Oral Fibroma Removal With a Er,Cr:YSSG Laser
Kayvon Javid, DDS; Gregori M. Kurtzman, DDS; and Audrey L. Boros, DDS, MSc
Abstract
Fibromas often occur orally and are typically due to trauma to underlying connective tissue resulting in tissue enlargement. To prevent continuing trauma to the tissue, excision may be recommended, and biopsy is often indicated for these masses to confirm their diagnosis and to rule out a potential malignant cause. This article presents a case report in which a Er,Cr:YSSG laser was utilized for excision of an oral fibroma. This approach enabled excellent tissue response compared to the use of a scalpel or monopolar electrosurgery, with minimal patient postoperative discomfort and improved healing.
Fibromas may present in the dental practice either by being casually identified during examination or as a patient primary complaint related to pain due to ongoing trauma to the mass. Fibromas are often found on the interior of the cheek, lips, or lateral borders of the tongue and result from tissue being trapped between the maxillary and mandibular teeth when occluding. These lesions are a benign, scar-like reaction due to persistent long-standing irritation in the mouth resulting in an increase in connective tissue at the site of repeated trauma. They are also known as traumatic fibromas, focal intraoral fibrous hyperplasia, fibrous nodules, or oral polyps. Commonly identified in older individuals, fibromas can occur at any age, affecting 1% to 2% of adults.1
An oral fibroma presents as a firm smooth lump and is usually similar in color to the surrounding soft tissue, although it may be paler due to less vascularity and increased connective tissue within it. A fibroma also may present darker if trauma has led to bleeding inside of it and may have an ulcerated surface related to recent trauma. Fibromas have dome-shaped geometry but may be pedunculated (polyp-like with a short stalk-like base). Apart from the physical obtrusive feel and appearance of an oral fibroma, the only palpable symptom is the patient's primary complaint of pain related to the continuing trauma of inadvertently biting the fibroma.
Intraoral fibromas develop over a period of weeks or months and reach a maximum size usually about 1 cm in diameter; however, they may grow to be larger in size. Typically, they are solitary lesions, and they have not been associated with oral cancer.2 Biopsy should be performed when they are removed to rule out other potential pathologies that occur in the mouth. Studies have reported comparing the incidence of fibroma in differential diagnosis to other oral pathologies as occurring between 5% and 8.4% of the time.3-6
Case Presentation
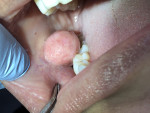
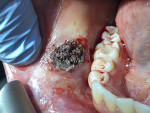
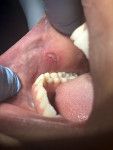
A 32-year-old female patient presented with a primary complaint of a mass on the interior of her right cheek adjacent to the mandibular first and second molars (Figure 1). She indicated the mass had been present for 3 to 4 years and that she periodically would bite it, causing pain and soreness. Her medical history revealed no known medical conditions, and the only medication she was taking was oral birth control.
The patient was informed that the mass was a suspected fibroma that most likely was initiated by trauma to the tissue due to biting the cheek, with damage having occurred to deep layers of tissue. As the mass enlarged it was bitten more frequently and traumatized, and further continued to enlarge. Recommended treatment was excision using an erbium, chromium-doped yttrium, scandium, gallium, and garnet (Er,Cr:YSGG) laser with biopsy and histological confirmation of the suspected fibroma. The patient agreed with the treatment recommendation and treatment was initiated.
A 20% benzocaine topical anesthetic (Benzo-Jel®, Henry Schein Dental, henryschein.com) was applied to the area with a cotton swab around the base of the mass. After allowing several minutes for the anesthetic to affect the soft tissue, one-third of a carpule of 2% lidocaine with 1:100,000 epinephrine (HSI, Henry Schein Dental) was administered on the mesial and distal aspects of the mass at its base.
