Smear Layer Removal Efficacy Using EndoActivator and EndoUltra Activation Systems: An Ex Vivo SEM Analysis
Priyatam Karade, MDS; Alexander Johnson, BS, MS; John Baeten, BS, MS; Rutuja Chopade, MDS; and Upendra Hoshing, MDS
Abstract
Objective:This study evaluated intracanal smear layer removal using syringe and needle irrigation (ie, conventional irrigation [CI]) with and without adjunctive activation using the EndoActivator® or EndoUltra®. Materials and Methods: Thirty-five premolars were divided into three experimental groups (n = 10) and a control (n = 5): (1) CI, (2) CI plus sonic activation (EndoActivator), (3) CI plus ultrasonic activation (EndoUltra), or (4) CI using saline. All teeth were prepared to a size #40 K file. Following irrigation, the teeth were split and imaged using scanning electron microscopy (SEM). SEM images were evaluated by: (1) blinded researchers who provided a smear layer score (1-5), and (2) a custom software algorithm that automatically and independently quantified the number of open tubules. Results: The use of both the EndoActivator and EndoUltra resulted in significantly cleaner smear layer scores at all canal thirds compared to CI (P < .001). The EndoUltra yielded significantly cleaner scores than the EndoActivator at the apical third (P < .001); however, no significant difference was evident in the middle and coronal thirds. The software analysis yielded the same conclusions as the smear layer scores except statistical significance was seen between the EndoUltra and EndoActivator at all canal thirds (P < .02). Of the adjunctive irrigant activators, the EndoUltra opened 94%, 117%, and 26% more tubules in the apex, middle, and coronal thirds, respectively, than the EndoActivator. Conclusions: The EndoUltra cleaned canals more effectively than the EndoActivator and CI.
The ultimate goal of endodontic treatment is to eradicate pathogenic microbes from the root canal system to prevent reinfection.1 To achieve this objective, root canals are thoroughly disinfected before obturation using mechanical instrumentation, intracanal irrigants and medicaments, and adjunctive activation devices during final irrigation. However, after mechanical instrumentation, an amorphous, irregular layer known as the smear layer is formed on root canal walls.2 The smear layer consists primarily of inorganic dentin material yet also contains organic material such as remnant pulp tissue and viable bacteria.3 Pashley proposed that smear layer containing bacteria might provide a reservoir of irritants that threaten the long-term effectiveness of endodontic therapy.4 Thus, complete removal of the smear layer is required to eliminate residual bacteria and irritants that might compromise therapy effectiveness.3
Root canal disinfection methods can be divided into two broad categories: irrigant delivery and irrigant agitation methods. Both encompass manual and machine-assisted techniques.5 Typical irrigant delivery includes positive pressure irrigation, which uses a syringe and side-vented needle (ie, conventional irrigation [CI]), or negative pressure irrigation using a syringe and disposable attachment (eg, EndoVac, Kerr, kerrdental.com). Machine-assisted positive pressure irrigation (eg, AutoSyringe, Vista Dental Products, vista-dental.com; RinsEndo, Dürr Dental, duerrdental.com) also can be used, can also machine-assisted negative pressure irrigation (EndoVac Pure, Kerr).
Manual irrigant agitation methods typically consist of hand "activation" using a gutta-percha point. Conversely, machine-assisted activation techniques usually involve equipment that applies sonic or ultrasonic energy to canal irrigants. Sonic devices utilize frequencies between 10 Hz to 200 Hz to activate the canal irrigant. Ultrasonic devices use frequencies around 40 kHz to activate the canal irrigant, which generates cavitation and acoustic streaming within the irrigant.6 Newer techniques that are available include use of a plastic rotary file and dental lasers.7,8
Recently, the EndoUltra® device (Vista Dental Products) was introduced with the intention of improving the irrigation stage of endodontic therapy by energizing the irrigant solution with ultrasonic energy. The cordless device oscillates an activator tip at ~40 kHz to create acoustical streaming and cavitation. The system is designed specifically for activation of endodontic irrigants during root canal therapy, as opposed to being adapted for use with ultrasonic hygiene scaler units. Thus, the EndoUltra is aimed at providing enhanced disinfection and cavitation within the root canal space.
