Improving Periodontal Disease Management With Artificial Intelligence
Erin A. Kierce, RDH, MS, MPH; and Robert J. Kolts, DDS, MBA
ABSTRACT
Periodontitis is a common inflammatory disease of the adult population, with an incidence positively correlated with age. A lack of standardization relating to periodontitis diagnosis and management, however, can often result in instances of undiagnosed and untreated oral disease. Progressive approaches to care, such as incorporating artificial intelligence (AI) technology into dental practice software, can assist dental providers in standardizing the diagnosis of periodontitis and contribute to increased treatment acceptance by improving patients' health literacy and understanding of their periodontal condition. The use of AI technology can enhance clinical efficiency, improve provider standardization, facilitate clinical decision-making, and promote intra- and interprofessional collaboration. AI-powered radiograph analysis provides objective data to assist dentists in their clinical decision-making and diagnostic consistency.
Periodontitis remains a significant oral health challenge, with high rates of untreated disease exhibited among certain high-risk and disadvantaged groups.1,2 It is a chronic dental disease that impacts an individual's oral health-related quality of life and overall well-being3 and affects an estimated 20% to 50% of the world's population.4-6 Periodontitis is the most common inflammatory disease of the adult population, with an incidence positively correlated with age.7 However, there is a lack of standardization relating to periodontitis diagnosis and management, resulting in instances of undiagnosed and untreated oral disease.8,9 Progressive approaches to care, such as incorporating artificial intelligence (AI) technology into dental practice software, can assist dental providers in standardizing the diagnosis of periodontitis and contribute to increased treatment acceptance by improving patients' health literacy and understanding of their periodontal condition.
Periodontitis is associated with the body's inflammatory response to the presence of oral bacteria and other etiological agents, resulting from inadequate oral hygiene practices and/or environmental factors, causing erythema, inflammation, and destruction of the crestal bone surrounding teeth.5,10-12 Oral inflammation, however, can also contribute to systemic inflammation by releasing endotoxins into the bloodstream, increasing the risk of systemic disease and health complications.11,13-15 Specifically, studies have associated the chronic inflammatory properties of untreated periodontitis with an increased risk of cardiovascular disease, Alzheimer's disease, respiratory disease, cerebrovascular disease, and adverse birth outcomes, as well as an increase in morbidity and mortality relating to diabetes, obesity, pregnancy complications, and osteoporosis.5,13-24
The management of periodontitis involves periodontal therapy, which includes periodontal debridement in conjunction with risk factor and behavioral modifications.25,26 A systematic review and meta-analysis by Luthra et al demonstrated that the treatment of periodontitis is associated with a reduction in the concentration of C-reactive proteins, which are an indicator of systemic inflammation, up to 6 months after treatment, even without the initial use of antibiotics as an adjunctive therapeutic modality. Therefore, the treatment of periodontitis results in a reduction of both local and systemic inflammation.7,13,14
Classifications of Periodontitis
In 2017, the World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions formulated a new classification of periodontitis characterized by a multifaceted staging and grading structure.27 "Staging" categorizes the severity and degree of a patient's disease centered on a quantifiable amount of damaged soft tissue resulting from active periodontitis. Staging also examines factors that may impact treatment and condition management.27 "Grading" aims to predict the rate of disease progression, a patient's likely response to standard therapy, and the disease's potential impact on the patient's systemic health.27 Current categories of periodontitis are "necrotizing periodontal diseases" (associated with papilla necrosis, bleeding, pain, and an impaired immune response), "periodontitis," and "periodontitis as a manifestation of systemic diseases" (ie, genetic disorders, neoplasms, and other metabolic and endocrine disorders).27-29
Current Health Impact of Periodontal Disease in United States
In the United States, approximately 42% of American adults have periodontitis, with 8% exhibiting severe periodontitis.1,2 Further, periodontitis is the causative factor for 80% of tooth loss among people aged 35 years and older.4,30 According to the US Centers for Disease Control and Prevention (CDC), periodontitis is considered to be a worldwide pandemic, causing disability, speech impairment, low self-esteem, and reduced quality of life.5,31
A higher prevalence of periodontitis exists among certain racial and ethnic groups, and higher rates of disease are inversely correlated with an individual's level of income, education, and health literacy.1,32,33 Specifically, the prevalence of total periodontitis, defined as the combination of mild, moderate, and severe periodontitis, is greatest among men (50.2%), Mexican Americans (59.7%), adults below 100% of the federal poverty level (60.4%), current smokers (62.4%), and people with self-reported diabetes (59.9%).1
Clinical Considerations for Periodontitis Diagnosis and Treatment Planning
The diagnosis of periodontitis historically has come from a combination of an analysis of a patient's radiographs, a soft-tissue periodontal assessment, and clinical examination. Dentists and dental hygienists rely on their education, training, and experience to identify disease indicators, such as erythema, inflammation, bone loss, and the presence of etiologic factors such as plaque and calculus, to determine the current state and severity of a patient's condition and generate a plan of treatment. This treatment plan may include nonsurgical or surgical periodontal therapy and adjunctive treatment modalities.