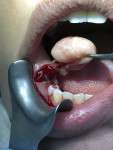
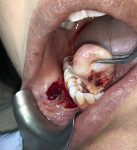
The Er,Cr:YSGG laser (Waterlase Express™, Biolase, biolase.com) unit was placed into Soft Tissue Biopsy mode and then set in Rapid Cut for removal of the lesion. Cutting was performed at 3.00 watts, 75 mill joules, and 40 hertz in a pulsed output. With a MZ6-14mm ZipTip (Biolase) tip on the handpiece (Figure 2) and with tension exerted on the mass with tissue forceps, the laser was used to cut and detach the mass at its base. Working from mesially to distally while continuing to exert pressure on the mass, the clinician moved the tip back and forth across the base as it cut the soft tissue (Figure 3). This continued until complete detachment was accomplished (Figure 4). Upon examination of the cut edges no noticeable thermal changes (burning of the tissue) were present and inflammation was minimal.
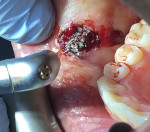
Hemostasis of the cut area was performed by placing the laser unit in the Hemostasis setting while still in Soft Tissue Biopsy mode. In this setting, hemostasis was performed at 1.00 watts, 20 mill joules, and 50 hertz in a pulsed output. Gently tugging on the fibroma either with a locking hemostat, as was done in this case, or via suture placed through the fibroma allows the practitioner to better visualize the base of the fibroma where the cut will be made. The laser tip was held 1 mm to 3 mm from the bleeding area and defocused, being moved in a circular motion over the area starting at the margins and moving to the center (Figure 5). Starting the incision marginally rather than at the center of the fibroma enabled good visualization and helped minimize accidental tissue removal. As hemostasis was achieved with the laser, the bleeding surface contracted and the exudate darkened.
The site was periodically dabbed with sterile gauze to remove any pooled blood on the surface, allowing the practitioner to verify that hemostasis had been achieved. Upon achieving hemostasis, the surface of the site had the appearance of dark coagulated blood with some scattered lighter areas (Figure 6). Hemostasis may be tested by gently pressing a piece of clean sterile gauze on the surface. Should any blood be apparent on the gauze surface, reapplication of the laser in hemostasis mode may be repeated until no bleeding is noted.
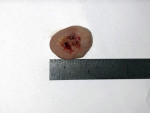
The mass measured almost 3 cm at its longest dimension (Figure 7). The excised tissue was placed into formalin in a sealable container to prepare the tissue for transportation. The contained specimen was sent for histological analysis to confirm the suspected diagnosis of fibroma and rule out a potentially malignant state of the tissue.
Postoperative instructions included a prescription for a chlorhexidine oral rinse (Peridex™, 3M Oral Care, 3m.com) and recommendation to use it twice daily for 30 seconds to 1 minute for the first week postoperatively. Chlorhexidine rinses aid in decreasing plaque and the associated bacteria within it on adjacent dentition, thereby reducing tissue inflammation potential at the surgical site during the initial healing period.7 The patient was not given a prescription for a narcotic pain medication and was advised to use an over-the-counter (OTC) nonsteroidal anti-inflammatory drug (NSAID) such as ibuprofen should she have any discomfort. She was appointed for follow-up 2 weeks post-surgically to assess healing.
Histological Findings
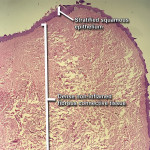
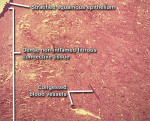
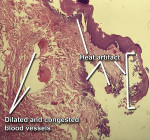
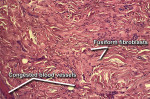
The pathology report noted the following: Clinical examination of the specimen submitted revealed formalin fixed tissue of 2.8 cm x 2 cm x 1.8 cm consisting of one nodule that was tan-gray soft tissue. The specimen was in two blocks due to its size and was serially sectioned and totally submitted. After tissue processing, microscopic examination of the specimen was performed at the histology laboratory. Examination of sections of the two blocks of specimen revealed nodular segments of tissue covered by stratified squamous epithelium. Most of the lesion was composed of collagenous fibrous tissue showing little functional organization. This was noted with low-power magnification of the specimens (Figure 8). Small-diameter blood vessels were present throughout the specimens, as seen under medium-power magnification (Figure 9). Additionally, examination of the specimens at medium-power magnification at the edges noted heat, or thermal, artifacts where the tissue had been cut by the laser (Figure 10). Because the depth of the heat artifacts was nominal, laser excision of the tissue did not hamper histological analysis. Dilated and congested blood vessels also were noted upon examination at medium-power magnification. At higher power, dense fibrous connective tissue with fusiform fibroblasts and numerous vascular channels was noted (Figure 11). Inflammation was not prominent, and no evidence of malignancy was observed in the sections studied. Histological diagnosis, as per the pathologist's findings, confirmed fibroma.
