Review of Intraoral Harvesting for Bone Augmentation: Selection Criteria, Alternative Sites, and Case Report
Federico Brugnami, DDS; Alfonso Caiazzo, DDS; and Cataldo Leone, DMD, DMSc
Abstract: The success of intraoral bone grafts depends, in part, on the choice of donor graft material as well as on how the material is handled. The evidence supporting the use of autogenous intramembranous bone, with or without the use of barrier membranes, is reviewed briefly. The rational of donor site selection also is presented. Advantages and disadvantages of harvest site options are discussed.
Learning Objectives:
After reading this article, the reader should be able to:
- better understand bone graft biology.
- select the most appropriate site for bone harvesting.
- optimize the small amount of bone graft available.
- recognize alternative sources for bone harvesting.
An adequate volume of bone is one of the factors critical to successful osseointegration and long-term retention of endosseous dental implants.1,2 In situations in which inadequate bone volumes exist, osseous ridge augmentation procedures often are necessary for predictable implant therapy. Although a number of different materials have been used for hard-tissue ridge augmentation during the past several decades, autogenous bone grafts are generally considered one of the more ideal augmentation materials.3,4 The choice of autogenous donor site is markedly influenced, however, by two important considerations: the quantity of bone required at the recipient site and the biologic qualities of the donor bone. In addition, successful augmentation of the recipient site is influenced by the technical, intraoperative surgical manipulations used.5
An extraoral donor site often required for ridge augmentation in totally edentulous patients, for example, where ridge resorption may be extreme and extensive.5 A popular and reasonably safe extraoral site is the posterior iliac crest, which can yield relatively large bone volumes from 70 cc to 140 cc.5 The surgical convenience of iliac grafts is negated, in part, by the additional procedural requirements and attendant patient morbidity; such procedures are longer, often require the use of general anesthesia, increase the likelihood of intra- and postoperative complications, and can result in considerable postoperative pain.6
In contrast, ridge defects in partially edentulous patients often are less severe and more localized, necessitating a smaller quantity of bone.6 This allows greater flexibility in autogenous donor site selection and, in particular, makes highly feasible the use of intraoral donor sites.6 In such cases, relatively modest bone volumes ranging from 2 cc to 10 cc from the mandibular symphysis or the ramus, for example, may be adequate for ridge augmentation (Figure 1, 2, 3, 4).6 Figure 5, 6, 7, 8, 9, 10, 11, 12 detail a case of a 60-year-old male who presented with edentulism in the posterior right mandible (missing his first, second, and third molars). Because of the limited quantity of bone, implant placement was impossible without prior bone augmentation. The treatment plan was for guided bone regeneration (GBR) for vertical augmentation, with nonresorbable membrane and autologous bone from his mandibular symphysis.
Intraoral sites generally allow for shorter procedures, avoid the need for general anesthesia, and are associated with few complications and less postoperative discomfort than extraoral sites.6 Somewhat less apparent than the bone quantity required, but no less important, are the biologic qualities of the transplanted bone. These include the bone’s embryologic origin, morphology, cytologic constituents, and biochemical composition of the extracellular matrix.6 Local harvesting is advantageous when bone volume demands are not prohibitively high because intraoral sites can serve as excellent, readily accessible sources of intramembranous bone.6 Within the mouth, the mandible tends to present more sources than the maxilla. The mandibular symphysis and ramus are excellent donor sites.6 However, these sites often are not contiguous with the area to be augmented, requiring the involvement of a second surgical site. Clearly, an alternative mandibular donor site that is contiguous with the recipient area would obviate the need for an extra surgical site. Such alternative sources for local harvesting in the mandible can be evaluated by careful clinical and radiographic examinations of the patient.6 It is important to emphasize, albeit obvious, that the anatomical factors limiting bone harvesting in the posterior mandible is the mandibular canal and associated neurovascular elements. Presurgical treatment planning, therefore, should include appropriate anatomical determinations when such alternative harvesting is considered.6
Case Report
A 42-year-old female presented for preprosthetic evaluation and treatment. Bilateral, posterior ridge augmentation was recommended, and informed consent was obtained. The clinical and radiographic findings in this case demonstrated the possibility of harvesting bone from locations contiguous with the recipient site. The left posterior mandible exhibited a moderate torus whereas the right posterior aspect presented with an edentulous anatomy that would allow resection of a corticocancellous block 2-mm thick.
