An Unusual Case of Gingival Enlargement:Peripheral Odontogenic Fibroma
Rania Livada, DDS, MS; Norman Fine, DMD, MS; K. Mark Anderson, DDS, MS; and Jacob Shiloah, DMD
Abstract:
The peripheral odontogenic fibroma (POdF) is an uncommon gingival enlargement. It is a benign, unencapsulated exophytic gingival mass, which is composed of fibrous connective tissue associated with various amounts of calcifications and islands of odontogenic epithelium.The lesion is usually firm to palpation and non-tender, and it could be mistaken for other more common exophytic gingival lesions, such as peripheral ossifying fibroma, pyogenic granuloma, or peripheral giant cell granuloma. Therefore, a histopathologic examination is necessary for accurate diagnosis. This article presents a well-documented case of POdF and a review of the literature in regards to its diagnostic features, treatment modalities, and recurrence.
The odontogenic fibroma is defined by the World Health Organization (WHO) as a benign odontogenic neoplasm of fibroblastic origin. It is characterized by relatively mature collagenous fibrous tissue and varying amounts of odontogenic epithelium. It may occur in either a central or extraosseous location. The extraosseous counterpart is designated peripheral odontogenic fibroma (POdF). Though peripheral odontogenic tumors are rare—comprising only 0.05% of all biopsy specimens—POdF is the most common peripheral odontogenic tumor,1 even more common than its central counterpart by a ratio of 1.4:1.
Gardner2 was the first to define POdF as a clinical entity. It was initially thought to be a rare extraosseous counterpart of the central odontogenic fibroma.3 In contrast to the reactive nature of peripheral ossifying fibroma (POF), this entity was considered a true fibroblastic neoplasm of odontogenic origin.4 The term peripheral odontogenic fibroma WHO type was suggested to distinguish between POdF and POF, which were once used interchangeably, especially in North America.
In contrast to the peripheral ossifying fibroma, the peripheral odontogenic fibroma is a rare lesion. It is believed to arise from the overlying gingival epithelium or the rest of the dental lamina remaining in an extraosseous location. The epithelial component resembles the dental lamina formed during the early stages of odontogenesis. The lesion provides some evidence that it originates from a recapitulation of dental lamina, because the epithelium is capable of producing inductive changes in the connective tissue that are similar to changes seen in the dental lamina during odontogenesis. The periodontal ligament has been proposed as a possible source for POdF because these tumors are found exclusively in tooth-bearing areas of the jaws.4
Approximately 175 cases of POdF have been described in the literature.5 Most of the available data on POdF are from case reports and small case series.
Case Report
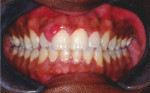
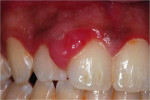
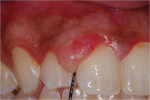
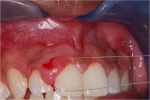
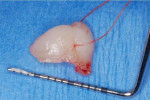
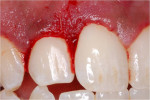
A 26-year-old African-American man was referred to the Periodontics Department of the College of Dentistry, University of Tennessee, Health Science Center for evaluation of an asymptomatic, slowly growing gingival mass in the right maxilla (Figure 1 and Figure 2). The patient reported having had a smaller lesion in the same area approximately 2 years previously, but that he had removed it himself. Since its reappearance, the lesion had been slowly growing, resulting in a much larger lesion than the previous one.
Clinical Examination
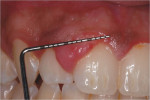
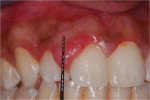
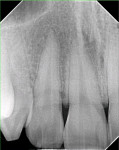
A clinical examination revealed a localized enlargement of approximately 12 mm x 9 mm (Figure 3 and Figure 4). It presented as an erythematous and inflamed sessile mass on the attached labial gingiva between the maxillary right lateral and the central incisor. The lesion was firm and painless. Radiographic examination showed no bony involvement or calcification in the lesion (Figure 5). Probing depths of teeth involved in the lesion and of adjacent teeth were within normal limits (Figure 6). Vitality tests on teeth Nos. 7 and 8 revealed vital pulps with no signs of pulpal pathology or symptoms.
Biopsy
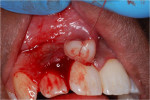
An excisional biopsy of the lesion was performed using a popular method—placing a suture under the lesion and holding the ends of the suture while incising the lesion. The specimen was placed in 10% neutral formalin and submitted for microscopic examination. The wound was sutured with resorbable sutures (Figure 7 through Figure 10).