Postoperative Follow-up
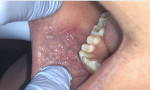
The patient presented for the 2-week postoperative appointment and indicated she had experienced no pain following the surgical excision of the mass. She noted she had not taken any OTC pain relievers either immediately following surgery or in the subsequent 2 weeks. Examination of the surgical site revealed minimal inflammation with slight redness at the cut edges. The area between the cut edges presented with granulation over the surface, indicating healing was progressing normally (Figure 12). At 4 weeks postoperative, re-examination of the surgical site noted no remaining inflammation and the only evidence that excision had occurred was a slight raised line at the site (Figure 13). The patient indicated overall there was no pain or irritation during the healing over the previous 4 weeks.
Discussion
Biopsy and oral fibroma removal traditionally has been performed with a scalpel and excision, resulting in bleeding margins. The raw bleeding edge left by scalpel excision frequently requires suture placement to control bleeding when the area exposed is as large as would be observed with removal of a significant fibroma. Additionally, these types of wounds have been associated with postoperative pain and inflammation.8-10 The use of sutures for wound management may exacerbate site irritation until their resorption or removal.11 Later techniques advocated the use of monopolar electrosurgery as an alternative to the scalpel blade. Electrosurgery, if used improperly, has the potential to cause tissue burns and increased site inflammation that may hamper healing.12,13 This method provides good hemostasis, however there is increased postoperative sensitivity with associated inflammation compared to the use of a scalpel.14,15
With the advent of lasers for soft-tissue surgeries, various types of laser devices have been used successfully for oral fibroma removal. Laser selection is practitioner dependent. Diode lasers may be used for fibroma removal; their utilization of the laser tip physically contacting the tissue being cut provides the practitioner a familiar feel to the use of a scalpel or electrosurgery. An advantage of diode laser application compared to scalpel use is the provision of a relatively bloodless surgical incision with minimal swelling and scarring postoperatively.16 Also, compared to an electrosurgery tip, the use of a diode laser reduces the zone of affected cells considerably, thus less tissue trauma and thermal burning and better healing have been reported.17 Consequently, the diode laser is suitable for excision of oral fibromas.18
Other lasers, including carbon dioxide (CO2), erbium-doped yttrium aluminum garnet (Er:YAG), neodymium-doped yttrium aluminum garnet (Nd:YAG), and Er,Cr:YSGG, have been reported in the literature with regard to use in soft-tissue excisional procedures. Use of an Er,Cr:YSGG laser resulted in less tissue damage than a diode laser,19,20 which, as stated earlier, is considered a good cutting device for oral mucosa. For excision of oral soft-tissue lesions, the CO2 and Er:YAG lasers both yield low postoperative pain. Although more bleeding was reported with the Er:YAG than the CO2 laser, the lower thermal effect of the Er:YAG laser seems advantageous for histopathological evaluation when a biopsy is indicated.21 Regardless of wavelength, all of these types of lasers have showed advantages for soft-tissue excision, including a bloodless surgical field, minimal to no postoperative pain, and ease of execution clinically.22 No studies have reported any clinical advantages when comparing various lasers with regard to soft-tissue surgery, but all of the aforementioned lasers demonstrate a clear advantage to the use of a scalpel.23 Again, laser selection is practitioner dependent.