Surgical Technique
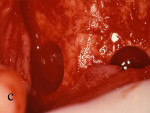
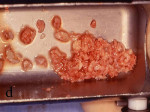
The patient was given amoxicillin 250 mg t.i.d. beginning the day of surgery, continuing for 1 week. A split-thickness incision was made, as described by Buser et al,12,13 after infiltration with local anesthesia and preoperative rinse with chlorhexidine gluconate 0.12%. Narrowness of the ridge was confirmed intraoperatively (Figure 13). The donor bone to be obtained from the retromolar area was identified, and fixation holes were prepared before graft removal (Figure 14). Then, the recipient site was prepared by intramarrow penetration with a round No. 2 carbide bur (Figure 15) followed by harvesting and fitting to the recipient site. The graft then was secured with a stainless screw (Figure 16). Next, corticocancellous chips were placed around the graft and a Gore-Tex® membrane was trimmed, shaped, placed over the graft, and stabilized with a stainless screw. Care was taken to leave space between the membrane and the adjacent tooth. On the contralateral side, pedunculated tori were present (Figure 17). The tori were removed (Figure 18), the recipient bed was prepared, and the graft was secured (Figure 19). In this case, shaping of the graft was not necessary because it sat well on the donor site. A Gore-Tex membrane was trimmed to allow coverage of the graft (Figure 20). Corticocancellous chips were additionally placed around the graft before tacking the membrane underneath the lingual flap. Wound closure was accomplished first with vertical mattress sutures, to cover the membrane and lift the flap as much as possible, then, followed by interrupted sutures to ensure primary closure. Postoperative management included chlorexidine rinses twice daily for 2 weeks, nonsteroidal analgesia as needed, and routine patient instructions.
Clinical Evaluation
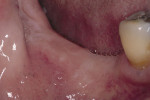
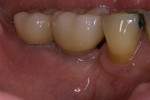
Six months after the ridge augmentation procedure, the membranes were removed (Figure 21), augmentation of the ridge assessed (Figure 22), and two endosseus implants placed in each area (Figure 23). During implant site preparation, a surgical trephine was used to obtain a bone core from the buccal of the newly formed ridge on each side (Figure 24). A preoperative radiograph and postoperative computed tomography scan showed substantial bone augmentation (Figure 25)
Histologic Evaluation
Bone cores were immediately fixed in 10% formalin and subsequently decalcified by 3 hours incubation in 100 mL of a standard solution of EDTA disodium, potassium sodium tartrate, and diluted hydrochloric acid in distilled water. The solution’s pH was < 1. Sections of 6-µm thickness were cut and stained with hematoxylin and eosin. A total of six histologic sections were evaluated for the two cores. The decalcified sections showed foci of bone with empty lacunae (consistent with the graft material) surrounded by intimately opposed woven and lamellar bone that was delineated by distinct cement lines. The new bone marrow demonstrated a mild degree of fibrosis without inflammatory reaction. There was some evidence of remodeling (Figure 26 and Figure 27).
