Plasma Cell Gingivitis: Clinical and Pathological Case Report
Celine Joyce Cornelius Timothius, BDS, MSc, MS; Ilanit Stern, DDS, MS; J. Kobi Stern, DDS, MS; Rafik A. Abdelsayed, DDS, MS; and Mark E. Peacock, DMD, MS
ABSTRACT
Introduction: Plasma cell gingivitis (PCG) is a rare benign condition usually found on marginal and attached gingiva. This case details a generalized PCG, to include the management of the patient and clinicopathologic characteristics of the disorder. Case Presentation: The patient, a 24-year-old African American female, was referred to the periodontics clinic for severe generalized gingival erythema and edema. Past medical history review was remarkable for sickle cell anemia and systemic lupus erythematosus. The patient was initially prescribed dexamethasone oral rinse (0.5 mg/5 ml) pending biopsy and medical consult for potential causes of any hypersensitivity reaction. The patient also was instructed to discontinue her current type of oral mouthrinse and dentifrice. Biopsy results confirmed a diagnosis of PCG. Resolution of signs and symptoms started 1 month later, and approximately 2 years after initial diagnosis the patient was clinically stable. Conclusion: This report describes the management of a diffuse plasma cell gingivitis and reviews pertinent literature on the lesion. The etiology of PCG, although unclear, may be attributable to a hypersensitivity reaction. PCG may mimic other pathological entities, which underscores the importance of microscopic examination in establishing a definitive diagnosis before treatment initiation.
Plasma cell gingivitis (PCG) is an extremely rare reactive inflammatory condition of the gingiva whose etiology is unclear. Its incidence and prevalence rate is ambiguous as only a few cases have been reported in the literature.1 PCG usually affects the maxillary anterior gingiva2 but can involve any marginal and/or attached gingival tissues and, in some cases, may affect the tongue and other loose oral tissues. The clinical appearance of PCG is often described as presenting with "fire engine red" gingiva with a distinct histological evidence of intense inflammatory cell infiltrates. If it is a generalized PCG, the cause is usually not limited to one source and the plasmacytic response tends to be widespread.3
PCG was first reported by Kerr et al as an "allergic gingivostomatitis," and the cause was attributed to a contact hypersensitivity-type reaction to chewing gum and/or hard candy.4 Other authors have also alluded to possible allergens, such as mint, cinnamon, herbal products, dentifrices, and other spices/flavoring agents, as a cause.5-13 One review even suggested that bacteria in dental plaque could elicit such a reponse.14 Silverman and Lozada proposed that fungal overgrowth may play a part in the etiology.12 There has even been skepticism as to whether PCG should even be considered a separate and distinct clinical condition from plaque-induced gingivitis and periodontitis.15
PCG presents clinically as a well-defined erythematous and edematous lesion affecting keratinized tissues, but not adjacent mucosa. The gingiva, though usually not ulcerated, appears smooth and almost glossy, and may also appear friable and be painful in some patients.16 Gingival tissues may bleed even with light, unintentional stimulation, leading some patients to think it is spontaneous. A recent report documented a PCG case that was originally treated as aggressive plaque-induced periodontal disease.17 Given that the clinical presentation can resemble other neoplastic diseases, including monoclonal plasma cell lesions,18 it is imperative to conduct early examination, establish a complete differential diagnosis, and perform a biopsy with appropriate histologic evaluation.
Therapy for PCG includes treating symptoms while concurrently evaluating the potential etiological agent(s) that may be the cause of the condition. Elimination treatment and dietary analysis may help identify a causative agent. Most patients respond to therapy, although recurrences are not uncommon; the progression of this condition to malignancy has not been reported.19
This case report documents the successful management of a patient with PCG and provides an accompanying review of pertinent literature.
Clinical Presentation
A 24-year-old African American woman was referred to the Periodontics Clinic of the Dental College of Georgia, Augusta University, Augusta, Georgia, in 2020, with a chief complaint that her "gums are bleeding and are very red and tender." Medical history review was significant for sickle cell anemia and systemic lupus erythematosus. Dental history was unremarkable except for an 18-month-long orthodontic treatment that was completed 5 years prior to the current presentation.
