Localized Idiopathic Apical Root Resorption: A Report of Five Cases With Emphasis on Differential Diagnosis
Robert J. Kanas, DDS; and Scott J. Kanas
Abstract
Seeking to describe the clinical and radiologic features of localized idiopathic apical root resorption (LIAR), the authors conducted a dental literature review of idiopathic external root resorption and performed a retrospective study of LIAR among the identified cases retrieved from a private general practice. Cases involved five patients—one female and four males—ranging in age from 15 to 48 years. The authors concluded that LIAR appears to be a distinct but uncommon clinicopathologic condition that is self-limiting, and therefore warrants no definitive treatment. However, the condition, which can present asymptomatically on routine radiographs or is sometimes associated with clinical symptoms, warrants differentiation from periapical replacement resorption as a result of orthodontics or trauma.
Idiopathic external root resorption of teeth is an infrequent occurrence. The dental literature on this topic consists chiefly of case reports with no large clinical studies completed. The etiology of this condition is unknown. However, factors such as trauma, infection and/or inflammatory sources, occlusal trauma, orthodontic forces, neoplasia and cysts, systemic disorders, and hormonal imbalances have been suggested.1 A diagnosis of idiopathic external root resorption is made after excluding these recognized causes of external root resorption.
Four types of idiopathic external root resorption have been described: localized idiopathic cervical resorption (LICR); multiple idiopathic cervical resorption (MICR); localized idiopathic apical resorption (LIAR); and multiple idiopathic apical resorption (MIAR).2 Both local and multiple idiopathic cervical resorptions are characterized by cavitation of the cervical root area near the cemento-enamel junction (CEJ), exhibiting an irregular radiolucency that initiates in the periodontal ligament (PDL) and may extend to involve the root canal system of the tooth.3,4 Conversely, both local and multiple idiopathic apical resorptions are characterized by periapical replacement resorption (PARR) or shortening of the apical root with replacement of the root by normal-appearing trabeculated bone radiographically.3,4 Local forms are limited to one to three teeth, whereas multiple forms involve more than three teeth.5 Idiopathic cervical resorption has been reported more frequently in females, while idiopathic apical resorption has a male predilection.6,7
Factors implicated in LICR include bacteria from periodontal disease, periodontal treatment, trauma, intra-coronal bleaching, orthodontics, bruxism, developmental tooth defects, as well as an idiopathic derivation.2 The exact pathogenesis is not clearly known. Invasive cervical resorption, extra-canal invasive resorption, cervical root resorption, and cervical inflammatory resorption are additional terms to describe this form of external root resorption. In addition, LICR may be misinterpreted as cervical root caries. Consequently, LICR is more commonly reported.
Conversely, MICR 6,8-20 and MIAR7,21-39 are rarely encountered in clinical practice. There are a limited number of case reports with no large clinical studies in the dental literature of these types of external root resorptions. These conditions are progressive and result in loss of teeth at an early age.6
LIAR is characterized by PARR without ankylosis in one to three teeth.5,40 PARR as a result of orthodontics or trauma occurs more frequently than LIAR. The incidence of PARR that follows orthodontic therapy has been reported to vary between 19% and 31% in all or some of the anterior teeth.40 PARR with ankylosis occurs frequently in teeth traumatized either from avulsion and subsequent reimplantation or intrusive luxation.40-42 Alternatively, LIAR is rarely referenced in the dental literature.31,40,43 Although, radiographically, LIAR appears to be similar to PARR from trauma or orthodontics, there are clinical differences between these entities. LIAR appears to occur in the distal roots of the mandibular first molars or mandibular premolars, sometimes bilaterally, whereas PARR from orthodontics or trauma involves chiefly anterior teeth or maxillary and mandibular premolars.31,40 The authors have documented their experience with five cases of LIAR over the course of 20 years; this article reports the clinical and radiographic features of this form of external root resorption, with an emphasis on the differential diagnosis of this entity.
