Correction of Large Oro-antral Communications From Previously Failed Implant Treatment: Reconstruction of Hard and Soft Tissues
Paul S. Petrungaro, DDS, MS; Santiago Gonzalez, DDS; and Carlos Villegas, DDS
ABSTRACT
As dental implants become more popular for the treatment of partial and total edentulism and treatment of “terminal dentitions,” techniques for the management of the atrophic posterior maxillae continue to evolve. Although dental implants carry a high success rate long term, attention must be given to the growing numbers of revisions or retreatment of cases that have had previous dental implant treatment and/or advanced bone replacement procedures that, due to either poor patient compliance, iatrogenic error, or poor quality of the pre-existing alveolar and/or soft tissues, have led to large osseous defects, possibly with deficient soft-tissue volume. In the posterior maxillae, where the poorest quality of bone in the oral cavity exists, achieving regeneration of the alveolar bone and adequate volume of soft tissue remains a complex procedure. This is made even more difficult when dealing with loss of dental implants previously placed, aggressive bone reduction required in various implant procedures, and/or residual sinus infections precluding proper closure of the oral wound margins. The purpose of this article is to outline a technique for the total closure of large oro-antral communications, with underlying osseous defects greater than 15 mm in width and 30 mm in length, for which multiple previous attempts at closure had failed, to achieve not only the reconstruction of adequate volume and quality of soft tissues in the area of the previous fistula, but also total regeneration of the osseous structures in the area of the large void.
The success of dental implants and bone regeneration procedures ancillary to dental implant placement has been well established in the dental literature.1-4 Dental implants as an option for natural tooth replacement are becoming more commonplace in contemporary dental practices, not only for single tooth replacement but also for partially and/or totally edentulous arches and for patients seeking permanent correction for “terminal dentitions.”5-9 As these types of procedures gain more acceptance in the reconstructive, cosmetic, and surgical disciplines of dentistry, an increasing number of clinicians are incorporating these techniques (basic-intermediate-advanced protocols) into their daily practice in a variety of patient case types. Many of these patients require some degree of bone replacement or bone reconstructive procedure to ensure adequate bone volume around a dental implant, which, in turn, allows for the formation of a stable and healthy cortical plate of bone and viable periosteum.10-12 This requirement for adequate bone contour and volume becomes more difficult to obtain in different areas of the oral cavity, as numerous variables affect wound healing issues and bone remodeling success rates.13,14 This is most notably present in the posterior maxillae, where generally the poorest bone quality, density, and volume is present.15-18
These circumstances led to the development and refinement of sinus elevation and augmentation procedures.19-25 When performed properly, these procedures can provide an adequate volume and quality of bone in the previously pneumatized sinus cavity region, to enable long-term success and stability for implant restorations in the posterior maxillae.22-25
Various procedures were developed to avoid more complex grafting protocols for the reconstruction of the atrophic posterior maxillae exhibiting pneumatized sinus cavities. In particular, the All-on-4® treatment complex allows for an immediate implant prosthesis to be placed on four dental implants inserted at various angles, engaging the cortical plate of the canine eminence and/or nasal floor.26-28 In theory, this approach eliminates the need for sinus grafting procedures and, depending on the prosthetic plan and the required reduction of the maxillary alveolar crest to ensure a stable and adequate restorative space (in general, 18 mm from the planned incisal edge of the central incisors to the crest of the alveolar ridge in the anterior pre-maxillae, and 15 mm in the first molar region26-28), any peri-implant grafting after tooth removal in “terminal dentition” cases. Often times, in the posterior maxillae this causes reduction of bone into the sinus cavity region. If improperly managed, this can create a large defect in the maxillae, with most, if not all, of the base cortical plate of the floor of the sinus being totally absent. When this is not managed with appropriate isolation, reconstruction, and closure procedures, residual oro-antral communications may develop that frequently are of such dimensions that traditional methods of oro-antral management and correction are unsuccessful.29-32
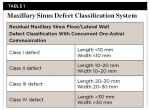
Recurrent sinus infections, patient discomfort, soft-tissue infections, and loss of the distal implant fixture in these types of complications have all been noted.29-32 Table 1 shows a classification system for residual maxillary sinus floor/lateral wall defects with concurrent oro-antral communication resulting from these types of procedures.