However, some health determinants of high-risk groups, including low health literacy, inadequate access to care, and low-income/socioeconomic status, have resulted in low rates of periodontal treatment and maintenance utilization. It has also been suggested that a lack of patient engagement or time constraints during the appointment contribute to a lack of patient understanding about their state of disease or infection, resulting in an inability to make sound decisions regarding recommended treatment.33,34 One study found that the pressure of time felt by clinicians deleteriously affected their ability to diagnose dental disease radiographically, reducing their sensitivity to recognize caries by 40% and identify crestal bone loss by 67%.34 Further, a current lack of standardization relating to periodontitis diagnosing and management results in inconsistencies among providers.8,9,35 Specifically, measurable clinical parameters, such as probing depth, bleeding on probing, attachment loss, and radiographic bone loss, provide a foundation to determine the current state of a patient's periodontal condition. The American Academy of Periodontology published guidelines that classify the severity and extent of periodontal disease based on factors such as probing depths and vertical bone loss.36 However, it has been concluded that a periodontal diagnosis is dependent on the benchmark reference used by a practitioner and his or her interpretation of the reference.8,9,35,37
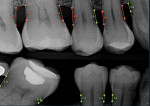
New technology and innovative approaches to care, such as incorporating AI technology into dental practice software, can improve the method and means of patient education, enhance clinical efficiency, improve provider standardization, facilitate clinical decision-making, and promote intra- and interprofessional collaboration.2 Some types of AI-powered radiograph analysis can measure from the cementoenamel junction (CEJ) to the crestal bone, which adds objective data to assist dentists in their clinical decision-making and diagnostic consistency.
The use of AI also can help improve patient engagement, which can facilitate optimal health outcomes and health service utilization.33 Further, improving health literacy via patient education enables patients to better understand their condition, which can lead to an improvement in health behaviors and self-care techniques, potentially reducing future treatment costs.2,38 Using AI technology, dental providers can empower patients by means of presenting unambiguous and visual health information. This empowerment can improve comprehension irrespective of underlying determinants and, ultimately, increase rates of treatment acceptance and utilization (Figure 1).