Materials and Methods
External root resorption cases were retrieved from the private practice files of one of the authors (RJK). Informed consent was obtained from each study participant. Medical history, clinical examination, periodontal probing, and radiographs were reviewed. Radiographs were obtained from the charts, and prior radiographs demonstrating complete root development were sought to use as a baseline for evidence of external root resorption. Since the study was done outside of a hospital, no institutional board review was required.
Radiographs demonstrating PARR in a single tooth or one to three teeth were included. PARR is defined as shortening or blunting of the apical root with replacement of the root with normal-appearing trabeculated bone, as previously described.2 All cervical resorption cases were excluded as well as any multiple-root resorption cases.
Cases of external apical root resorption were excluded if the apical resorption was due to a necrotic or devitalized tooth, periapical inflammatory disease, advanced periodontal disease, history of a systemic disorder associated with apical root resorption, history of trauma, or history of orthodontics. Pulp tests were performed on any suspected tooth by placing a cold ice cube on the offending tooth and recording the responses. In addition to the cold ice pulp tests, teeth were subjected to percussion, palpation, periodontal probe, and mobility tests. An electric pulp tester was not employed. The only positive test results in the study group were for cold ice pulp tests in some of the teeth. A vital pulp test was recorded if the tooth responded to cold, but the cold response faded rapidly when the cold was removed. A non-vital test was recorded if a tooth did not respond to cold. Irreversible pulpitis was recorded if lingering pain persisted after removal of the cold ice.
Results
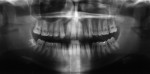
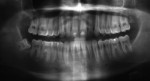
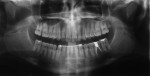
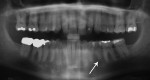
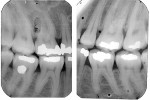
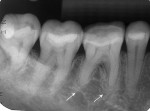
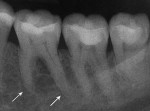
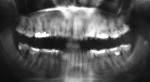
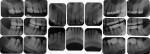
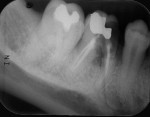
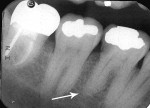
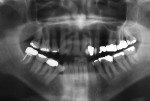
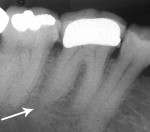
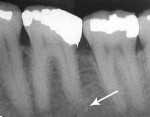
Five cases met the criteria as set forth in the study and are summarized in Table 1; radiographs of the five cases are shown in Figure 1 through Figure 21. Ages ranged from 15 to 48 years. Four patients were male and one female. All five cases involved Caucasian patients. Two cases involved the distal roots of mandibular first molars bilaterally (Figure 1, Figure 2, Figure 3, Figure 4, Figure 5, Figure 6, Figure 7, Figure 8, and Figure 18, Figure 19, Figure 20, and Figure 21); one case involved both roots of the mandibular first molars bilaterally (Figure 11, Figure 12, Figure 13, Figure 14); and two cases involved the mandibular second premolar unilaterally (Figure 9 and Figure 10, and Figure 15, Figure 16, and Figure 17). Two of the patients presented with symptoms similar to cracked-tooth syndrome (Table 1, Case 3 and Case 4), one with “chronic reversible pulpitis” (Table 1, Case 1), and two were asymptomatic (Table 1, Case 2 and Case 5).
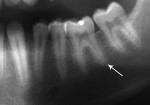
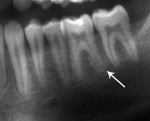
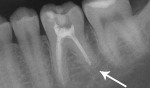
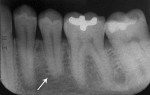
Patients in all five cases exhibited PARR of the involved roots, as previously described (Figure 1 through Figure 21).40 The roots showed shortening and blunting of the apex of greater than 2 mm in all five cases. In three of the five cases, the PARR self-arrested without any treatment. The mandibular left first molar in Case 1 (Figure 5, Figure 6, Figure 7, and Figure 8) received conventional root canal therapy (RCT), while the mandibular right first molar in Case 3 (Figure 11, Figure 12, Figure 13, Figure 14) received a bonded ceramic crown. The PARR appears to have arrested in these two cases, and the outcome of the treatment in relation to the resorption could not be determined. The symptoms the patients were experiencing did eventually resolve. Consequently, all five study cases became symptom-free, including the patient who received conventional RCT (Case 1).