The objective of this ex vivo study was to evaluate and compare the effect of two different irrigant activation systems, the EndoActivator® (Dentsply Sirona, dentsplysirona.com) and the EndoUltra, on the removal of smear layer.
Materials and Methods
A total of 35 freshly extracted, noncarious human intact premolars were selected (age group 18 to 25 years). Endodontic access was obtained with a round diamond bur and a #10 K file (Dentsply Maillefer, dentsplymaillefer.com). Working lengths were set by deducting 1 mm from file lengths as they extruded beyond the apical foramina. Crowns were sectioned using a diamond disc (Brasseler, brasselerusa.com) to obtain a standard working length (16 mm) for all samples.
To simulate clinical conditions, apices were sealed with soft modeling wax (Cera Reus S.A., cerareus.com) and subjected to manual root canal instrumentation, using the step-back method. Manual instrumentation was used in the study to mimic a worst-case scenario, as manual instrumentation tends to generate more dentin debris than rotary instrumentation. The root canals were first instrumented manually with K files up to a #40 master apical size; 5 mL of 5.25% sodium hypochlorite (NaOCl) was flushed after each file.
The step-back phase of the apical third began with a #45 K file, followed by five larger K files up to a #70 K. These roots were then randomly divided into three experimental groups of 10 and a negative control group of five.
Conventional Needle Irrigation (CI) Regiment
Each sample was subjected to final irrigation by utilizing three different irrigation/activation methods, using 5 mL of 5.25% NaOCl (Vishal Dental Products), followed by 5 mL of 17% ethylenediaminetetraacetic acid (EDTA; Pulpdent EDTA Solution, Pulpdent, pulpdent.com) followed by 5 mL 5.25% NaOCl and 0.9% normal saline (Amanta Healthcare, amanta.co).9 All irrigants were delivered at 1 mL/min using a 30-gauge monoject endodontic irrigation needle with syringes (Unolok®, Hindustan syringes), and the needle was inserted 3 mm short of the apex. All canals were dried with absorbent paper points (Dentsply Maillefer) after final irrigation and activation.
Group 1: CI
This group consisted of conventional needle irrigation without irrigant activation.
Group 2: CI + EndoActivator
Each irrigant in the CI regiment was activated using the EndoActivator for 60 seconds at 10,000 cycles per minute (167 Hz) using a #15/02 polymer tip placed 1 mm short of the working length in a cyclic axial movement as described in the manufacturer's instructions-for-use.
Group 3: CI + EndoUltra
Each irrigant in the CI regiment was activated using the EndoUltra for 60 seconds at 40 kHz using a #15/02 metal activator tip in an up-and-down motion as described in the manufacturer's instructions-for-use.
Group 4: Negative Control
15mL of 0.9% normal saline was irrigated using the same specifications detailed in the CI regiment, ie, flow rate, use of a 30-gauge irrigation needle, needle placement, etc. Only normal saline was irrigated in this group.
Sample Sectioning
Using diamond discs, deep grooves were made on the buccal and palatal surfaces of the roots without perforating the canal. The roots were then split longitudinally using a chisel. One half of each root was selected for examination with a scanning electron microscope (SEM).
Scanning Electron Microscope Evaluation
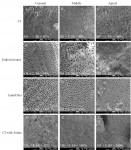
After assembly on coded stubs, the specimens were platinum sputtered and examined using SEM (1000X magnification). The dentinal wall of the coronal, middle, and apical thirds were observed to evaluate the presence/absence of smear layer. Photomicrographs were taken at the coronal, middle, and apical thirds representing overall cleanliness at each canal third. Two independent, calibrated, blinded observers evaluated the SEM images and provided a qualitative smear layer score (1-5) as described by Goel et al,9 where a score of "5" denotes the presence of a thick smear layer, "4" denotes the presence of smear layer but tubules are visible, "3" denotes the presence of a thin smear layer with few tubules open, "2" denotes the presence of a thin smear layer with some tubules open, and "1" denotes all tubules open with no visible smear layer present.