Artificial Intelligence in Dentistry
Since 2020, the utilization of AI capabilities has increased among all industries. Artificial intelligence refers to a technology that can replicate the intelligence of humans to perform a particular task and evolve based on retrieved information.4 Some types of AI enable algorithms to self-learn and make decisions, resulting in enhanced speed, ability, and accuracy of tasks compared to humans.4 In dentistry, dental clinicians are collaborating with scientists to develop and program algorithms to measure and analyze radiographs, photographs, clinical assessments, and chart notes with dentist-level accuracy and understanding.2,39,40 This can result in a highly accurate output and clinically relevant results, improving patient care, enhancing diagnostics, and reducing errors.39,40
Improving Periodontal Disease Diagnosis and Management with AI
Currently, AI is projected to be useful in disease diagnosis, predicting the prognosis of a specific condition, and developing patient-specific treatment plans.41,42 Further, AI may be able to assist dentists in making time-sensitive clinical decisions, reducing the human element of error, and providing standardized quality of care while reducing provider stress.41 Also, algorithms and AI-enhanced software can help dentists improve patient communication and demonstrate the necessity for treatment. The algorithms are built on millions of data points learned through inputs, resulting in an ability to achieve accuracy comparable to single clinician accuracy.2 According to research, one AI model exhibited periodontal diagnosing accuracy rates of 81% and 76.7% in premolars and molars, respectively,40 while another model has been used to recognize periodontitis by examining a patient's subgingival plaque to differentiate microbial profiles.40,43
Nakano et al used deep learning to detect oral malodor from microbiota and was found to have a predictive accuracy of 97%.44 Danks et al used a deep neural network to measure periodontal bone loss by analyzing periapical radiographs.45 The system achieved a total percentage of correct key points of 89.9%. Similarly, Tonetti et al employed a deep learning model to detect and measure periodontal bone loss from panoramic images, which was then used to stage periodontitis.36 The deep learning model had high accuracy and excellent reliability, suggesting AI may be capable of generating an automatic diagnostic recommendation of periodontal disease and being used as a routine surveillance tool.36,41,46 However, current capabilities of AI only provide partial information required for decision-making or treatment.41
Conclusion
Unmet oral health needs continue to impact systemic health and remain prevalent among the most vulnerable populations.32,47-50 Disparities, such as low health literacy and inadequate access to care, result in higher rates of oral disease and poorer health outcomes and can exacerbate or increase the risk of systemic chronic conditions, including diabetes, cancer, and cardiovascular disease.47-49,51 In addition, a lack of standardization among dental clinicians pertaining to the diagnosis, treatment, and management of periodontitis is associated with inconsistencies relating to care utilization.33,34 Therefore, approaches to care that promote and improve patient engagement, health literacy, and care calibration should be prioritized. Dentistry has long practiced on the standards of delivering comprehensive care through means reflective of emerging technology and scientific evidence. AI can empower patients by standardizing the process of care and improving the accessibility of health information.
ABOUT THE AUTHORS
Erin A. Kierce, RDH, MS, MPH
Chief Compliance/Hygiene Officer, Passion Dental, Marlborough, Massachusetts
Robert J. Kolts, DDS, MBA
Clinical Director, Overjet, Inc., Boston, Massachusetts
REFERENCES
1. Eke PI, Thornton-Evans GO, Wei L, et al. Periodontitis in US adults: National Health and Nutrition Examination Survey 2009-2014. J Am Dent Assoc. 2018;149(7):576-588.e6.
2. Kierce E, Balaban C. The AI revolution: transforming dental hygiene care. Inside Dental Hygiene. 2021;17(5)spec iss 2:16-18.
3. Jaumet L, Hamdi Z, Julia C, et al. Periodontitis assessed with a new screening tool and oral health-related quality of life: cross-sectional findings among general-population adults. Qual Life Res. 2023;32(1):259-272.
4. Shen KL, Huang CL, Lin YC, et al. Effects of artificial intelligence-assisted dental monitoring intervention in patients with periodontitis: a randomized controlled trial. J Clin Periodontol. 2022;49(10):988-998.
5. Bui FQ, Almeida-da-Silva CLC, Huynh B, et al. Association between periodontal pathogens and systemic disease. Biomed J. 2019;42(1):27-35.
6. Nazir MA. Prevalence of periodontal disease, its association with systemic diseases and prevention. Int J Health Sci (Qassim). 2017;11(2):72-80.
7. Luthra S, Orlandi M, Hussain SB, et al. Treatment of periodontitis and C-reactive protein: a systematic review and meta-analysis of randomized clinical trials. J Clin Periodontol. 2023;50(1):45-60.
8. Savage A, Eaton KA, Moles DR, Needleman I. A systematic review of definitions of periodontitis and methods that have been used to identify this disease. J Clin Periodontol. 2009;36(6):458-467.
9. Simpson TC, Needleman I, Wild SH, et al. Treatment of periodontal disease for glycaemic control in people with diabetes. Cochrane Database Syst Rev. 2010;5:CD004714.
10. Cuevas-González MV, Suaste-Olmos F, García-Calderón AG, et al. Expression of microRNAs in periodontal disease: a systematic review. Biomed Res Int. 2021;2021:2069410. doi: 10.1155/2021/2069410.