The Case 1 patient, who was the son of the Case 2 patient, was observed in preoperative orthodontic records prior to initiating orthodontic therapy (Figure 1 and Figure 2). The Case 1 patient also demonstrated PARR of the mandibular anterior incisors post-orthodontic therapy (Figure 8). The Case 5 patient had serial extractions completed as a child, but no fixed or removable orthodontic appliances were utilized. Past medical histories were all noncontributory (Table 1). No cases of MICR or MIAR were identified in any of the files reviewed.
Discussion
LIAR is a specific clinicopathologic entity of unknown etiology. It typically involves one to three posterior teeth5—primarily distal roots of mandibular first permanent molars or mandibular premolars, sometimes bilaterally.24,31,40 Males are usually affected more than females over a wide age range.6,7 The involved teeth have been reported to be asymptomatic with an occasional complaint of mobility. PARR is the characteristic radiologic feature of LIAR.40 The apical portion of the root is resorbed and replaced by normal-appearing trabeculated bone. The apical root canal space is patent and there is no ankylosis. No treatment is recommended due to the self-limiting nature of LIAR.
LIAR is similar both radiographically and in clinical location to MIAR. Both conditions exhibit PARR without ankylosis involving the mandibular molars and premolars. However, MIAR involves additional teeth and is progressive, with resultant loss of teeth, whereas LIAR involves one to three teeth and is self-limiting. Clinical history and a full-mouth radiographic series or panoramic radiograph would aid in differentiating these two conditions. Utilization of cone beam computed tomography (CBCT) imaging may be of benefit when available, but its utilization in LIAR cases would require further study.
LIAR has rarely been referenced in the dental literature. The authors identified five cases in a 20-year retrospective analysis. Although five cases represent a limited number for a study, it would appear from the dental literature to be the largest to date. The localized, self-limiting features of this condition, together with the rarity of the disorder, may contribute to the lack of reports in the dental literature. It is the authors’ estimation that this entity occurs in general dental practices in greater number than is represented in the literature and textbooks, but may be overlooked due to its benign nature. In their small general practice consisting of 1,114 active patients, the authors were able to document five cases, reflecting a prevalence rate of 0.5% of their patient population.
The pathogenesis of LIAR is undetermined. These five cases shed very little information in this regard. Possible mechanisms to investigate include occlusal trauma, genetic factors, and inflammatory cytokines. Interleukin-1β polymorphism and RANK/RANKL/OPG (nuclear factor-қ B receptor and ligand and osteoproterin) pathways have been recently implicated as potential molecular models of root resorption.44-47 Osteoclasts and osteoblasts activation and survival in both normal and pathologic conditions, with resultant bone resorption, may be influenced by these pathways. Two of the cases involved a father and son, suggesting a possible genetic link.
The reported age of LIAR presentation for the patients in these five cases was 15 to 48; four of the five patients were between 30 and 48 years of age.
Males have been reported to develop idiopathic apical root resorption more often than females.6,7 Four of the five cases in the study involved males. In both trauma- and orthodontic-related PARR, gender appears not to be a clinical factor.
LIAR primarily occurs in the mandibular first molars and mandibular premolars, sometimes bilaterally. In two cases, LIAR occurred in the second premolars unilaterally while the other three cases involved the mandibular first molars, bilaterally. Cases 1 and 5 involved the distal root of the mandibular first molars bilaterally, while Case 3 involved both mesial and distal roots bilaterally.
LIAR has been reported as an asymptomatic process identified by routine radiographs. However, in three of the five cases in the present study, the teeth were symptomatic, with two cases being asymptomatic and discovered on routine radiographs. Two of the three symptomatic cases presented with cracked-tooth-like symptoms with vital pulp tests. One case presented with “chronic reversible pulpitis,” which the patient had experienced for over a 1-year period. This case was treated with conventional RCT, while one of the cracked-tooth-like syndrome cases was treated with a bonded ceramic crown. No treatment was rendered in three of the five cases.