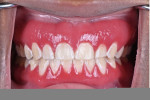
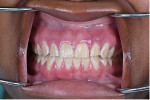
The patient reported daily use of a desensitizing toothpaste along with an antimicrobial mouthrinse (essential oil-based). Oral examination revealed diffuse erythematous and edematous gingival generalized on the facial of maxillary and mandibular keratinized tissues (Figure 1). The tissue was friable, which caused it to bleed easily upon palpation and toothbrushing. Probing depths were generalized 2 mm to 3 mm with generalized bleeding on probing at the affected areas. There was no evidence of attachment loss, mobility, or furcation involvement throughout the dentition.
Case Management
Prior to clinical intervention, a chairside consultation with an oral pathologist elicited a differential diagnosis of PCG, cicatricial pemphigoid, and lichen planus. Cicatricial pemphigoid or mucous membrane pemphigoid was considered, as it typically presents with painful blisters on the oral mucous membrane and as areas of desquamative gingivitis. Similarly, oral lichen planus affects the gingiva, causing areas of redness, swelling, and peeling or blistering. The onset of oral lichen planus also could be triggered by allergy, which was the suspected etiology at the primary evaluation. The histological appearance of lichen planus presents with distinct features such as the characteristic epithelial hyperplasia leading to a saw-toothed form with wedge-shaped hypergranulosis. Cicatricial pemphigoid, on the other hand, is confirmed by histology and direct immunofluorescence. Histological features include subepithelial blisters with mixed infiltration of inflammatory cells.20,21
Under local anesthesia of 2% lidocaine with 1:50,000 epinephrine and 4% articaine with 1:100,000 epinephrine, a 3 mm to 4 mm deep incisional biopsy was obtained from the maxillary left vestibule over the attached gingiva. The patient was prescribed dexamethasone oral rinse (0.5 mg/5 ml) and prophylactically advised to discontinue her mouthrinse, dentifrice brand, and oral contraceptives. She also was referred to her physician for a full blood work-up to identify any potential allergens that may have been causing the condition and had skin-patch tests done based on her physician's recommendation.
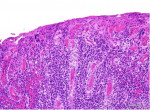
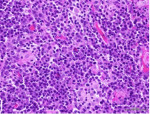
Histologic examination of the specimen from the attached gingiva exhibited erosion of the surface epithelium and supporting intense inflammatory cell infiltrate, predominantly composed of plasma cells (Figure 2). The specimen was covered by fibrinous deposits and supported superficial dilated blood vessels engorged with erythrocytes interspersed by occasional vertical streaks of residual epithelial cells exhibiting uniform basophilic nuclei surrounded by variable amounts of eosinophilic cytoplasm. With an indistinct basal layer, the lamina propria supported an intense inflammatory cell infiltrate, chiefly consisting of densely packed sheets of plasma cells with their characteristic eccentric basophilic nuclei with cartwheel-appearing chromatin surrounded by eosinophilic cytoplasm with paranuclear clear zones (Figure 3). The specimen obtained from the left maxillary gingiva presented with severe chronic mucositis with predominant plasma cell infiltrate, epithelial erosion, and stromal eosinophilia, consistent with PCG. The presence of stromal eosinophils is consistent with allergy-based gingival inflammation.22,23 None of the specimens presented with evidence of neoplasia or granulomatosis. The clinical presentation and histopathological features led the authors to a diagnosis of PCG.
Dexamethasone was the only treatment utilized in this case. No allergens were identified.
Clinical Outcome
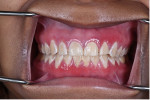
The patient was followed-up at 4 weeks after the initial evaluation appointment and reported improved symptoms since the time of that appointment (Figure 4). Two years after the initial appointment, the patient presented with improved clinical signs and symptoms on the previously affected areas (Figure 5). Gingival inflammation and edema markedly reduced with the appearance of abandoned islands of keratinization. At this final 2-year re-evaluation appointment, the patient presented with remarkably improved clinical findings. Overall gingival appearance was healthy with no apparent edema or periodontal attachment loss. Complete clinical resolution of the PCG was reported with no adverse symptoms.