11. Munz M, Richter GM, Loos BG, et al. Meta-analysis of genome-wide association studies of aggressive and chronic periodontitis identifies two novel risk loci. Eur J Hum Genet. 2019;27(1):102-113.
12. Larjava H, Koivisto L, Hakkinen L, Heino J. Epithelial integrins with special reference to oral epithelia. J Dent Res. 2011;90(12):1367-1376.
13. D'Aiuto F, Gkranias N, Bhowruth D, et al; TASTE Group. Systemic effects of periodontitis treatment in patients with type 2 diabetes: a 12 month, single-centre, investigator-masked, randomised trial. Lancet Diabetes Endocrinol. 2018;6(12):954-965.
14. Machado V, Botelho J, Escalda C, et al. Serum C-reactive protein and periodontitis: a systematic review and meta-analysis. Front Immunol. 2021;12:706432. doi: 10.3389/fimmu.2021.706432.
15. Isola G, Santonocito S, Distefano A, et al. Impact of periodontitis on gingival crevicular fluid miRNAs profiles associated with cardiovascular disease risk. J Periodontal Res. 2023;58(1):165-174.
16. Moliner-Sanchez CA, Iranzo-Cortes JE, Almerich-Silla JM, et al. Effect of per capita income on the relationship between periodontal disease during pregnancy and the risk of preterm birth and low birth weight newborn. Systematic review and meta-analysis. Int J Environ Res Public Health. 2020;17(21):8015.
17. Zardawi F, Gul S, Abdulkareem A, et al. Association between periodontal disease and atherosclerotic cardiovascular diseases: revisited. Front Cardiovasc Med. 2021;7:625579.
18. Ayed MS, Alsharif AF, Divakar DD, et al. Evaluating the possible association between systemic osteoporosis and periodontal disease progression in postmenopausal women. Dis Mon. 2019;65(6):193-215.
19. Nascimento GG, Leite FRM, Vestergaard P, et al. Does diabetes increase the risk of periodontitis? A systematic review and meta-regression analysis of longitudinal prospective studies. Acta Diabetol. 2018;55(7):653-667.
20. Arigbede AO, Babatope BO, Bamidele MK. Periodontitis and systemic diseases: a literature review. J Indian Soc Periodontol. 2012;16(4):487-491.
21. Paquette DW. The periodontal infection-systemic disease link: a review of the truth or myth. J Int Acad Periodontol. 2002;4(3):101-109.
22. Moutsopoulos NM, Madianos PN. Low-grade inflammation in chronic infectious diseases: paradigm of periodontal infections. Ann N Y Acad Sci. 2006;1088:251-264.
23. Fowler EB, Breault LG, Cuenin MF. Periodontal disease and its association with systemic disease. Mil Med. 2001;166(1):85-89.
24. DeBowes LJ. The effects of dental disease on systemic disease. Vet Clin North Am Small Anim Pract. 1998;28(5):1057-1062.
25. Siow DSF, Goh EXJ, Ong MMA, Preshaw PM. Risk factors for tooth loss and progression of periodontitis in patients undergoing periodontal maintenance therapy. J Clin Periodontol. 2023;50(1):61-70.
26. Sanz M, Herrera D, Kebschull M, et al; EFP Workshop Participants and Methodological Consultants. Treatment of stage I-III periodontitis-the EFP S3 level clinical practice guideline. J Clin Periodontol. 2020;47(suppl 22):4-60.
27. Papapanou PN, Sanz M, Buduneli N, et al. Periodontitis: consensus report of workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J Clin Periodontol. 2018;45 suppl 20:S162-S170.
28. Jepsen S, Caton JG, Albandar JM, et al. Periodontal manifestations of systemic diseases and developmental and acquired conditions: consensus report of workgroup 3 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J Clin Periodontol. 2018;45 suppl 20:S219-S229.
29. Albandar JM, Susin C, Hughes FJ. Manifestations of systemic diseases and conditions that affect the periodontal attachment apparatus: case definitions and diagnostic considerations. J Clin Periodontol. 2018;45 suppl 20:S171-S189.