Complications of this magnitude in the posterior maxillae can also be seen in cases where previously failed implant treatment causes significant bone loss and sinus infections, failed sinus elevation and grafting procedures, large perforations of the Schneiderian membrane in sinus grafting and elevation procedures where primary wound closure cannot be obtained, and residual oro-antral defects and bone loss in residual tooth extractions where significant amounts of bone destruction is noted apical and lateral to the tooth/teeth to be removed. Additionally, osteomyletis radiation therapy or trauma also can cause the pathological communication.
Previous surgical methods to rectify an oro-antral communication consist of rotation of the buccal fat pad33-35 or treatment of the oro-antral fistula with autogenous bone graft and placement of a resorbable membrane.36-38 These methods present various rates of success in the literature33-39 and are effective for gaining soft-tissue closure of the oro-antral communication. Scattarella et al described a method where a non-absorbable expanded polytetrafluoroethylene (ePTFE) titanium-reinforced membrane was used in conjunction with an autogenous bone graft to repair a small defect following tooth removal.39
The purpose of this article is to outline a new technique, which is a unique approach to the resolution of large oro-antral communications that not only is aimed at repairing the soft-tissue defect but also restoring the posterior maxillary alveolar bone. The management of persistent oro-antral communications is demonstrated, as underlying maxillary alveolar defects in 25 posterior defects were treated. Multiple surgical procedures had been attempted on all of the defects in an effort to resolve the communication, and the procedures all resulted in worsening of the symptoms, including additional scarring and stricture of the wound margins, which made closure and wound margin approximation more difficult. Table 2 lists the maxillary residual defects in the 25 sinus regions treated.
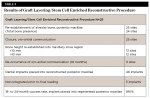
The patients in this treatment group consisted of those who had previously failed implant treatment in the maxillary arch and who had the All-on-4 treatment protocol done, with significant bone reduction required and performed. Each patient had at least one attempt at closure of the oro-antral defect prior to having the graft layering/stem cell enriched reconstruction procedure performed. Eight patients required the placement of additional implants to aid in proper distribution for anterior-posterior (A-P) spread to enable the required distal occlusion in the maxillary prosthesis. The remaining patients, consisting of nine of the treated oro-antral complication sites, were treated for correction of the oro-antral communication only. Table 3 outlines the results of the graft layering/stem cell enriched reconstruction procedure.
Case 1
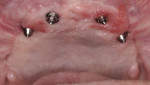
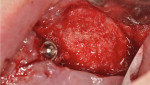
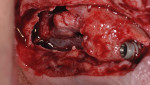
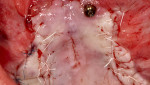
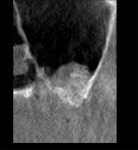
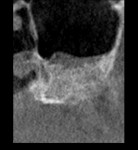
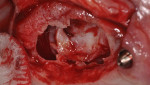
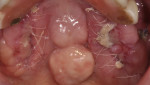
A 60-year-old, non-smoking man presented with persistent oro-antral communications bilaterally after completion of treatment with the All-on-4 concept (Figure 1). The oral antral communications were attempted bilaterally two separate times, once by rotation of the buccal flap, palatally, and once by resorbable membrane placement with buccal flap advancement. Both procedures failed to correct the communication. The genesis of the oro-antral communication was the initial All-on-4 procedure. The required reduction of the alveolar crest was carried past the floor of the sinuses bilaterally, and there was a failure to address the large antral opening with manipulation of the sinus by crestal elevation and insufficient wound closure techniques, occurring at the initial surgical procedure (Figure 2 and Figure 3).
After consultation with the patient and review of a multi-stage correction procedure to both address the oro-antral communication and rebuild the alveolar ridge and floor of the sinus, the patient agreed to the correction procedure for final resolution of the defect present.