A software macro was written in ImageJ (US National Institutes of Health, nih.gov) to quantify the number of open tubules in the SEM images. The macro executed the following functions in order: (1) despeckle and invert the image, (2) adjust image brightness/contrast to enhance open tubule visualization, (3) threshold to remove background and make the remaining pixels binary, (4) analyze to count the number of open tubules (settings: size > 50 pixels2, circularity 0-1, show - outlines, and summarize). At this point the macro summarized the number of open tubules in a table and a new image. This procedure is summarized in Figure 1.
Sample Size Justification
A sample size of n = 10 per experimental group was calculated using a power analysis (target power = 80%) and a Cohen's d of 1.3, which was assumed because differences between the experimental groups were anticipated to be large and easily discernible.
Statistical Analysis
Inter-observer reliability was verified using the kappa test. Kruskal-Wallis tests and Mann-Whitney U-tests (α = 0.05) were performed to compare the smear layer scores and number of tubules between the groups at each canal location (apical, middle, and coronal).
Results
Smear Layer Score
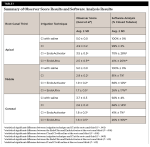
Inter-observer smear layer scores showed good agreement in the coronal third (κ = 0.70) and very good agreement in the middle and apical thirds (κ = 0.83 and κ = 0.88, respectively). The EndoUltra and EndoActivator groups provided statistically significantly better smear layer scores than CI at all canal thirds (P < .001) (Figure 2 and Table 1). The EndoUltra resulted in statistically significantly better smear layer scores at the apical third compared to the EndoActivator (P = .001), whereas the coronal and middle thirds were not statistically different (P = .301 and P = .423, respectively). The negative control (saline irrigation) group resulted in minimal smear layer removal in all canal thirds. Representative SEM images of the experimental and control groups are provided in Figure 3. These results illustrate that activation of irrigants, especially when using the EndoUltra, provides enhanced canal cleanliness and smear layer removal compared to CI.
Tubule Quantification via Software Analysis
The software analysis method provided a quantitative measure of canal cleanliness by counting the number of open tubules in each SEM image. A pilot study verified the software methodology by correctly analyzing images containing known numbers of tubules (not shown). The number of tubules for each image was then normalized to the maximum number of tubules at each canal third, which allowed for the calculation of a "closed tubule percentage" metric for each image using Equation 1:
Equation 1: [closed tubule percentage] = (1-[number of open tubules] / [maximum number of open tubules at that image's root location])*100
The average closed tubule percentage for each experimental group can be seen in Figure 2 and Table 1. Activation using the EndoUltra or EndoActivator resulted in more open tubules than CI alone at all canal thirds (P < .005). The EndoUltra showed statistically significantly more open tubules at all canal thirds compared to the EndoActivator (P ≤ .05). More specifically, the EndoUltra resulted in 94%, 117%, and 26% more open tubules at the apical, middle, and coronal thirds, respectively, than the EndoActivator.
Discussion
This study focused on evaluating the effectiveness of smear layer removal using CI with and without the aid of irrigant activation units (ultrasonic = EndoUltra; sonic = EndoActivator). SEM imaging was performed to obtain representative images at the apical, middle, and coronal canal thirds. SEM images were then analyzed by two blinded observers to provide a smear layer cleanliness score (1-5). Smear layer scores showed that CI plus irrigant activation (either EndoUltra or EndoActivator) was more effective at smear layer removal than CI alone. Moreover, the EndoUltra resulted in statistically significantly cleaner observer scores in the apical third than the EndoActivator.
Images were also subjected to software analysis using a custom-designed macro in ImageJ to quantify the number of closed tubules. Here CI plus irrigant activation resulted in statistically significantly more open tubules than CI alone at all canal thirds, and irrigant activation using the EndoUltra resulted in statistically significantly more open tubules than the EndoActivator at all canal thirds.