Discussion
Quantity of bone available is a critical factor when selecting a donor site for harvesting. Somewhat less apparent than the bone quantity required—but no less important—are the biologic qualities of the transplanted bone.7 Although detailed review of each of these properties is beyond the scope of this article, further discussion of the embryologic origin of donor bone is warranted. The development of any given bone proceeds along one of two general pathways: endochondral or intramembranous ossification.8 In endochondral ossification, bone replaces a hyaline cartilage precursor. Long bones such as the tibia, fibula, and femur, as well as the iliac crest, are formed in this way.8 Intramembranous ossification proceeds by direct mineralization of the organic matrix, without a cartilaginous intermediate. The bones of the craniofacial complex, with limited exceptions, form via intramembranous ossification. The calvaria, maxillary bones, mandibular body, and mandibular ramus, in particular, are intramembranous; the mandibular condyles are exceptions because they are of endochondral origin.8
The particular embryologic origin of donor bone is recognized as one factor in the success of bone transplantation procedures. From comparative studies of craniofacial reconstruction in animals and man, it appears that intramembranous grafts tend to maintain their volume whereas endochondral grafts undergo variable degrees of resorption over variable periods of time.9-11 Thus, all other factors being equal, intramembranous rather than endochondral bone autografts may be preferred in head and neck/intraoral applications. From the preceding discussion, the relative attractiveness of intraoral sites for the harvesting of donor bone can be appreciated.
The mandibular symphysis, which is an excellent donor site, is almost invariably not contiguous with the area to be augmented. Clearly, an alternative mandibular donor site that is contiguous with the recipient area would obviate the need for an extrasurgical site. Such alternative sources for local harvesting in the mandible can be evaluated by careful clinical and radiographic examinations of the patient. Tori and exostoses, which are common intraoral exophitic findings,12 are suitable alternative bone sources. Retromolar and edentulous areas also can be accessed.
After harvesting, the donor bone must be adapted to the recipient site. Several investigators have examined the various technical considerations in this regard.13-15 These intraoperative considerations include the adequacy of donor bone volume, use of block grafts vs ground bone, method of fixation, concomitant use of barrier membranes, and degree of flap coaptation. In 1993, Buser and colleagues13 presented a technique for localized ridge augmentation using stainless steel pins to maintain space underneath a barrier membrane. Subsequently, this group modified their technique by adding corticocancellous bone grafts harvested from the retromolar area.13 Bone chips harvested from contiguous areas also were packed into the augmentation site. The rationale for using autologous bone with barrier membranes was that the bone had both space-maintaining and bone-growth-promoting properties. The benefit of the combined use of bone grafts and membranes was confirmed by Jensen et al15 who found, using a canine model, less resorption of autologous block grafts when membranes were used.
This article presented a case that demonstrated the successful treatment of localized ridge defects by combined autologous intraoral bone transplant and GBR.16-20 The bone volume required was small enough to allow harvesting from intraoral sites. Intraoral bone donor sites provide convenient surgical access, decreased procedure time, and lower morbidity.6 In addition, the donor and recipient sites are comprised of bone having the same embryologic origin (ie, intramembraneous). There seems to be some difference in treatment outcomes, intraorally, between endochondral and intraoral donor bone. Endochondral grafts have been widely used in oral and maxillofacial reconstructions, with and without osseointegrated implants. Typical donor sites are the anterior and posterior iliac crest, rib,5 and tibia.21,22 However, endochondral bone grafts are associated with delayed, sometimes dramatic resorption,11 and the associated implant success rates range from 25% to 86%.11,23-25 As a consequence, intramembraneous bone tends to be preferred in craniofacial reconstructions, again with or without implant placement.26-37 Intramembraneous, mandibular symphysis grafts have shown less delayed resorption and less morbidity than extraoral endochondral grafts.36,37 The placement of implants in areas grafted with chin bone has been documented.32-35 In particular, Jensen and Sindet-Pedersen32 reported a 94% success rate of 107 implant fixtures in 26 patients grafted with chin bone, followed up to 32 months. Other locations in the mandible also have been used to obtain intramembraneous bone; these include the retromolar region,14 ramus,6,38 and tori.38-40
Conclusion
The use of appropriate surgical techniques, supported with sound knowledge of bone biology, optimizes ridge augmentation procedures. Based on the presented review, the authors propose a hierarchy of clinical preferences for ridge augmentation in the partially edentulous patient. First, intraoral bone of intramembraneous origin is preferred over extraoral bone of either intramembraneous or endochondral origin. Second, mandibular donor bone is preferred over maxillary bone. Third, when feasible, donor bone that is contiguous with the recipient site is preferred over intraoral bone from a second distinct location. Fourth, when the transplanted bone is insufficient, additional chips of bone can be collected and packed to achieve a ridge with the desired size and shape. Fifth, when the volume of transplanted bone is barely sufficient, the concomitant use of a barrier membrane, in accordance with the principles of guided-tissue regeneration, is preferred over bone transplantation without membranes. In the presented case, all of these preferences were applied. The combined use of localized transplanted posterior mandibular bone exostoses and a barrier membrane resulted in good clinical results.