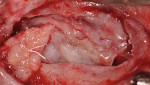
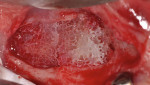
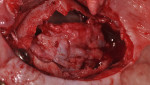
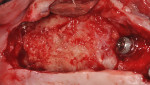
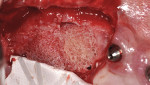
After administration of appropriate sedation and local anesthetic, a palatal incision was made from the tuberosity region, mid-palatal, to the distal aspect of the pre-existing multi-abutment with anterior vertical release into the vestibular space bilaterally. Full-thickness flap elevation was started at the most palatal aspect of the incision, with dissection of the palatal connective tissue circumferentially around the oral antral fistula tissue, incorporating the dissected tissues and fistula into a pedical tissue that was elevated superiorly to become the new superior aspect of the sinus, thereby raising the oro-antral communication into the sinus. Figure 4 shows the right dissection and elevation of the aforementioned procedure with ligation of the greater palatine contents. Figure 4 also demonstrates the large crestal defect in the right posterior maxillae. After elevation of the dissected tissue, a resorbable collagen membrane, 30 mm x 40 mm, (OraMem®, Salvin Dental, salvin.com) was trimmed and inserted superiorly into the sinus. This was then followed by placement of a 1-cc Allostem® stem cell bone growth substitute (Allosource, allosource.com), cut in half. This cancellous bone graft carries stem cells and will help to stimulate bone formation in the non-existence of stimulating autogenous bone cells (Figure 5). Additionally, its “cube”-like structure allows for additional support in maintaining the dissected oro-antral tissue superior in the sinus cavity.
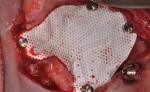
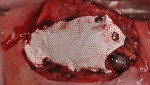
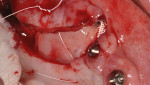
A cortico-cancellous allogenic graft complex was then placed inferior to the Allostem stem cell bone growth substitute to give additional support of the elevated tissues and for the stimulation and structure of the posterior maxillary crest. A large posterior ePTFE titanium-reinforced membrane (Osteogenics Biomedical, osteogenics.com) was then sized to cover the defect margins and molded to form the contour of the newly planned posterior maxillae (Figure 6). Several membrane tacks were used to fix the ePTFE membrane in place into the surrounding palatal and buccal bone (Figure 6). An amniotic/chorionic membrane (AmnioGenix)was then placed over the ePTFE membrane to allow for localized delivery of growth factors to the re-approximated margins of the wound prior to closure. Closure was then accomplished with 4.0 ePTFE sutures by horizontal mattress and continuous sling suture methods.
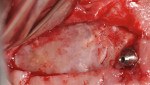
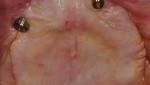
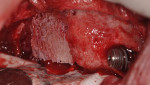
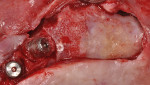
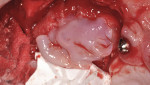
The patient was then observed over a 5-month healing phase in which total alleviation of the communication between the oral cavity and sinus nasal structures was observed. After this healing phase, the membrane tacks and titanium-reinforced ePTFE membrane were removed. The regenerated posterior maxillae can be observed in Figure 7. Figure 8 shows the pre-corrected oro-antral defect and alveolar defect on the left side. Figure 9 shows the 5-month regenerated posterior maxillae.
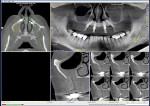
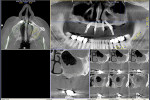
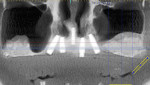
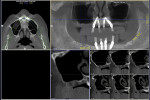
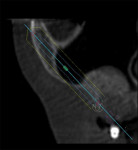
CT scans 1.5 years post-correction are shown in Figure 10, right posterior maxillae, and Figure 11, left posterior maxillae. Note the total reconstruction of the posterior maxillae alveolar bone and closure of the communication.
Case 2
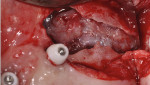
A 71-year-old, non-smoking man presented post All-on-4 treatment in the maxillary arch with bilateral, oro-antral communication in the posterior maxillae (Figure 12). Three previous attempts of closure of the bilateral defects failed, including resorbable collagen membrane placement, rotation of the buccal vestibular tissues, and buccal flap advancement. After the procedure to rectify the oro-antral defect and regenerate the posterior maxillary alveolar bone as previously described, the right maxillary defect was visible, as shown in Figure 13, while Figure 14 shows the left maxillary defect in the posterior maxillae, both crestally and buccally.
Placement of the 1-cc Allostem stem cell bone growth substitute into the right and left sinus defects (Figure 15) preceded the placement of the allogenic, cortico-cancellous graft complex inferior to the stem cell bone growth substitute. Posterior, extra-large ePTFE titanium-reinforced membranes were then seated, covering the margins of the defects and secured with membrane fixation tacks (Figure 16). Prior to closure, amnion/chorionic membranes were placed over the ePTFE membranes, and closure with 4.0 ePTFE sutures was accomplished (Figure 17).