Histologic Findings
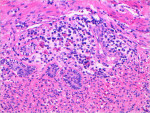
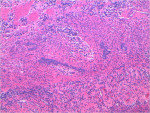
Histologic sections showed a proliferation of cellular fibrous connective tissue covered by an unremarkable layer of parakeratinized stratified squamous epithelium. Scattered collections of chronic inflammatory cells were seen beneath the mucosal layer (Figure 11). Throughout the connective tissue proliferation, strands and nests of odontogenic epithelium were interspersed (Figure 12). Some vacuolization was evident in cells arranged at the periphery of these nests. The histologic features are consistent with a diagnosis of peripheral odontogenic fibroma, WHO type.
Patient Follow-up
The patient returned in a week for follow-up. Healing of the area was uneventful and the patient did not report any subsequent problems.
Discussion
The peripheral odontogenic fibroma usually presents clinically on the gingiva as either a pedunculated or a sessile firm enlargement with a pink, smooth, nonulcerated mucosal surface. It is asymptomatic and slowly increases in size, usually ranging from 0.5 cm to 3.5 cm in diameter.4,6 However, Chen et al7 reported a unique case of a chronic untreated lesion measuring 4.5 cm × 4 cm (the only one reported over 3 cm) exhibiting the growth potential of POdF if left untreated.
The peripheral odontogenic fibroma is usually asymptomatic and its location and size vary. It does not affect the surrounding hard and soft tissues. However, in rare cases, where the lesion is larger than 3 cm in size,7 tilting of adjacent teeth and even exfoliations have been reported. Alveolar bone resorption and mild facial asymmetry have been reported as well.7 Interdental lesions often cause tooth migration. Bonetti et al8 reported a case where the POdF caused localized obstruction of permanent teeth eruption in a 12-year-old boy.
The lesion may appear at any age, starting during the first decade of life, with incidence showing a slight increase in the third decade of life. Souza9 reported in 2006 a case of POdF removal from a 4-month-old baby. That lesion had been present since birth. At 16-month follow-up, no recurrence was noted and normal primary incisors eruption was observed.
It has an equal gender distribution, with some studies finding a slight female predilection,4 while others reported a slight male predilection.1,10 It appears to affect Caucasians more often than it does African-Americans11; however, some studies have noted a higher prevalence in African-Americans.4
The POdF can arise throughout either arch but tends to occur in the mandibular canine-premolar and maxillary anterior areas.12
Radiographic changes are not commonly seen, especially in early lesions, but mineralization may be visible in larger lesions. Focal calcifications in the form of bone, cementum, and dentin are occasionally present. Large lesions may reveal saucerization of the cortical bone or some widening of the cervical portion of the periodontal space.13
The histologic spectrum of this lesion varies. Connective tissue ranges from loose (almost myxomatous) to markedly cellular or to relatively cellular and well organized.14 Islands of odontogenic epithelium are scattered throughout the connective tissue, which may be prominent or scarce. Hard tissue resembling dentin and its protein matrix are occasionally seen in both central and peripheral entities, but this finding has no prognostic importance.15
POdF is treated by complete excision of the soft lesion, including the underlying periosteum, to minimize the likelihood of recurrence. Attempts to superficially remove the lesion without curettage of the underlying bone often result in recurrence. Budding of the surface epithelium and calcification in apposition to odontogenic epithelial rests are histologic predictors of recurrence.16
There is a general lack of information on optimal management of POdF due to its rarity and lack of clinical trials. In particular, the number of studies with follow-up is limited, and therefore its potential for recurrence remains unclear.17 Except for two recent studies by Dayley et al18 and Ritwik et al16 showing recurrence rates of 38% and 50%, respectively, when follow-up information was available, the recurrence rate is considered to be low.7,18 Typically, recurrent lesions develop 1 to 4 years after surgical removal. When there is a recurrence within the first year, it is likely that this represents incomplete removal of the lesion. Recently Armas et al19 reported a case where a lesion of POdF recurred three times. The last time not only occurred after an 11-year gap—which is the longest reported time to recurrence—but also presented in association with a central odontogenic fibroma, although there were no radiographic findings on the previous two lesions. This case emphasizes the importance of radiographic survey and follow-ups.
Differential Diagnosis
Clinical differential diagnosis for peripheral odontogenic fibroma may include fibroma, peripheral giant cell granuloma, pyogenic granuloma, and peripheral ossifying fibroma (Table 1).20 These may have similar clinical appearances and courses; hence, clinical findings alone are not sufficient for a diagnosis of PODF (WHO-type), and biopsy and histopathologic examination are necessary for a definitive diagnosis. Histologically, the key feature is the presence of odontogenic epithelium and dysplastic dentin and/or cementum-like material in the POdF.