Clinically, laser usage for dental soft-tissue treatment offers improved tissue response and predictable results. Er:YAG and Er,Cr:YSSG lasers have a narrow zone of affected cells at the cut margins, which permits them to be used for biopsy procurement intraorally. They may not be suitable for small biopsies in which the differential diagnosis required includes malignancies, ulcerations, vesiculobullous conditions, or dysplasia, or when interpretation of margins is requested. This is because lasers produce artifactual changes that complicate and often times entirely prevent definitive interpretation of the affected tissue in small samples, as insufficient biopsy tissue may remain for analysis from the margins. Should the specimen being removed be 10 mm (1 cm) or greater in each dimension then the laser may be well suited for biopsy as sufficient unaltered tissue from the laser excised margins of the sample will not affect histological evaluation. Compared to other surgical methods, lasers offer a lower inflammatory reaction, more pseudomembrane coverage, and minimal damage of adjacent mucoperiosteal tissue.24,25 When comparing tissue reactions between the use of a scalpel and Er,CR:YSGG laser in animal studies, wound healing was enhanced with the laser, with improvement histologically at the wound margins.19,25 Hemostasis management affects the postoperative experience from both a healing and patient comfort standpoint.
Conclusion
Oral fibromas result from trauma to underlying connective tissue, leading to tissue enlargement. Because these growths tend to occur in areas where the trauma is due to teeth clamping down on the tissue (eg, lips, cheek, and lateral border of the tongue), as they enlarge they become further prone to biting and being traumatized, creating further enlargement and continuing the growth cycle. When fibromas are identified, excision should be recommended to prevent continuing trauma to the tissue.
Biopsy is indicated for these masses to confirm their diagnosis and rule out a potential malignant cause. In the case presented, a Er,Cr:YSSG laser was used to excise an oral fibroma, resulting in excellent tissue response, minimal patient postoperative discomfort, and good healing. Other types of lasers may also have been applicable in this case, with the same surgical approach taken irrespective of the laser's wavelength.
Acknowledgment
The photomicrographs (Figure 8 through Figure 11) are the property of Dr. Boros.
About the Authors
Kayvon Javid, DDS
Fellow, International Congress of Oral Implantologists; Private Practice, Lomita, California
Gregori M. Kurtzman, DDS
Master, Academy of General Dentistry; Diplomate, International Congress of Oral Implantologists; Private Practice, Silver Spring, Maryland
Audrey L. Boros, DDS, MSc
Assistant Professor, Ostrow School of Dentistry, University of Southern California, Los Angeles, California; Private Practice, Santa Monica, California
References
1. Esmeili T, Lozada-Nur F, Epstein J. Common benign oral soft tissue masses. Dent Clin North Am. 2005;49(1):223-240.
2. Panta P. Traumatic fibroma. Pan Afr Med J. 2015;21:220. doi:10.11604/pamj.2015.21.220.7498.
3. Khan SU, O'Connor TE, Keogh IJ. A series of oral lesions presenting to an otolaryngology department. Ir Med J. 2015;108(6):177-179.
4. Arslan S, Çobanoğlu B, Ural A, et al. A 15-year retrospective study of 160 cases of benign lip lesions. J Laryngol Otol. 2015;129(12):1224-1227.
5. Valério RA, de Queiroz AM, Romualdo PC, et al. Mucocele and fibroma: treatment and clinical features for differential diagnosis. Braz Dent J. 2013;24(5):537-541.
6. Khan SU, O'Connor TE, Keogh IJ. A series of oral lesions presenting to an otolaryngology department. Ir Med J. 2015;108(6):177-179.
7. Chye RML, Perrotti V, Piattelli A, et al. Effectiveness of different commercial chlorhexidine-based mouthwashes after periodontal and implant surgery: a systematic review. Implant Dent. 2019;28(1):74-85.
8. Saeed S, Ali A, Zainab S, Junaid Khan MT. Scalpel versus diathermy of midline skin incisions: comparison of mean pain scores on second post-operative day. J Pak Med Assoc. 2017;67(10):1502-1505.