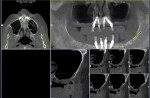
After a 5-month healing and maturation phase, the ePTFE membranes were once again removed. Figure 18 shows a 5-month post-correction CT scan panoramic view, with not only closure of the oro-antral osseous and tissue communication, but also a significant amount of bone regenerated in the graft regions. Figure 19 shows the 5-month re-entry of the right posterior maxillae, and Figure 20 depicts the left posterior maxillae. Figure 13 and Figure 19 can be compared to ascertain in the elevated right posterior the amount of osseous structures regenerated. The patient then went on to have an anterior implant removed (with exposed multiple threads) and a conventional implant prosthesis constructed for the maxillary final implant restoration.
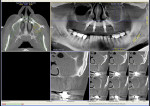
Figure 21 and Figure 22 show the 2-year post-correction CT scan serial views of the right and left sinuses, respectively. Note the regeneration of the posterior maxillary obtained from the procedure outlined.
Case 3
A 66-year-old non-smoking woman presented for an initial All-on-4 treatment concept procedure in both arches. The initial surgical procedure consisted of aggressive alveolar reduction of the maxilla through both maxillary sinuses and placement of four implants with poor A-P spread and low initial torque. Upon initial healing, the patient noticed communication between the oral cavity and the nasal cavity on the right side (Figure 23). After initial healing had occurred, soft-tissue closure of the right defect was noted; however, the prosthodontist requested additional posterior implant support bilaterally, with the placement of implants to support the poor A-P spread and the initial implants placed (Figure 23 and Figure 24). Examination of the CT scans demonstrated no alveolar structures at the base of both the right and left sinuses, complicating the sinus grafting procedures. The initial management decision was to complete bilateral sinus grafting procedures in an attempt to add osseous structures to the sinus cavity for additional implant placement.
Following the procedure outlined in the aforementioned text, a full-thickness flap was elevated, with dissection of the connective tissue from the soft-tissue closure of the antral communication elevated into the sinus cavity (Figure 25). Note the large defect in the crest of the right maxillae, complicating the addition of bone into the sinus region. Figure 26 depicts the Allostem stem cell bone growth substitute cube graft placed into the defect in the right maxillary sinus cavity. Prior to placement of the titanium-reinforced ePTFE membrane, an umbilical membrane (UC Material, Neox® Cord, Amniox Medical, amnioxmedical.com) was placed over the stem cell cube/graft complex (Figure 27). Then the ePTFE membrane was secured with membrane tacks prior to closure (Figure 28) to aid in rapid soft-tissue wound healing to promote soft-tissue dehiscence over the ePTFE membrane, affecting the success of the graft procedure. Closure was then completed by a combination of continuous sling and horizontal mattress suturing techniques with 4.0 ePTFE suture material.
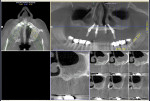
The 10-day postoperative clinical view can be seen in Figure 29. The patient underwent an uneventful healing phase for 5 months, followed by removal of the ePTFE membranes bilaterally. The patient was then allowed to have the sinus regions undergo additional healing and maturation. Figure 30 shows the CT scan serial view 1-year post-corrective procedure of the right sinus region, with the area now treatment-planned in NobelClinician® (Nobel Biocare, nobelbiocare.com) for single zygoma implants posterior bilaterally. Note the alveolar structures once again regenerated in an area that was totally void of a cortical plate of bone pretreatment.
Conclusion
Oro-antral communications that occur post-tooth extraction or as a result of failed implant procedures or other surgical procedures in the maxillary sinus regions are common postoperative complications. Oftentimes smaller communications may close on their own, or a minor buccal flap advancement procedure may be used for the closure of the communication only. However, larger defects, resulting from aggressive alveolar reduction in surgical procedures, failed previous treatment by dental implants and/or sinus grafting, or other pathologic-type lesions may require the restoration of a cortical plate at the apical aspect of the sinus region and regeneration of alveolar structures superior to this crest of bone for the retreatment of the area with dental implants or for permanent closure of recurrent oro-antral defects.
The procedure outlined in this article has been shown to provide total closure of the oro-antral communication in more than 25 oro-antral fistulas with large underlying osseous defects present. It has been shown to be a predictable method in which the regeneration of the posterior maxillary alveolar structure can be achieved and can potentially allow additional implants to again be placed into these areas.