Two image analysis techniques were used in this study to compare differences between smear layer score results or a software analysis method. Smear layer scores as described by Goel et al9 were used because the authors implemented a 1-5 scoring scale, which offers more resolution (ie, more categories to segregate the data) than other typically used smear layer scores (1-3). Nevertheless, the software analysis seems to be more advantageous as it increases the dataset's resolution by yielding any numerical result between 0% and 100% (ie, infinite possible results), whereas the smear layer scores are restricted to whole integers 1-5 (ie, only five possible result categories). This is best illustrated in Figure 4, which summarizes the result distribution provided by smear layer scores or the software analysis. Approximately 64% of the smear layer scores were either a 2 or 3 score, while the software scores were more evenly distributed. For these reasons, inter-group statistical analysis is limited for qualitative smear layer score datasets, because categorizing data into whole integer values results in lower dataset resolution. This is likely why statistical significance between the EndoUltra and EndoActivator was not apparent in the middle and coronal thirds for observer scores (P = .431 and P = .301, respectively), yet this significance was discernable via software analysis (P < .05). To avoid observer bias and increase dataset robustness, the described software analysis technique is recommended for future SEM image analysis studies.
The results of this study demonstrate that the EndoUltra produced cleaner canals than the EndoActivator at all canal thirds. This is likely attributed to the EndoUltra's use of ultrasonics, which creates acoustical streaming and cavitation that enhances cleaning efficacy.10-12 Similar to the present results, Sabins et al13 and Caper et al14 reported that passive ultrasonic irrigation produced significantly cleaner canals than passive sonic irrigation. Results from Jiang's study also showed significantly more dentin debris removal when ultrasonic irrigant activation was used compared to sonic irrigant activation.15 However, Rodig et al16 showed significantly greater smear layer removal when the EndoActivator was used rather than ultrasonic irrigant agitation and a canal brush; however, this study did not use the EndoUltra as the ultrasonic unit. Uroz-Torres et al17 used the EndoActivator with a final rinse of 1 mL of 17% EDTA or 4% NaOCl for 1 minute. The EndoActivator did not enhance the removal of smear layer as compared with conventional Max-I-Probe® (Dentsply Maillefer) irrigation with NaOCl and EDTA. This disagrees with the present authors' results as the EndoActivator was found to be more beneficial than CI alone. All aforementioned literature used smear layer scores to create their dataset. As such there is a concern that those statistical analyses may not be robust due to the limitations previously described. Thus, the present authors believe the use of software analysis can greatly aid prior and future SEM studies to elucidate trends that may not be apparent from only evaluating smear layer scores.
The results of this study have several clinical implications. Most importantly, CI does not provide adequate cleaning of the canal system, especially in the apical third. This is concerning because only 45% of endodontists utilize adjunctive ultrasonic or sonic activation,18 with 55% using only CI. The study's results also suggest that the use of ultrasonic irrigant activation, in particular the EndoUltra, removes more smear layer than CI or CI plus the EndoActivator. Specifically, the use of the EndoUltra resulted in two times more open tubules in the apical third, which may help decrease endodontic therapy retreatment/failure rates. Lastly, it was qualitatively observed that the EndoActivator resulted in substantial wall contact during activation, which seemed to dampen its file movement. Conversely, this did not seem to be a concern with the EndoUltra because the file oscillates at ultrasonic frequency and smaller displacements.
Due to normal variations in patient tubule number and density, a limitation of this study is that the software results were normalized to the greatest number of tubules at each root canal third. As a result, the software's quantitative measure of open tubules is likely lower than reality. Although SEM images were obtained to represent the entire root third's cleanliness, another limitation of the study is that only one image at each root third was obtained. It is not expected that these limitations have a profound impact on the study's results and conclusions.
Conclusion
Within the limitations of this study, none of the techniques completely removed smear layer from the apical third. However, the EndoUltra cleaned the canal more efficiently than the other techniques used in this study at all canal thirds. To the authors' knowledge, this is the first study that utilized a software program to automatically quantify the number of open tubules in a SEM image. This method is robust and provides significant advantages over other methods such as observer scores.
Disclosure
The authors disclose that there was no external funding for this study.
Acknowledgments
The authors thank the Department of Sophisticated Analytical Instrument Facility (SAIF), Indian Institute of Technology, Mumbai, India, for assistance with SEM imaging.
