis: case definitions and diagnostic considerations. J Periodontol. 2018;45(suppl 20):S278-S285.
9. Albrektsson T, Zarb GA, Worthington P, Eriksson AR. The long-term efficacy of currently used dental implants: a review and proposed criteria of success. Int J Oral Maxillofac Implants. 1986;1(1):11-25.
10. Schwarz F, Derks J, Monje A, Wang HL. Peri-implantitis. J Periodontol. 2018;89(suppl 1):S267-S290.
11. Misch CE, Perel ML, Wang HL, et al. Implant success, survival, and failure: the International Congress of Oral Implantologists (ICOI) Pisa Consensus Conference. Implant Dent. 2008;17(1):5-15.
12. Padial-Molina M, Suarez F, Rios HF, et al. Guidelines for the diagnosis and treatment of peri-implant diseases. Int J Periodontics Restorative Dent. 2014;34(6):e102-e111.
13. Passi D, Singh M, Dutta SR, et al. Newer proposed classification of peri-implant defects: a critical update. J Oral Biol Craniofac Res. 2017;7(1):58-61.
14. Ramanauskaite A, Juodzbalys G. Diagnostic principles of peri-implantitis: a systematic review and guidelines for peri-implantitis diagnosis proposal. J Oral Maxillofac Res. 2016;7(3):e8.
15. Peri-implant mucositis and peri-implantitis: a current understanding of their diagnoses and clinical implications. J Periodontol. 2013;84(4):436-443.
16. Dreyer H, Grischke J, Tiede C, et al. Epidemiology and risk factors of peri-implantitis: a systematic review. J Periodontal Res. 2018;53(5):657-681.
17. Heitz-Mayfield LJ. Peri-implant diseases: diagnosis and risk indicators. J Clin Periodontol. 2008;35(8 suppl):292-304.
18. Lang NP, Berglundh T, Working Group 4 of Seventh European Workshop on Periodontology. Periimplant diseases: Where are we now?-Consensus of the Seventh European Workshop on Periodontology. J Clin Periodontol. 2011;38(suppl 11):178-181.
19. Strietzel FP, Reichart PA, Kale A, et al. Smoking interferes with the prognosis of dental implant treatment: a systematic review and meta-analysis. J Clin Periodontol. 2007;34(6):523-544.
20. Schou S, Holmstrup P, Worthington HV, Esposito M. Outcome of implant therapy in patients with previous tooth loss due to periodontitis. Clin Oral Implants Res. 2006;17(suppl 2):104-123.
21. Galindo-Moreno P, Fauri M, Avila-Ortiz G, et al. Influence of alcohol and tobacco habits on peri-implant marginal bone loss: a prospective study. Clin Oral Implants Res. 2005;16(5):579-586.
22. Lindhe J, Meyle J, Group D of European Workshop on Periodontology. Peri-implant diseases: consensus report of the Sixth European Workshop on Periodontology. J Clin Periodontol. 2008;35(8 suppl):282-285.
23. Alani A, Kelleher M, Bishop K. Peri-implantitis. Part 1: Scope of the problem. Br Dent J. 2014;217(6):281-287.
24. Tallarico M, Cannullo L, Caneva M, Özcan M. Microbial colonization at the implant-abutment interface and its possible influence on periimplantitis: a systematic review and meta-analysis. J Prosthodont Res. 2017;61(3):233-241.
25. Padial-Molina M, Lopez-Martinez J, O'Valle F, Galindo-Moreno P. Microbial profiles and detection techniques in peri-implant diseases: a systematic review. J Oral Maxillofac Res. 2016;7(3):e10.
26. Lin GH, Kapila Y, Wang HL. Parameters to define peri-implantitis: a review and a proposed multi-domain scale. J Oral Implantol. 2017;43(6):491-496.
27. Araujo MG, Lindhe J. Peri-implant health. J Periodontol. 2018;89(suppl 1):S249-S256.
28. Hashim D, Cionca N, Combescure C, Mombelli A. The diagnosis of peri-implantitis: a systematic review on the predictive value of bleeding on probing. Clin Oral Implants Res. 2018;29(suppl 16):276-293.
29. Nguyen-Hieu T, Borghetti A, Aboudharam G. Peri-implantitis: from diagnosis to therapeutics. J Investig Clin Dent. 2012;3(2):79-94.
30. Tallarico M, Cannullo L, Wang HL, et al. Classification systems for peri-implantitis: a narrative review with a proposal of a new evidence-based etiology codification. Int J Oral Maxillofac Implants. 2018;33(4):871-879.
31. Piattelli A, Scarano A, Faero L, et al. Clinical and histologic aspects of dental implants removed due to mobility. J Periodontol. 2003;74(3):385-390.
32. Valente NA, Andreana S. Peri-implant disease: What we know and what we need to know. J Periodontal Implant Sci. 2016;46(3):136-151.
33. Han J, Lulic M, Lang NP. Factors influencing resonance frequency analysis assessed by Osstell mentor during implant tissue integration: II. Implant surface modifications and implant diameter. Clin Oral Implants Res. 2010;21(6):605-611.
34. Greenstein G, Cavallaro J. Failed dental implants: diagnosis, removal and survival of reimplantations. J Am Dent Assoc. 2014;145(8):835-842.
35. García-García M, Mir-Mari J, Benic GI, et al. Accuracy of periapical radiography in assessing bone level in implants affected by peri-implantitis: a cross-sectional study. J Clin Periodontol. 2016;43(1):85-91.