Discussion
Plasma cell gingivitis is an uncommon inflammatory benign condition characterized by polyclonal proliferation of plasma cells extending into the mucogingival junction. The condition is also referred to as atypical, allergic, or idiopathic gingivostomatitis and plasmacytosis.18
Typically hypothesized to be a hypersensitivity reaction to certain antigens found in chewing gums, toothpastes, and lozenges, the etiology is still unclear.24 Kerr et al in 1971 reported a case of PCG from an allergic reaction to cinnamon, which was added as a flavoring agent in chewing gums. Inflammatory reactions were seen in both attached and free gingiva from flavoring agents added to toothpastes and chewing gums.4 The pathogenesis of the disease could be a result of chronic inflammatory changes leading to local immunological dysregulation, inducing plasma cell migration with pro-inflammatory cytokines. This mechanism triggers B-cell proliferation that ultimately results in the clinical presentation of PCG.25
PCG has a varied clinical presentation but is found as painful, edematous, and erythematous lesions that bleed easily with or without ulcerations.22,26-28 Histological examination usually reports the appearance of dense plasma infiltration into the subepithelial connective tissue and could mimic neoplasms, autoimmune vesicular bullous diseases, and lymphoproliferative disorders. Hence, a differential diagnosis of the condition is crucial to eliminate outliers.18,29,30
Due to the unknown etiology of the condition, therapy is often aimed at providing symptomatic relief and identifying and eliminating triggering factors. First-line therapy includes corticosteroids, immunomodulators, antibiotics, and plaque-control mouthwashes.27,31-33
Conclusion
This case report documented an atypical inflammatory gingival condition: plasma cell gingivitis. The cause of this condition is generally believed to be a hypersensitivity allergic reaction, but the distinct etiology is still unresolved. Detection of antigenic cause may take time and lead to delayed treatment/resolution. Successful management of this condition requires thorough evaluation, treatment, and histological examination of the lesions. In this case, biopsy results confirmed a diagnosis of PCG, and resolution of signs and symptoms started 1 month later. The treatment provided was an oral dexamethasone rinse and elimination of potential allergens. Approximately 2 years after the initial diagnosis the patient was clinically stable.
ABOUT THE AUTHORS
Celine Joyce Cornelius Timothius, BDS, MSc, MS
Assistant Professor, Department of Periodontics, Dental College of Georgia at Augusta University, Augusta, Georgia
Ilanit Stern, DDS, MS
Instructor, Department of Oral Biology and Diagnostic Sciences, Dental College of Georgia at Augusta University, Augusta, Georgia
J. Kobi Stern, DDS, MS
Program Director, Professor, Department of Periodontics, Dental College of Georgia at Augusta University, Augusta, Georgia
Rafik A. Abdelsayed, DDS, MS
Professor, Department of Oral Biology and Diagnostic Sciences, Dental College of Georgia at Augusta University, Augusta, Georgia
Mark E. Peacock, DMD, MS
Professor, Department of Periodontics and Department of Oral Biology and Diagnostic Sciences, Dental College of Georgia at Augusta University, Augusta, Georgia
REFERENCES
1. Janam P, Nayar Br, Mohan R, Suchitra A. Plasma cell gingivitis associated with cheilitis: a diagnostic dilemma! J Indian Soc Periodontol. 2012;16(1):115-119.
2. Holmstrup P, Plemons J, Meyle J. Non-plaque-induced gingival diseases. J Clin Periodontol. 2018;89(suppl 1):S28-S45.
3. Peacock ME, Hokett SD, Hellstein JW, et al. Gingival plasma cell granuloma. J Periodontol. 2001;72(9):1287-1290.
4. Kerr DA, McClatchey KD, Regezi JA. Allergic gingivostomatitis (due to gum chewing). J Periodontol. 1971;42(11):709-712.
5. Kumar V, Tripathi AK, Saimbi CS, Sinha J. Plasma cell gingivitis with severe alveolar bone loss. BMJ Case Rep. 2015;2015:bcr2014207013.
6. Agarwal S, Ajmera N, Chatterjee A, Madaiah H. An unusual plasma cell gingivitis case related to use of herbal toothpowder. Clin Adv Periodontics. 2012;2(2):107-112.
7. Parashis AO, Vardas E, Tosios K. Generalized aggressive periodontitis associated with a plasma cell gingivitis lesion: a case report and non-surgical treatment. Clin Adv Periodontics. 2015;5(2):91-98.
8. Ranganathan AT, Chandran CR, Prabhakar P, et al. Plasma cell gingivitis: treatment with chlorpheniramine maleate. Int J Periodontics Restorative Dent. 2015;35(3):411-413.
9. Isaac-Renton M, Li MK, Parsons LM. Cinnamon spice and everything not nice: many features of intraoral allergy to cinnamic aldehyde. Dermatitis. 2015;26(3):116-121.