About the Authors
Priyatam Karade, MDS
Assistant Professor
Department of Conservative Dentistry and Endodontics
Vasantdada Patil Dental College and Hospital
Kavalapur, Sangli, India
Alexander Johnson, BS, MS
Senior Product Development Engineer
Inter-Med, Inc.
Racine, Wisconsin
John Baeten, BS, MS
Director of Engineering / R&D
Inter-Med, Inc.
Racine, Wisconsin
Rutuja Chopade, MDS
Assistant Professor
Department of Conservative Dentistry and Endodontics
Bharati Vidyapeeth Deemed University Dental College and Hospital
Kavalapur, Sangli, India
Upendra Hoshing, MDS
Professor
Department of Conservative Dentistry and Endodontics
Vasantdada Patil Dental College and Hospital
Kavalapur, Sangli, India
References
1. Haapasalo M, Shen Y, Qian W, Gao Y. Irrigation in endodontics. Dent Clin N Am. 2010;54(2):291-312.
2. Lui JN, Kuah HG, Chen NN. Effect of EDTA with and without surfactants or ultrasonics on removal of smear layer. J Endod. 2007;33(4):472-475.
3. Drake DR, Wiemann AH, Rivera EM, Walton RE. Bacterial retention in canal walls in vitro: effect of smear layer. J Endod. 1994;20(2):78-82.
4. Pashley DH. Smear layer: physiological considerations. Oper Dent Suppl. 1984;3:13-29.
5. de Gregorio C, Estevez R, Cisneros R, et al. Efficacy of different irrigation and activation systems on the penetration of sodium hypochlorite into simulated lateral canals and up to working length: an in vitro study. J Endod. 2010;36(7):1216-1221.
6. de Gregorio C, Estevez R, Cisneros R, et al. Effect of EDTA, sonic, and ultrasonic activation on the penetration of sodium hypochlorite into simulated lateral canals: an in vitro study. J Endod. 2009;35(6):891-895.
7. Bahcall J, Olsen FK. Clinical introduction of a plastic rotary endodontic finishing file. Endo Prac. 2007;10:17-20.
8. Chopra S, Murray PE, Namerow KN. A scanning electron microscopic evaluation of the effectiveness of the F-file versus ultrasonic activation of a K-file to remove smear layer. J Endod. 2008;34(10):1243-1245.
9. Goel S, Tewari S. Smear layer removal with passive ultrasonic irrigation and the Navitip FX: a scanning electron microscopic study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108(3):465-470.
10. Ahmad M, Roy RA, Kamarudin AG. Observations of acoustic streaming fields around an oscillating ultrasonic file. Endod Dent Traumatol. 1992;8(5):189-194.
11. Ahmad M, Pitt Ford TR, Crum LA, Walton AJ. Ultrasonic debridement of root canals: acoustic cavitation and its relevance. J Endod. 1988;14(10):486-493.
12. Ahmad M, Pitt Ford TJ, Crum LA. Ultrasonic debridement of root canals: acoustic streaming and its possible role. J Endod. 1987;13(10):490-499.
13. Sabins RA, Johnson JD, Hellstein JW. A comparison of the cleaning efficacy of short-term sonic and ultrasonic passive irrigation after hand instrumentation in molar root canals. J Endod. 2003;29(10):674-678.
14. Caper D¸ Aydinbelge HA. Effectiveness of various irrigation activation protocols and the self-adjusting file system on smear layer and debris removal. Scanning. 2014;36(6):640-647.
15. Jiang LM, Verhaagen B, Versluis M, van der Sluis LW. Evaluation of a sonic device designed to activate irrigant in the root canal. J Endod. 2010;36(1):143-146.
16. Rödig T, Döllmann S, Konietschke F, et al. Effectiveness of different irrigant agitation techniques on debris and smear layer removal in curved root canals: a scanning electron microscopy study. J Endod. 2010;36(12):1983-1987.
17. Uroz-Torres D, González-Rodríguez MP, Ferrer-Luque CM. Effectiveness of the EndoActivator system in removing the smear layer after root canal instrumentation. J Endod. 2010;36(2):308-311.
18. Dutner J, Mines P, Anderson A. Irrigation trends among American Association of Endodontists members: a web-based survey. J Endod. 2012;38(1):37-40.