30. Al-Shammari KF, Al-Khabbaz AK, Al-Ansari JM, et al. Risk indicators for tooth loss due to periodontal disease. J Periodontol. 2005;76(11):1910-1918.
31. CDC researchers find close to half of American adults have periodontitis. J Can Dent Assoc. 2012;78:c136.
32. Northridge ME, Kumar A, Kaur R. Disparities in access to oral health care. Annu Rev Public Health. 2020;41:513-535.
33. Bombard Y, Baker GR, Orlando E, et al. Engaging patients to improve quality of care: a systematic review. Implement Sci. 2018;13(1):98.
34. Plessas A, Nasser M, Hanoch Y, et al. Impact of time pressure on dentists' diagnostic performance. J Dent. 2019;82:38-44.
35. Hausmann E, Allen K, Clerehugh V. What alveolar crest level on a bite-wing radiograph represents bone loss? J Periodontol. 1991;62(9):570-572.
36. Tonetti MS, Greenwell H, Kornman KS. Staging and grading of periodontitis: framework and proposal of a new classification and case definition. J Periodontol. 2018;89(suppl 1):S159-S172.
37. Martin JA, Grill AC, Matthews AG, et al. Periodontal diagnosis affected by variation in terminology. J Periodontol. 2013;84(5):606-613.
38. Albano MG, d'Ivernois JF, de Andrade V, Levy G. Patient education in dental medicine: a review of the literature. Eur J Dent Educ. 2019;23(2):110-118.
39. Chen YW, Stanley K, Att W. Artificial intelligence in dentistry: current applications and future perspectives [erratum appears in Quintessence Int. 2020;51(5):430]. Quintessence Int. 2020;51(3):248-257.
40. Lee JH, Kim DH, Jeong SN, Choi SH. Diagnosis and prediction of periodontally compromised teeth using a deep learning-based convolutional neural network algorithm. J Periodontal Implant Sci. 2018;48(2):114-123.
41. Patil S, Albogami S, Hosmani J, et al. Artificial intelligence in the diagnosis of oral diseases: applications and pitfalls. Diagnostics (Basel). 2022;12(5):1029.
42. Shan T, Tay FR, Gu L. Application of artificial intelligence in dentistry. J Dent Res. 2021;100(3):232-244.
43. Feres M, Louzoun Y, Haber S, et al. Support vector machine-based differentiation between aggressive and chronic periodontitis using microbial profiles. Int Dent J. 2018;68(1):39-46.
44. Nakano Y, Suzuki N, Kuwata F. Predicting oral malodour based on the microbiota in saliva samples using a deep learning approach. BMC Oral Health. 2018;18(1):128.
45. Danks RP, Bano S, Orishko A, et al. Automating periodontal bone loss measurement via dental landmark localisation. Int J Comput Assist Radiol Surg. 2021;16(7):1189-1199.
46. Chang HJ, Lee SJ, Yong TH, et al. Deep learning hybrid method to automatically diagnose periodontal bone loss and stage periodontitis. Sci Rep. 2020;10(1):7531.
47. Liu Y, Li Z, Walker MP. Social disparities in dentition status among American adults. Int Dent J. 2014;64(1):52-57.
48. Borrell LN, Beck JD, Heiss G. Socioeconomic disadvantage and periodontal disease: the Dental Atherosclerosis Risk in Communities study. Am J Public Health. 2006;96(2):332-339.
49. Malecki K, Wisk LE, Walsh M, et al. Oral health equity and unmet dental care needs in a population-based sample: findings from the Survey of the Health of Wisconsin. Am J Public Health. 2015;105(suppl 3):S466-S474.
50. Lee PH, McGrath CPJ, Kong AYC, Lam TH. Self-report poor oral health and chronic diseases: the Hong Kong FAMILY project. Community Dent Oral Epidemiol. 2013;41(5):451-458.
51. VanWormer JJ, Acharya A, Greenlee RT, Nieto FJ. Oral hygiene and cardiometabolic disease risk in the Survey of the Health of Wisconsin. Community Dent Oral Epidemiol. 2013;41(4):374-384.