9. Giovannacci I, Mergoni G, Meleti M, et al. Postoperative discomfort in oral soft tissue surgery: a comparative perspective evaluation of Nd:YAG Laser, quantic molecular resonance scalpel and cold blade. Minerva Stomatol. 2015;64(1):9-20.
10. Peneva M, Gjorgjeska A, Gjorgova ST, et al. Evaluation of pain following the use of scalpel versus electrosurgery for skin incisions in the facial regions. Pril (Makedon Akad Nauk Umet Odd Med Nauki). 2018;39(2-3):107-112.
11. Kulkarni SS, Chava VK. Comparison of cyanoacrylate and silk sutures on healing of oral wounds-an animal model study. Indian J Dent Res. 2003;14(4):254-258.
12. Huschak G, Steen M, Kaisers UX. Principles and risks of electrosurgery [article in German]. Anasthesiol Intensivmed Notfallmed Schmerzther. 2009;44(1):10-13.
13. Odell RC. Surgical complications specific to monopolar electrosurgical energy: engineering changes that have made electrosurgery safer. J Minim Invasive Gynecol. 2013;20(3):288-298.
14. Ozcelik O, Haytac MC, Akkaya M. Iatrogenic trauma to oral tissues. J Periodontol. 2005;76(10):1793-1797.
15. Chandra RV, Savitharani B, Reddy AA. Comparing the outcomes of incisions made by colorado microdissection needle, electrosurgery tip, and surgical blade during periodontal surgery: a randomized controlled trial. J Indian Soc Periodontol. 2016;20(6):616-622.
16. Azma E, Safavi N. Diode laser application in soft tissue oral surgery. J Lasers Med Sci. 2013;4(4):206-211.
17. Stübinger S, Saldamli B, Jürgens P, et al. Soft tissue surgery with the diode laser--theoretical and clinical aspects [article in French, German]. Schweiz Monatsschr Zahnmed. 2006;116(8):812-820.
18. Bargiela-Pérez P, González-Merchan J, Díaz-Sanchez R, et al. Prospective study of the 532 nm laser (KTP) versus diode laser 980 nm in the resection of hyperplastic lesions of the oral cavity. Med Oral Patol Oral Cir Bucal. 2018;23(1):e78-e85.
19. Jin JY, Lee SH, Yoon HJ. A comparative study of wound healing following incision with a scalpel, diode laser or Er,Cr:YSGG laser in guinea pig oral mucosa: a histological and immunohistochemical analysis. Acta Odontol Scand. 2010;68(4):232-238.
20. Tracey R. Soft-tissue surgery: use of the Er,Cr:YSGG laser. Dent Today. 2008;27(2):156-159.
21. Suter VG, Altermatt HJ, Bornstein MM. A randomized controlled clinical and histopathological trial comparing excisional biopsies of oral fibrous hyperplasias using CO2 and Er:YAG laser. Lasers Med Sci. 2017;32(3):573-581.
22. Romeo U, Palaia G, Tenore G, et al. Excision of oral mucocele by different wavelength lasers. Indian J Dent Res. 2013;24(2):211-215.
23. Monteiro L, Delgado ML, Garcês F, et al. A histological evaluation of the surgical margins from human oral fibrous-epithelial lesions excised with CO2 laser, Diode laser, Er:YAG laser, Nd:YAG laser, electrosurgical scalpel and cold scalpel. Med Oral Patol Oral Cir Bucal. 2019;24(2):e271-e280.
24. Qu W, Shang J, Liu L, et al. Comparative study on the incision healing of the palatal mucosa by using Er:YAG laser or traditional scalpel in the SD rats. Lasers Med Sci. 2018;33(5):1019-1024.
25. Ryu SW, Lee SH, Yoon HJ. A comparative histological and immunohistochemical study of wound healing following incision with a scalpel, CO2 laser or Er,Cr:YSGG laser in the guinea pig oral mucosa. Acta Odontol Scand. 2012;70(6):448-454.