References
1. Albrektsson T, Dahl E, Enbom L, et al. Osseointegrated oral implants. A Swedish multicenter study of 8139 consecutively inserted Nobelpharma implants. J Periodontol. 1988;59(5):287-2976.
2. Adell R, Lekholm U, Rockler B, et al. A 15-year study of osseointegrated implants in the treatment of the edentulous jaw. Int J Oral Surg. 1981;10(6):387-416.
3. Brugnami F, Caiazzo A, Leone C. Local intraoral autologous bone harvesting for dental implant treatment: alternative sources and criteria of choice. Keio J Med. 2009;58(1):24-28.
4. Hammack BL, Enneking WF. Comparative vascularization of autogenous and homogenous bone transplants. J Bone Joint Surg Am. 1960;42(A):811-817.
5. Klijn RJ, Meijer GJ, Bronkhorst EM, et al. A sinus floor augmentation surgery using autologous bone grafts from various donor sites: a meta-analysis of the total bone volume. [published online ahead of print January 17, 2010]. Tissue Eng Part B Rev.
6. Misch CM. Ridge augmentation using mandibular ramus bone grafts for the placement of dental implants: presentation of a technique. Pract Periodontics Aesthet Dent. 1996;8(2):127-135.
7. Ten Cate AR. Oral Histology: Development, Structure, and Function. 4th ed. St. Louis, MO: Mosby; 1994.
8. Smith JD, Abramson M. Membraneous vs endochondral bone autografts. Arch Otolaryngol. 1974;99(3):203-205.
9. Zins JE, Whitaker LA. Membraneous versus endochondral bone: implications for craniofacial reconstruction. Plast Reconstr Surg. 1983;72(6):778-785.
10. Breine U, Brånemark PI. Reconstruction of alveolar jaw bone. An experimental and clinical study of immediate and preformed autologous bone grafts in combination with osseointegrated implants. Scand J Plast Reconstr Surg. 1980;14(1):23-48.
11. Shafer WG, Hine MK, Levy BM. A Textbook of Oral Pathology. 4th ed. Philadelphia, PA: W.B. Sanders Company; 1983.
12. Buser D, Dula K, Belser U, et al. Localized ridge augmentation using guided bone regeneration. I. Surgical procedure in the maxilla. Int J Periodontic Restorative Dent. 1993;13(1):29-45.
13. Buser D, Dula K, Belser UC, et al. Localized ridge augmentation using guided bone regeneration. II. Surgical Procedure in the mandible. Int J Periodontic Restorative Dent. 1995;15(1):10-29.
14. Donos N, Kostopoulos L, Tonetti M, et al. Long-term stability of autogenous bone grafts following combined application with guided bone regeneration. Clin Oral Implants Res. 2005;16(2):133-139.
15. Becker W, Becker BE, Handelsman M, et al. Bone formation at dehisced dental implant sites treated with implant augmentation material. A pilot study in dogs. Int J Periodontic Resorative Dent. 1990;10(20:92-101.
16. Brugnami F, Then P, Moroi H, et al. Histologic evaluation of human extraction sockets treated with demineralized freeze-dried bone allograft (DFDBA) and a cell occlusive membrane. J Periodontol. 1996;67(8):821-825.