ABOUT THE AUTHORS
Paul S. Petrungaro, DDS, MS
Private Practice, Periodontics and Implantology, Chicago, Illinois
Santiago Gonzalez, DDS
Clinical Professor, Autónoma University School of Dentistry, Manizales, Colombia; Private Practice, Oral Surgery, Manizales, Colombia
Carlos Villegas, DDS
Private Practice, Oral Surgery, Medellin, Colombia
REFERENCES
1. Buser D, Ingimarsson S, Dula K, et al. Long-term stability of osseointegrated implants in augmented bone: A 5-year prospective study in partially edentulous patients. Int J Periodontics Restorative Dent. 2002;22(2)109-117.
2. Simion M, Jovanovic SA, Tinti C, Benfenati SP. Long-term evaluation of osseointegrated implants inserted at the time or after vertical ridge augmentation. A retrospective study on 123 implants with 1-5 year follow-up. Clin Oral Implants Res. 2001;12(1)35-45.
3. Adell R, Lekholm U, Rockler B, Brånemark PI. A 15-year study of osseointegrated implants in the treatment of the edentulous jaw. Int J Oral Surg. 1981;10(6):387-416.
4. Adell R, Eriksson B, Lekholm U, et al. Long-term follow-up study of osseointegrated implants in the treatment of totally edentulous jaws. Int J Oral Maxillofac Implants. 1990;5(4)347-359.
5. Wolfinger GJ, Balshi TJ, Rangert B. Immediate functional loading of Branemark System implants in edentulous mandibles: clinical report of the results of developmental and simplified protocols. Int J Oral Maxillofac Implants. 2003;18(2):250-257.
6. Malo P, Rangert B, Nobre M. All-on-4 immediate-function concept with Branemark System implants for completely edentulous maxillae: a 1-year restrospective clinical study. Clin Implant Dent Relat Res. 2005;7(suppl 1):S88-S94.
7. Rocci A, Martignoni M, Gottlow J. Immediate loading in the maxilla using flapless surgery, implants placed in predetermined positions, and prefabricated provisional restorations: a restrospective 3-year clinical study. Clin Implant Dent Relat Res. 2003;5(suppl 1)29-36.
8. Chee W, Jivraj S. Efficiency of immediately loaded mandibular full-arch implant restorations. Clin Implant Dent Relat Res. 2003;5(1):52-56.
9. Cooper LF, Rahman A, Moriarty J, et al. Immediate mandibular rehabilitation with endosseous implants: simultaneous extraction, implant placement, and loading. Int J Oral Maxillofac Implants. 2002;17(4):517-525.
10. Petrungaro PS. An update on implant placement and provisionalization in extraction, edentulous and sinus grafted sites: A clinical report on 3,200 sites over 8 years. Compend Contin Educ Dent. 2008;29(5):288-300.
11. De Rouck T, Collys K, Wyn I, Cosyn J. Instant provisionalization of immediate single-tooth implants is essential to optimize esthetic treatment outcome. Clin Oral Implants Res. 2009;20(6):566-570.
12. Tarnow DP, Chu SJ, Salama MA, et al. Flapless postextraction socket implant placement in the esthetic zone: Part 1. The effect of bone grafting and/or provisional restoration on facial-palatal ridge dimension change-a retrospective cohort study. Int J Periodontics Restorative Dent. 2014;34(3):323-331.
13. Mattsson T, Kondell PA, Gynther GW, et al. Implant treatment without bone grafting in severely resorbed edentulous maxillae. J Oral Maxillofac Surg. 1999;57(3):281-287.
14. Aparicio C, Perales P, Rangert B. Tilted implants as an alternative to maxillary sinus grafting: a clinical, radiologic, and periotest study. Clin Implant Dent Relat Res. 2001;3(1):39-49.
15. Kuabara MR, Ferreira EJ, Gulinelli JL, Panzarini SR. Use of 4 immediately loaded zygomatic fixtures for retreatment of atrophic edentulous maxilla after complications of maxillary reconstruction. J Craniofac Surg. 2010;21(3):803-805.
16. Bedrossian E, Rangert B, Stumpel L, Indresano T. Immediate function with the zygomatic implant: a graftless solution for the patient with mild to advanced atrophy of the maxilla. Int J Oral Maxillofac Implants. 2006;21(6):937-942.
17. Cannizzaro G, Felice P, Minciarelli AF, et al. Early implant loading in the atrophic posterior maxilla: 1 stage lateral versus crestal sinus lift and 8 mm hydroxyapatite-coated implants: A 5-year randomised controlled trial. Eur J Oral Implantol. 2013;6(1):13-25.