The symptoms reported in three cases of LIAR may be a transient feature of this condition; however, the relationship between the symptoms and the apical resorption could not be determined. LIAR is considered to be self-limiting and asymptomatic; it usually arrests spontaneously. Definitive dental treatment, such as RCT and crowns, should be discouraged in these cases unless other clinical signs or symptoms warrant such treatment. The RCT was performed in Case 1 due to the length of the pulpitis and young age of the patient. The pulp was determined to be vital at the time of therapy. The ceramic crown was placed on the tooth in Case 3 due to the prolonged cracked-tooth-like symptoms, which had lasted for over 1 year. The teeth in both cases did become asymptomatic post-treatment. The possibility that the symptoms may have self-arrested without treatment must be considered.
The radiologic feature of PARR is characteristic of LIAR. PARR was readily apparent in all five cases (Figure 1 through Figure 21). Two or more millimeters of apical root were resorbed and replaced with normal-appearing trabeculated bone. The PDL space and remnants of the lamina dura appeared to be intact. The apical root canal space appeared patent but somewhat narrowed. The teeth were not ankylosed. Conversely, PARR due to trauma results in ankylosis with bone replacement resorption from both the apical and lateral directions of the root with obliteration of the root canal space and PDL space.1,40-42 PARR from orthodontics would be impossible to distinguish from LIAR radiographically; however, a history of prior orthodontics and the location of the involved teeth would separate this process from LIAR (Table 2).
A differential diagnosis would be limited for this entity due to the benign nature and radiologic features of this condition. Possible diagnoses are listed in Table 3.
The most likely diagnoses to exclude would be PARR from orthodontics or trauma. LIAR occurs in the mandibular first molars and mandibular premolars, whereas PARR from orthodontics occurs in the maxillary and mandibular anteriors and/or premolars. Trauma-associated PARR usually involves anterior teeth and exhibits pulp obliteration and ankylosis. A history of trauma or prior orthodontic therapy would also differentiate these conditions.
Apical root resorption from occlusal trauma and idiopathic osteosclerosis exhibit a similar distribution to LIAR.29,48-52 The mandibular first molars and second premolars are frequent sites for both conditions. The radiographic changes in occlusal trauma, however, consist of dense sclerotic opaque bone at the apex of the tooth, which sometimes masks the apical root area, in contrast to PARR in LIAR.29,48 Idiopathic osteosclerosis infrequently produces apical root resorption when the lesion is in contact with the root apex.53 This condition is usually separated from the roots of teeth and may occur anywhere in the jaws.49-52 In addition, the sclerotic masses of idiopathic osteosclerosis on dental radiographs are dissimilar to the finely trabeculated bone in PARR.
Condensing osteitis and focal cemento-osseous dysplasia frequently occur in the posterior mandible.4 Apical root resorption has been reported in isolated cases of condensing osteitis.54 The teeth usually have irreversible pulpitis or necrotic pulps, and the apical changes consist of widened PDL space with an increased zone of radiodensity at the root apex in contrast to LIAR. Focal cemento-osseous dysplasia is distinguished by a radiolucent border with a radiopaque center and predominates in females.4
Metabolic and systemic disturbances—such as hormonal imbalances,12 Paget’s disease of bone,55,56 hyperparathyroidism,25 Gaucher’s disease,40 hypophosphatasia,57 Papillon-Lefevre syndrome,58 hemifacial atrophy,59 renal and liver diseases,26,60 focal dermal hypoplasia (Goltz syndrome),61 familial expansile osteolysis,62 and osteogenesis imperfecta63—may need to be considered. However, these disease processes produce a radiographic pattern similar to MIAR and MICR but should be considered in LIAR. A full-mouth radiographic series or panoramic film would be indicated in suspected cases. CBCT images may be beneficial if available.