36. Salvi GE, Monje A, Tomasi C. Long-term biological complications of dental implants placed either in pristine or in augmented sites: a systematic review and meta-analysis. Clin Oral Implants Res. 2018;29
(suppl 16):294-310.
37. Botticelli D, Berglundh T, Lindhe J. Hard-tissue alterations following immediate implant placement in extraction sites. J Clin Periodontol. 2004;31(10):820-828.
38. Rodrigo D, Martin C, Sanz M. Biological complications and peri-implant clinical and radiographic changes at immediately placed dental implants. A prospective 5-year cohort study. Clin Oral Implants Res. 2012;23(10):1224-1231.
39. Tarnow DP, Cho SC, Wallace SS. The effect of inter-implant distance on the height of inter-implant bone crest. J Periodontol. 2000;71(4):546-549.
40. Grunder U, Gracis S, Capelli M. Influence of the 3-D bone-to-implant relationship on esthetics. Int J Periodontics Restorative Dent. 2005;25(2):113-119.
41. Buser D, Martin W, Belser UC. Optimizing esthetics for implant restorations in the anterior maxilla: anatomic and surgical considerations. Int J Oral Maxillofac Implants. 2004;19(suppl):43-61.
42. Albouy JP, Abrahamsson I, Persson LG, Berglundh T. Implant surface characteristics influence the outcome of treatment of peri-implantitis: an experimental study in dogs. J Clin Periodontol. 2011;38(1):58-64.
43. Huynh-Ba G, Pjetursson BE, Sanz M, et al. Analysis of the socket bone wall dimensions in the upper maxilla in relation to immediate implant placement. Clin Oral Implants Res. 2010;21(1):37-42.
44. Marcantonio C, Nicoli LG, Marcantonio E, Zandim-Barcelos DL. Prevalence and possible risk factors of peri-implantitis: a concept review. J Contemp Dent Pract. 2015;16(9):750-757.
45. Bornstein MM, Horner K, Jacobs R. Use of cone beam computed tomography in implant dentistry: current concepts, indications and limitations for clinical practice and research. Periodontol. 2000;73
(1):51-72.
46. Shulze RK, Berndt D, d'Hoedt B. On cone-beam computed tomography artifacts induced by titanium implants. Clin Oral Implants Res. 2010;21(1):100-107.
47. Fienitz T, Schwarz F, Ritter L, et al. Accuracy of cone beam computed tomography in assessing peri-implant bone defect regeneration: a histologically controlled study in dogs. Clin Oral Implants Res. 2012;23(7):882-887.
48. Razavi T, Palmer RM, Davies J, et al. Accuracy of measuring the cortical bone thickness adjacent to dental implants using cone beam computed tomography. Clin Oral Implants Res. 2010;21(7):718-725.
49. Jacobs R, Vranckx M, Vanderstuyft T, et al. CBCT vs other imaging modalities to assess peri-implant bone and diagnose complications: a systematic review. Eur J Oral Implantol. 2018;11(suppl 1):77-92.
50. Bornstein MM, Scarfe WC, Vaughn VM, Jacobs R. Cone beam computed tomography in implant dentistry: a systematic review focusing on guidelines, indications, and radiation dose risks. Int J Oral Maxillofac Implants. 2014;29(suppl):55-77.
51. Koyanagi T, Sakamoto M, Takeuchi Y, et al. Comprehensive microbiological findings in peri-implantitis and periodontitis. J Clin Periodontol. 2013;40(3):218-226.
52. Gouvoussis J, Sindhusake D, Yeung S. Cross-infection from periodontitis sites to failing implant sites in the same mouth. Int J Oral Maxillofac Implants. 1997;12(5):666-673.
53. Salomo-Coll O, Mate-Sanchez de Val JE, Ramırez-Fernandez MP, et al. Topical applications of vitamin D on implant surface for bone-to-implant contact enhance: a pilot study in dogs Part II. Clin Oral Implants Res. 2016;27(7):896-903.
54. Albrektsson T, Canullo L, Cochran D, De Bruyn H. "Peri-implantitis": a complication of a foreign body or a man-made "disease." Facts and Fiction. Clin Implant Dent Relat Res. 2016;18(4):840-849.
55. Noronha Oliveira M, Schunemann WVH, Mathew MT, et al. Can degradation products released from dental implants affect peri-implant tissues? J Periodontal Res. 2018;53(1):1-11.
56. Heyman O, Koren N, Mizraji G, et al. Impaired differentiation of Langerhans cells in the murine oral epithelium adjacent to titanium dental implants. Front Immunol. 2018;9:1712.
57. Petkovic-Curcin A, Zeljic
K, Cikota-Aleksic B, et al. Association of cytokine gene polymorphism with peri-implantitis risk. Int J Oral Maxillofac Implants. 2017;32(5):e241-e248.
58. Dirschnabel AJ, Alvim-Pereira F, Alvim-Pereira CC, et al. Analysis of the association of IL1B(C-511T) polymorphism with dental implant loss and the clusterization phenomenon. Clin Oral Implants Res. 2011;22(11):
1235-1241.
59. Andreiotelli M, Koutayas SO, Madianos PN, Strub JR. Relationship between interleukin-1 genotype and peri-implantitis: a literature review. Quintessence Int. 2008;39(4):289-298.