Apical Barrier Technique with Mineral Trioxide Aggregate Using Internal Matrix: A Case Report
Ravi Sood, BDS; Manoj Kumar Hans, MDS; and Shashit Shetty, MDS
Abstract
Apexification is a procedure well recognized and accepted by clinicians and researchers alike. Calcium hydroxide has long been used as an apexification material. The newly developed mineral trioxide aggregate (MTA) has also been shown to be effective in artificial root-end closure. The major problem in cases of a wide open apex is the need to limit the apexification material at the apex, thus avoiding the extrusion of a large amount of material into the periodontal tissue. The use of a matrix is advisable because its placement in the area of bone destruction provides a base on which the sealing material, especially MTA, can be placed and packed. This article presents a case report in which an internal matrix was formed using absorbable collagen sponge to provide a base for MTA placement at the apex.
One serious dental healthcare challenge is when pulp devitalizes in an immature permanent tooth after traumatic injury. Root canal therapy is the most effective treatment; however, obturation may be difficult if roots are not completely formed. Root canal rehabilitation in such cases is significantly challenging because of the size of the canal; the thin and fragile dentin walls; and the large, open apex. Calcium hydroxide (Ca(OH)2) has been the material of choice for apexification.1 Recently, mineral trioxide aggregate (MTA) has been popularized in endodontics due to an abundant amount of research indicating the beneficial properties of the material in terms of biocompatibility, ease of manipulation and placement, and a wide array of applications.2 It has been used for procedures ranging from direct pulp capping to perforation repair as well as for inducing an artificial barrier in open-apex cases.3
A study comparing the effectiveness of Ca(OH)2 versus MTA has shown that MTA has been found to be as effective as Ca(OH)2 for the treatment of cases with an open apex. MTA has shown to be effective in performing the same procedure in a considerably lesser period of time with predictable results. The authors also concluded that the chances for biological calcific bridge formation are favorable when the root canal apices are flush or underfilled with MTA.4
Lemon5 advocated the use of a matrix when the perforation diameter is larger than 1 mm to avoid extrusion of the sealing material. The use of a matrix is advisable since its placement in the area of bone destruction provides a base on which the sealing material, especially MTA, can be placed and packed in the perforation.6,7 Several materials have been recommended to create a matrix in cases of perforations as well as teeth with incomplete formation of apex. These materials include calcium hydroxide, hydroxyapatite, absorbable collagen, and calcium sulphate.6,8
This report presents a case of large periapical abscess associated with a tooth with an open apex that was treated nonsurgically, and apexification was carried out with MTA using an internal matrix.
Case Report
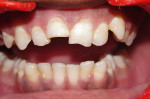
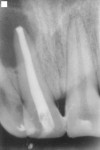
A 15-year-old male patient presented with a primary complaint of a discolored maxillary anterior tooth. Clinical examination revealed fracture in both the upper right and upper left central incisors (Figure 1). The patient had a history of trauma that occurred 8 years previously. No significant medical history was reported. A periapical radiograph demonstrated an incompletely formed root apex and a large radiolucent area around the upper right central and lateral incisors (Figure 2). The upper right central incisor elicited negative responses on electric as well as thermal pulp testing, although adjacent and contralateral incisors elicited positive responses. Based on subjective and objective findings, a diagnosis was made of Ellis Class IV fracture in the upper right central incisor and Ellis Class I fracture in the upper left central incisor. A decision was made to perform apexification in the upper right central incisor along with a composite restoration in the upper left lateral incisor.
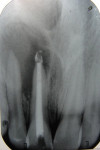
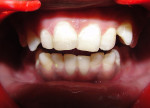
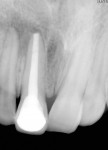
With a rubber dam in place and after access to the pulp chamber was gained, a working length radiograph of the upper right central incisor was taken (Figure 3). The working length was established 1-mm short of the radiographic apex. Cleaning and shaping was carefully performed using stainless-steel K-files. Copious irrigation with alternating solutions of 2.5% sodium hypochlorite and 17% EDTA was conducted throughout the cleaning and shaping procedure. After drying the canal system with paper points, calcium hydroxide paste (Metapex™, MetaBiomed, www.meta-biomed.com) was placed in the root canal system for 2 weeks, and a temporary coronal seal was established with Cavit™ (3M ESPE, www.3MESPE.com) (Figure 4). Calcium hydroxide was used to disinfect the root canal system in order to reduce root canal infection below a certain threshold level. After 2 weeks of Ca(OH)2 therapy, dressing was removed from the root canal system using sodium hypochlorite irrigation and endodontic files. Small pieces of CollaTape® (Zimmer Dental, www.zimmerdental.com), a synthetic collagen material, were gently compacted using hand pluggers to produce a barrier at the level of the apex. Gray MTA-Angelus (Angelus, www.angelus.ind.br) plug was placed in the canal with a carrier and pluggers to form an apical plug approximately 4-mm thick. Placement of MTA was confirmed radiographically (Figure 5). A small amount of Ca(OH)2 was extruded into the periapical region during removal (Figure 5). A sterile moist cotton pellet was placed over the MTA, and the access cavity was sealed with Cavit. The patient was recalled after 24 hours, and hardness of the apical barrier was checked at the follow-up appointment. Subsequently, backfill was performed using Obtura (Obtura Spartan Endodontics, www.obtura.com) (Figure 6). The access cavity was sealed and both fractured teeth restored with composite resin (Figure 7). The patient was recalled after every 3 months to check the vitality status of the upper right lateral incisor. The tooth was restored with a porcelain-fused-to-metal (PFM) crown, and periapical healing was confirmed by a radiograph at 1 year (Figure 8). The upper right lateral incisor elicited positive response on electric as well as thermal pulp testing after 1 year.