Cementoblastoma and osteoblastoma are neoplastic processes of cementum and bone, respectively. Cementoblastoma is usually fused to the roots of the tooth, and osteoblastoma is separated from the root apex. These lesions arise in the mandibular molar and premolar region before 30 years of age. Pain and swelling are frequent, and the roots of the involved tooth/teeth are obscured or resorbed as a result of the neoplastic process.4
Lastly, short-root anomaly (SRA) may occur sporadically but is considered a developmental disorder.64 The roots are undersized upon eruption as opposed to fully developed roots that resorb apically. Also, SRA occurs in the maxillary incisors and premolars and mandibular second premolars and is more common in females.
Conclusion
LIAR is a rare clinicopathologic entity of unknown etiology. It occurs in the mandibular first molars or mandibular premolars involving one to three teeth. The characteristic radiologic feature is PARR without ankylosis. There is a male preponderance, but no specific age limitation. The disorder may be symptomatic or asymptomatic and is sometimes discovered on routine radiographs. Clinicians should refrain from treating this disorder once diagnosed due to its self-limiting nature. Formulating a differential diagnosis of this entity will aid the clinician in arriving at the proper diagnosis.
Acknowledgment
The first author (RJK) would like to extend gratitude to Drs. Jerald Jensen and Al Abrams for their mentorship, without which this article would not have been possible.
References
1. Bakland LK. Root resorption. Dent Clin North Am. 1992;36(2):491-507.
2. Kanas RJ, Kanas SJ. Dental root resorption: A review of the literature and proposed new classification system. Compend Cont Educ Dent. 2011(32)3. https://www.dentalaegis.com/cced/2011/04/dental-root-resporption-a-review-of-the-literature-and-a-proposed-new-classification. Accessed January 20, 2012.
3. Regezi JA, Sciubba J, Jordan R. Abnormalities of the Teeth. In: Oral Pathology: Clinical-Pathologic Correlations. 6th ed. St. Louis, MO: Saunders Elsevier; 2012:373-389.
4. Neville BW, Damm DD, Allen CM, Bouquot JE. Oral and Maxillofacial Pathology. 3rd ed. St. Louis, MO: Saunders Elsevier; 2009.
5. Stafne EC, Slocumb CH. Idiopathic resorption of teeth. Am J Orthod Oral Surg. 1944;30:41-49.
6. Liang H, Burkes EJ, Frederiksen NL. Multiple idiopathic cervical root resorption: systematic review and report of four cases. Dentomaxillofac Radiol. 2003;32(3):150-155.
7. Cholia SS, Wilson PHR, Makdissi J. Multiple idiopathic external apical root resorption: report of four cases. Dentomaxillofacial Radiol. 2005;34(4):240-246.
8. Mueller E, Rony HR. Laboratory studies of an unusual resorption. J Am Dent Assoc. 1930;17:326-333.
9. Carr HG. Multiple idiopathic resorption of teeth. Br Dent J. 1958;
105:455-456.
10. Kerr DA, Courtney RM, Burkes EJ. Multiple idiopathic root resorption. Oral Surg Oral Med Oral Pathol. 1970;29(4):552-565.
11. Hopkins R, Adams D. Multiple idiopathic resorption of the teeth. Br Dent J. 1979;146(10):309-312.
12. George DI, Miller RL. Idiopathic resorption of teeth: a report of three cases. Am J Orthod. 1986;89(1):13-20.
13. Lydiatt DD, Hollins RR, Peterson G. Multiple idiopathic root resorption: diagnostic considerations. Oral Surg Oral Med Oral Pathol. 1989;67(2):208-210.
14. Moody AB, Speculand B, Smith AJ, Basu MK. Multiple idiopathic external resorption of teeth. Int J Oral Maxillofac Surg. 1990;19(4):200-202.
15. Moody GH, Muir KF. Multiple idiopathic root resorption. J Clin Periodontol. 1991;18(8):577-580.
16. Leache EB, Moreno Gonzalez JP. Spontaneous loss of permanent teeth: A case report. Am J Dent. 1989;2(2):64-66.
17. Beckett HA, Gilmour AG. Multiple idiopathic cervical root resorption in a male. Br Dent J. 1993;175(1):33-34.
18. Kim PH, Heffez HB. Multiple idiopathic resorption in the primary dentition: review of the literature and case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999;88(4):501-505.