18. Esposito M, Grusovin MG, Felice P, et al. The efficacy of horizontal and vertical bone augmentation procedures for dental implants – a Cochrane systemic review. Eur J Oral Implantol. 2009;2(3):167-184.
19. Tatum H Jr. Maxillary and sinus implant reconstruction. Dent Clin North Am. 1986;30(2):207-229.
20. Misch CE. Maxillary sinus augmentation for endosteal implants: organized alternative treatment plans. Int J Oral Implantol. 1987;4(2):49-58.
21. Hirsch JM, Ericsson J. Maxillary sinus augmentation using mandibular bone grafts and simultaneous installation of implants: a surgical technique. Clin Oral Implants Res. 1991;2(2):91-96.
22. Marx RE, Carlson ER, Eichstaedt RM, et al. Platelet-rich plasma: growth factor enhancement for bone grafts. Oral Surg Oral Med Oral Pathol. 1998;85(6):638-646.
23. Wallace SS, Froum SJ, Tarnow DP. Histologic evaluation of sinus elevation procedure: a clinical report. Int J Periodontics Restorative Dent. 1996;16(1):47-51.
24. Misch CM, Misch CE, Resnik R et al. Reconstruction of maxillary alveolar defects with mandibular symphysis grafts for dental implants: a preliminary procedural report. Int J Oral Maxillofac Implants. 1992;7(3):360-366.
25. Ad-Hoc Committee on Sinus Graft Consensus. Academy of Osseointegration: Sinus Graft Consensus Conference. November 16-17, 1996. Wellesley, MA. Int J Oral Maxillofac Implants. 1998;13(suppl):5-46.
26. Balshi SF, Wolfinger GJ, Balshi TJ. A prospective study of immediate functional loading, following Teeth in a Day protocol: a case series of 55 consecutive edentulous maxillas. Clin Implant Dent Relat Res. 2005;7(1):24-31.
27. Malo P, Nobre Mde A, Petersson U, Wigren S. A pilot study of complete edentulous rehabilitation with immediate function using a new implant design: case series. Clin Implant Dent Relat Res. 2006;8(4):223-232.
28. Malo P, de Araujo Nobre M, Lopes A, et al. A longitudinal study of the survival of All-on-4 implants in the mandible with up to 10 years of follow-up. J Am Dent Assoc. 2011;142(3):310-320.
29. Sandhya G, Bal Reddy P, Jeevan Kumar KA, et al. Surgical management of oro-antral communications using resorbable GTR membranes and FDMB sandwich technique: a clinical study. J Maxillofac Oral Surg. 2013;12(3):254-259.
30. Guven O. A clinical study on oroantral fistulae. J Craniomaxillofac Surg. 1998;26(4):267-271.
31. Del Junco R, Rappaport I, Allison GR. Persistent oral antral fistulas. Arch Otolaryngol Head Neck Surg. 1988;114(11):1315-1316.
32. Punwutikorn J, Waikakul A, Pairuchvej V. Clinically significant oro-antral communications – a study of incidence and site. Int J Oral Maxillofac Surg. 1994;23(1):19-21.
33. Yilmaz T, Suslu AE, Fursel B. Treatment of oroantral fistula: experience with 27 cases. Am J Otolaryngol. 2003;24(4):221-223.
34. Baumann A, Ewers R. Application of the buccal fat pad in oral reconstruction. J Oral Maxillofac Surg. 2000;58(4):389-392.
35. Kim YK. The use of pedicled buccal fat pad graft for bone coverage in primary palatorrhaphy: a case report. J Oral Maxillofac Surg. 2001;59(12):1499-1501.
36. Lee BK. One-stage operation of large oroantral fistula closure, sinus lifting, and autogenous bone grafting for dental implant installation. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;105(6):707-713.
37. Haas R, Watzak G, Baron M, et al. A preliminary study of monocortical bone grafts for oroantral fistula closure. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003;96(3):263-266.
38. Gacic B, Todorovic L, Kokovic V, et al. The closure of oroantral communications with resorbable PLGA-coated beta-TCP root analogs, hemostatic gauze, or buccal flaps: a prospective study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;68(1):229-230.
39. Scattarella A, Ballini A, Grassi FR, et al. Treatment of oroantral fistula with autologous bone graft and application of a non-resorbable membrane. Int J Med Sci. 2010;7(5):267-271.