Discussion
A number of methods have been recommended for artificial apical closure. For a long time, Ca(OH)2 has been used as an apexification material. Newly developed MTA has also been shown to be effective in artificial root-end closure. Both materials were found to stimulate the formation of mineralized tissue, thereby providing an adequate seal in the apical region.9 MTA, a biocompatible material, can be used to create a physical barrier that also helps in formation of bone and periodontium around its interface.2 In addition to biocompatibility, MTA has been reported to have such desirable properties as fibroblast stimulation, antimicrobial activity, and sealing capacity with an ability to set in a moist environment.10 Ca(OH)2 was used to disinfect the root canal system so that root canal infection would be reduced below a certain threshold level. Although earlier studies recommended the use of a Ca(OH)2 prior to MTA placement for barrier formation, recent studies report that the initial use of Ca(OH)2 paste was not necessary for apexification to occur, and apexification has shown to be strongly related to the extrusion of MTA and formation of barriers beyond the limits of the root canal walls.2 The major problem in cases of a wide open apex is the need to limit the apexification material to the perforation, thus avoiding the extrusion of a large amount of material into the periodontal tissue.11
A large volume of the extruded material may set before it disintegrates and gets resorbed. This might result in the persistence of the inflammatory process, which may complicate or even prevent repair of the tissue.11 Use of a matrix prevents the extrusion of the material into the periodontal tissues, reduces leakage in the sealing material, and allows a favorable response of the periodontal tissues.
Orthograde obturation with MTA as an apexification material also represents an attempt to strengthen immature tooth roots.12
Though MTA is not bonded to dentin, interaction of the released calcium and hydroxyl ions of MTA with a phosphate-containing synthetic body fluid results in the formation of apatite-like interfacial deposits.13 These deposits fill any gaps induced during the material shrinkage phase and improve the frictional resistance of MTA to the root canal walls, which likely accounts for the seal of MTA in orthograde obturation.14
Conclusion
The use of an internal matrix of absorbable collagen provides good control for condensation of MTA against the internal matrix. In the present case, the area of disease healed because the root canal infection was reduced below a certain threshold by placing Ca(OH)2 for 2 weeks, while the role of the MTA plug was to prevent additional toxins from communicating with the periradicular tissues. If the plug had not been placed correctly, the seal would be poor. Therefore, the CollaTape matrix enabled the MTA to be placed correctly, which in turn maximized the seal and, thus, healing took place. Further studies are needed to develop the internal matrix placement approach into a standardized method so that the matrix will be placed correctly each time it is used.
About the Authors
Ravi Sood, BDS
Post-Graduate Student
Department of Conservative Dentistry and Endodontics
K.D. Dental College and Hospital
Mathura, Uttar Pradesh, India
Manoj Kumar Hans, MDS
Reader
Department of Conservative Dentistry and Endodontics
K.D. Dental College and Hospital
Mathura, Uttar Pradesh, India
Shashit Shetty, MDS Professor and Head, Department of Conservative Dentistry and Endodontics, K.D. Dental College and Hospital, Mathura, Uttar Pradesh, India
References
1. Rafter M. Apexification: a review. Dent Traumatol. 2005;21(1):1-8.
2. Felippe WT, Felippe MC, Rocha MJ. The effect of mineral trioxide aggregate on the apexification and periapical healing of teeth with incomplete root formation. Int Endod J. 2006;39(1):2-9.
3. Torabinejad M, Chivian N. Clinical applications of mineral trioxide aggregate. J Endod. 1999;25(3):197-205.
4. Pradhan DP, Chawla HS, Gauba K, Goyal A. Comparative evaluation of endodontic management of teeth with unformed apices with mineral trioxide aggregate and calcium hydroxide. J Dent Child (Chic). 2006;73(2):79-85.
5. Lemon RR. Nonsurgical repair of perforation defects. Internal matrix concept. Dent Clin North Am. 1992;36(2):439-457.
6. Bargholz C. Perforation repair with mineral trioxide aggregate: a modified matrix concept. Int Endod J. 2005;38(1):59-69.
7. Al-Daafas A, Al-Nazhan S. Histological evaluation of contaminated furcal perforation in dogs’ teeth repaired by MTA with or without internal matrix. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;103(3):e92-e99.
8. Alhadainy HA, Himel VT, Lee WB, El Baghdady YM. Use of a hydroxylapatite-based material and calcium sulfate as artificial floors to repair furcal perforations. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1998;86(6):723-729.
9. Holden DT, Schwartz SA, Kirkpatrick TC, Schindler WG. Clinical outcomes of artificial root-end barriers with mineral trioxide aggregate in teeth with immature apices. J Endod. 2008;34(7):812-817.
10. Matt GD, Thorpe JR, Strother JM, McClanahan SB. Comparative study of white and gray mineral trioxide aggregate (MTA) simulating a one- or two-step apical barrier technique. J Endod. 2004;30(12):876-879.
11. Holland R, Mazuqueli L, Souza V, et al. Influence of the type of vehicle and limit of obturation on apical and periapical tissue response in dogs’ teeth after root canal filling with mineral trioxide aggregate. J Endod. 2007;33(6):693-697.
12. Tay FR, Pashley DH. Monoblocks in root canals: a hypothetical or a tangible goal. J Endod. 2007;33(4):391-398.
13. Bozeman TB, Lemon RR, Eleazer PD. Elemental analysis of crystal precipitate from gray and white MTA. J Endod. 2006;32(5):425-428.
14. De-Deus G, Petruccellli V, Gurgel-Filho E, Coutinho-Filho T. MTA versus Portland cement as repair material for furcal perforations: a laboratory study using a polymicrobial leakage model. Int Endod J. 2006;39(4):293-298.