19. Iwamatsu-Kobayashi Y, Satoh-Kuriwada S, Yamamoto T, et al. A case of multiple idiopathic external root resorption: a 6-year follow-up study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005;100(6):772-779.
20. Neely AL, Gordon SC. A familial pattern of multiple idiopathic cervical root resorption in a father and son: A 22-year follow-up. J Periodontol. 2007;78(2):367-371.
21. Sachs RL. Progressive idiopathic root resorption. Oral Surg Oral Med Oral Pathol. 1964;17:745-746.
22. Soni NN, La Velle WE. Idiopathic root resorption. Report of a case. Oral Surg Oral Med Oral Pathol. 1970;29(3):387-389.
23. Cowie P, Wright BA. Multiple idiopathic root resorption (case report). J Can Dent Assoc. 1981;47(2):111-112.
24. Brooks JK. Multiple idiopathic apical external root resorption. Gen Dent. 1986;34(5):385-386.
25. Belanger GK, Coke JM. Idiopathic external root resorption of the entire permanent dentition: report of case. ASDC J Dent Child. 1985;52(5):359-363.
26. Pankhurst CL, Eley BM, Moniz C. Multiple idiopathic external root resorption. A case report. Oral Surg Oral Med Oral Pathol. 1988;65(6):754-756.
27. Saravia ME, Meyer ML. Multiple idiopathic root resorption in monozygotic twins: case report. Pediatr Dent. 1989;11(1):76-78.
28. Postlethwaite KR, Hamilton M. Multiple idiopathic external root resorption. Oral Surg Oral Med Oral Pathol Oral Radiol Endod.1989;68(5):640-643.
29. Yusof WZ, Ghazali MN. Multiple external root resorption. J Am Dent Assoc. 1989;118(4):453-455.
30. Counts AL, Widlak RA. Generalized idiopathic external root resorption. J Clin Orthod. 1993;27(9):511-513.
31. Rivera EM, Walton RE. Extensive idiopathic apical root resorption. A case report. Oral Surg Oral Med Oral Pathol. 1994;78(5):673-677.
32. Snelgrove RA. Generalized idiopathic apical root resorption as an incidental finding in an adolescent: a case history. Dent Update. 1995;22(7):276-278.
33. Di Domizio P, Orsini G, Scarano A, Piattelli A. Idiopathic root resorption: report of a case. J Endod. 2000;26(5):299-300.
34. Schätzle M, Tanner SD, Basshardt DD. Progressive, generalized, apical idiopathic root resorption and hypercementosis. J Periodont. 2005;76(11):2002-2011.
35. Moazami F, Karami B. Multiple idiopathic apical root resorption: a case report. Int Endod J. 2007;40(7):573-578.
36. Gupta R. Prakash V. Bilateral extensive idiopathic apical root resorption in supraerupted maxillary molars: a case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;106(3):e44-e47.
37. McMullin A, Fleming PS, DiBiase AT. Idiopathic generalized apical root resorption: a report of three cases. Int J Paediat Dent. 2008;18(4):312-316.
38. Armas JM, Savarrio L, Brocklebank LM. External apical root resorption: two case reports. Int Endod J. 2008;41(11):997-1004.
39. Soğur E, Soğur HD, Baksi BG, Sen BH. Idiopathic root resorption of the entire permanent dentition: systematic review and report of a case. Dent Traumatol. 2008;24(4):490-495.
40. Bender IB, Byers MR, Mori K. Periapical replacement resorption of permanent, vital, endodontically treated incisors after orthodontic movement: report of two cases. J Endod. 1997;23(12):768-773.
41. Fuss Z, Tsesis I, Lin S. Root resorption—diagnosis, classification and treatment choices based on stimulation factors. Dent Traumatol. 2003;19(4):175-182.
42. Ne RF, Witherspoon DE, Gutmann JL. Tooth resorption. Quintessence Int. 1999;30(1):9-25.
43. Weisman MI. Idiopathic tooth resorption. Oral Surg Oral Med Oral Pathol. 1966;21(5):618.
44. Al-Qawasmi RA, Hartsfield JK Jr, Everett ET, et al. Genetic predisposition to external apical root resorption. Am J Orthod Dentofacial Orthop. 2003;123(3):242-252.