Summary
This report includes a well-documented case of peripheral odontogenic fibroma, along with a thorough review of the literature and published cases. The need for biopsy, histological evaluation, and follow-up of this lesion is strongly recommended.
DISCLOSURES
The authors do not report any financial, economic, or professional interests that may have influenced this article.
ABOUT THE AUTHORS
Rania Livada, DDS, MS
Assistant Professor, Department of Periodontology, University of Tennessee Health Science Center, Memphis, Tennessee
Norman Fine, DMD, MS
Private Practice, Greenville, South Carolina
K. Mark Anderson, DDS, MS
Associate Professor, Department of Diagnostic Sciences and Oral Medicine and Director of Oral and Maxillofacial Diagnostic Services, University of Tennessee Health Science Center, Memphis, Tennessee
Jacob Shiloah , DMD
Professor, Department of Periodontology, University of Tennessee Health Science Center, Memphis, Tennessee
REFERENCES
1. Buchner A, Merrell PW, Carpenter WM. Relative frequency of peripheral odontogenic tumors: a study of 45 new cases and comparison with studies from the literature. J Oral Pathol Med. 2006;35(7):385-391.
2. Gardner DG. The central odontogenic fibroma: an attempt at clarification. Oral Surg Oral Med Oral Pathol. 1980;50(5):425-432.
3. Gardner DG. The peripheral odontogenic fibroma: an attempt at clarification. Oral Surg Oral Med Oral Pathol. 1982;54(1):40-48.
4. de Villiers Slabbert H, Altini M. Peripheral odontogenic fibroma: a clinicopathologic study. Oral Surg Oral Med Oral Pathol. 1991;72(1):86-90.
5. Neville BW, Damm DD, Allen CM, Bouquot JE, eds. Oral & Maxillofacial Pathology. Philadelphia: WB Saunders Co; 1995:374-376, 536.
6. Garcia BG, Johann AC, da Silveira-Júnior JB, et al. Retrospective analysis of peripheral odontogenic fibroma (WHO-type) in Brazilians. Minerva Stomatol. 2007;56(3):115-119.
7. Lin CT, Chuang FH, Chen JH, et al. Peripheral odontogenic fibroma in a Taiwan chinese population: a retrospective analysis. Kaohsiung J Med Sci. 2008;24(8):415421.
8. Bonetti GA, Marini I, Zucchelli G, Checchi L. Obstruction of the eruption pathway by peripheral odontogenic fibroma: report of a patient. Am J Orthod Dentofacial Orthop. 2008;133(2):303-307.
9. Martelli-Júnior H, Mesquita RA, de Paula AM, et al. Peripheral odontogenic fibroma (WHO type) of the newborn: a case report. Int J Paediatr Dent. 2006;16(5):376-379.
10. Kenney JN, Kaugars GE, Abbey LM. Comparison between the peripheral ossifying fibroma and peripheral odontogenic fibroma. J Oral Maxillofac Surg. 1989;47(4):378-382.
11. Gardner DG. The peripheral odontogenic fibroma: an attempt at clarification. Oral Surg Oral Med Oral Pathol. 1982;54(1):40-48.
12. Rinaggio J, Cleveland D, Koshy R, et al. Peripheral granular cell odontogenic fibroma. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104(5):676-679.
13. Sapp JP, Eversole LR, Wysocki GP. Odontogenic Tumors. In: Sapp JP, Eversole LR, Wysocki GP, eds. Contemporary Oral and Maxillofacial Pathology. 2nd ed. St. Louis, MO: Mosby International; 2004:150.
14. Buchner A, Ficarra G, Hansen LS. Peripheral odontogenic fibroma. Oral Surg Oral Med Oral Pathol. 1987;64(4):432-438.
15. Gardner DG. Central odontogenic fibroma current concepts. J Oral Pathol Med. 1996;25(10):556-561.
16. Ritwik P, Brannon RB. Peripheral odontogenic fibroma: a clinicopathologic study of 151 cases and review of the literature with special emphasis on recurrence. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;110(3):357-363.
17. Curran AE. Peripheral odontogenic tumors. Oral Maxillofac Surg Clin North Am. 2004;16(3):399-408.
18. Daley TD, Wysocki GP. Peripheral odontogenic fibroma. Oral Surg Oral Med Oral Pathol. 1994;78(3):329-336.
19. Armas JM, Hunter KD, Jenkins WMM. Odontogenic fibroma: an unusual presentation. J Oral Maxillofac Pathol. 2008;12(2):68-71.
20. Rajendran R. Benign and malignant tumors of the oral cavity. In: Shafer WG, Hine MK, Levy BM, eds. Shafer’s Textbook of Oral Pathology. 5th ed. Philadelphia, PA: W.B. Saunders Co.; 2006:113-308.