10. Joshi C, Shukla P. Plasma cell gingivitis. J Indian Soc Periodontol. 2015;19(2):221-223.
11. Prasanna JS, Mutyap DA, Pantula VR, et al. Plasma cell gingivitis - a conflict of diagnosis. J Clin Diagn Res. 2016;10(11):ZD01-ZD03.
12. Silverman S Jr, Lozada F. An epilogue to plasma-cell gingivostomatitis (allergic gingivostomatitis). Oral Surg Oral Med Oral Pathol. 1977;43(2):211-217.
13. Solomon LW, Wein RO, Rosenwald I, Laver N. Plasma cell mucositis of the oral cavity: report of a case and review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;106(6):853-860.
14. Hedin CA, Karpe B, Larsson A. Plasma-cell gingivitis in children and adults. A clinical and histological description. Swed Dent J. 1994;18(4):117-124.
15. Jadwat Y, Meyerov R, Lemmer J, et al. Plasma cell gingivitis: does it exist? Report of a case and review of the literature. SADJ. 2008;63(7):394-395.
16. Román CC, Yuste CM, González MA, et al. Plasma cell gingivitis. Cutis. 2002;69(1):41-45.
17. Helmy HA, Fadel AF, Mansour KM, et al. Unusual presentation of maxillary plasma cell gingivitis mistakenly treated as aggressive periodontitis. (A case report). Int J Surg Case Rep. 2022;93:106998.
18. Joshi M, Prasanth T, Kosala M, Bhandari SK. Plasma cell gingivitis: a perplexing and rare clinical entity. J Indian Soc Periodontol. 2022;26(4):397-400.
19. Negi BS, Kumar NR, Haris PS, et al. Plasma-cell gingivitis a challenge to the oral physician. Contemp Clin Dent. 2019;10(3):565-570.
20. Mollaoglu N. Oral lichen planus: a review. Br J Oral Maxillofac Surg. 2000;38(4):370-377.
21. Chan LS. Ocular and oral mucous membrane pemphigoid (cicatricial pemphigoid). Clin Dermatol. 2012;30(1):34-37.
22. Palmer RM, Eveson JW. Plasma-cell gingivitis. Oral Surg Oral Med Oral Pathol. 1981;51(2):187-189.
23. Zigante M, Spalj S, Prpic J, et al. Immunohistochemical and histopathological features of persistent gingival enlargement in relation to metal allergic sensitisation during orthodontic treatment. Materials (Basel). 2022;16(1):81.
24. Poswillo D. Plasmacytosis of the gingiva. Br J Oral Surg. 1968;5(3):194-202.
25. Fujimura T, Furudate S, Ishibashi M, et al. Successful treatment of plasmacytosis circumorificialis with topical tacrolimus: two case reports and an immunohistochemical study. Case Rep Dermatol. 2013;5(1):79-83.
26. Lubow RM, Cooley RL, Hartman KS, McDaniel RK. Plasma-cell gingivitis. Report of a case. J Periodontol. 1984;55(4):235-241.
27. Senol M, Ozcan A, Aydin NE, et al. Intertriginous plasmacytosis with plasmoacanthoma: report of a typical case and review of the literature. Int J Dermatol. 2008;47(3):265-268.
28. Smith ME, Crighton AJ, Chisholm DM, Mountain RE. Plasma cell mucositis: a review and case report. J Oral Pathol Med. 1999;28(4):183-186.
29. Dos Santos HT, Cunha JLS, Santana LAM, et al. Plasma cell cheilitis: the diagnosis of a disorder mimicking lip cancer. Autops Case Rep. 2019;9(2):e2018075.
30. Madhavarajan S, Tighe J. Orofacial plasmacytosis: a management conundrum. Br J Oral Maxillofac Surg. 2015;53(4):399-402.
31. Farrier JN, Perkins CS. Plasma cell cheilitis. Br J Oral Maxillofac Surg. 2008;46(8):679-680.
32. Yamaguchi Y, Nishie W, Ito T, Shimizu H. Plasma cell cheilitis successfully treated with topical calcineurin inhibitors. Eur J Dermatol. 2016;26(6):609-610.
33. Rocha N, Mota F, Horta M, et al. Plasma cell cheilitis. J Eur Acad Dermatol Venereol. 2004;18(1):96-98.