45. Al-Qawasmi RA, Hartsfield JK Jr, Everett ET, et al. Genetic predisposition to external apical root resorption in orthodontic patients: linkage of chromosome-18 marker. J Dent Res. 2003;82(5):356-360.
46. Hartsfield JK Jr. Pathways in external apical resorption associated with orthodontia. Orthod Craniofacial Res. 2009;12(3):236-242.
47. Bastos Lages EM, Drummond AF, Pretti H, et al. Association of functional gene polymorphism IL-1β in patients with external root resorption. Am J Orthod Dentofacial Orthop. 2009;136(4):542-546.
48. Rawlinson A. Treatment of root and alveolar bone resorption associated with bruxism. Br Dent J. 1991;170(12):445-447.
49. Geist JR, Katz JO. The frequency and distribution of idiopathic osteosclerosis. Oral Surg Oral Med Oral Pathol. 1990;69(3):388-393.
50. McDonnell D. Dense bone island: A review of 107 patients. Oral Surg Oral Med Oral Pathol. 1993;76(1):124-128.
51. MacDonald-Jankowski DS. Idiopathic osteosclerosis in the jaws of Britons and of the Hong Kong Chinese: radiology and systemic review. Dentomaxillofac Radiol. 1999;28(6):357-363.
52. Williams TP, Brooks SL. A longitudinal study of idiopathic osteosclerosis and condensing osteitis. Dentomaxillofac Radiol. 1998;27(5):275-278.
53. Cheng YS, Kessler HP, Lucente J. Oral and maxillofacial pathology case of the month: Idiopathic osteosclerosis. Tex Dent J. 2008;125(10):1026,1030-1031.
54. Baughman R. Testing your diagnostic skills: Case no. 2: condensing osteitis. Today’s FDA. 2006;18(4):20-21,23.
55. Smith BJ, Eveson JW. Paget’s disease of bone with particular reference to dentistry. J Oral Pathol. 1981;10(4):233-247.
56. Barnett F, Elfenbein L. Paget’s disease of the mandible—a review and report of a case. Endod Dent Traumatol. 1985;1(1):39-42.
57. Newman WG. Possible etiologic factors in external root resorption. Am J Orthod. 1975;67(5):522-539.
58. Rügiger S, Berglundh T. Root resorption and signs of repair in Papillon-Lefèvre syndrome. A case study. Acta Odontol Scand. 1999;57(4):221-224.
59. Fayad S, Steffensen B. Root resorption in a patient with hemifacial atrophy. J Endod. 1994;20(6):299-303.
60. Moskow BS. Periodontal manifestations of hyperoxaluria and oxalosis. J Periodontol. 1989;60(5):271-278.
61. Baxter AM, Shaw MJ, Warren K. Dental and oral lesions in two patients with focal dermal hypoplasia (Goltz syndrome). Br Dent J. 2000;189(10):550-553.
62. Mitchell CA, Kennedy JG, Owens PD. Dental histology in famlilial expansile osteolysis. J Oral Pathol Med. 1990;19(2):65-70.
63. Thoma KH, Sosman MC, Bennett GA. An unusual case of hereditary fibrous osteodysplasia (fragilitas ossium) with replacement of dentin by osteocementum. Am J Orthod Oral Surg. 1943;29:1-30.
64. Apajalahti S, Hölttä P, Turtola L, Pirinen S. Prevalence of short-root anomaly in healthy young adults. Acta Odontol Scand. 2002;60(1):56-59.
About the Authors
Robert J. Kanas, DDS
Associate Professor
Oral and Maxillofacial Pathology
College of Dental Medicine
Roseman University of Health Sciences
South Jordon, Utah
Former Staff Dentist
Crittenton Medical Center
Rochester Hills, Michigan
Former Private Practice
Lakeville, Michigan
Scott J. Kanas
Predoctorate Student
University of Detroit Mercy
School of Dentistry
Detroit, Michigan