17. Simion M, Jovanovic SA, Tinti C, et al. Long-term evaluation of osseointegrated implants inserted at the time or after vertical ridge augmentation. A retrospective study on 123 implants with 1-5 year follow-up. Clin Oral Implants Res. 2001;12(1):35-45.
18. Degidi M, Scarano A, Piattelli A. Regeneration of the alveolar crest using titanium micromesh with autologous bone and a resorbable membrane. J Oral Implantol. 2003;29(2):86-90.
19. Brugnami F, Caleffi C. Prosthetically driven implant placement. How to achieve the appropriate implant site development. Keio J Med. 2005;54(4):172-178.
20. Kfir E, Kfir V, Kaluski E. Immediate bone augmentation after infected tooth extraction using titanium membranes. J Oral Implantol. 2007;33(3):133-138.
21. Catone GA, Reimer BL, McNeir D, et al. Tibial autogenous cancellous bone as an alternative donor site in maxillofacial surgery: a preliminary report. Int J Oral Maxillofac Surg 1992;50(12):1258-1263.
21. O’Keefe RM Jr, Reimer BL, Botterfield SL. Harvesting of autogenous bone graft from the proximal tibial metaphysis. A review of 230 cases. J Orthop Trauma. 1991;5(4):469-474.
22.Keller EE, Van Roekel NB, Desjardins RP, et al. Prosthetic-surgical reconstruction of the severely resorbed maxilla with iliac bone grafting and tissue-integrated prostheses. Int J Oral Maxillofac Implants. 1987;2(3):155-165.
23. Kahnberg KE, Nystrom E, Bartholdsson L. Combined use of bone grafts and Branemark fixtures in the treatment of severely resorbed maxillae. Int J Oral Maxillofac Implants. 1989;4(4):297-304.
24. Jensen J, Simonsen EK, Sindet-Pedersen S. Reconstruction of the severely resorbed maxilla with bone grafting and osseointegrated implants: a preliminary report. J Oral Maxillofac Surg. 1990;48(1):27-33.
25. Kusiak JF, Zins JE, Whitaker LA. The early revascularization of membranous bone. Plast Reconstr Surg. 1985;76(4):510-516.
26. Hardesty RA, Marsh JL. Craniofacial onlay bone grafting: a prospective evaluation of graft morphology, orientation, and embryonic origin. Plastic Reconstr Surg. 1990;85(1):5-15.
27. Moskalewsky S, Osiecka A, Maleczyk J. Comparison of bone formed intramuscularly after transplantation of scapular and calvarial osteoblasts. Bone. 1988;9(2):101-106.
28. Koole R, Bosker H, van der Dussen FN. Late secondary autogenous bone grafting in cleft patients comparing mandibular (ectomesenchymal) and iliac crest (mesenchymal) grafts. J Craniomaxillofac Surg. 1989;17(suppl 1):28-30.
29. Donovan MG, Dickerson NC, Hanson IJ, et al. Maxillary and mandibular reconstruction using calvarial bone grafts and Branemark implants: a preliminary report. J Oral Maxillofac Surg. 1994;52(6):588-594.
30. Gary JJ, Donovan M, Garner FT, et al. Rehabilitation with calvarial bone grafts and osteointegrated implants after partial maxillary resection: a clinical report. J Prosthetic Dent. 1992;67(6):743-746.
31. Jensen J, Sindet-Pedersen S. Autogenous mandibular bone grafts and osseointegrated implants for reconstruction of the severely atrophied maxilla: a preliminary report. J Oral Maxillofac Surg. 1991;49(12):1277-1287.
32. Jensen J, Sindet-Pedersen S, Oliver AJ. Varying treatment strategies for reconstruction of maxillary atrophy with implants: results in 98 patients. J Oral Maxillofac Surg. 1994;52(3):210-218.
33. Misch CM. Enhance maxillary implant sites through symphysis bone graft. Dent Implantol Update. 1991;2:101-104.
34. Misch CM, Misch CE. Autogenous mandibular bone graft for reconstruction of ridge deficiencies prior to implant placement. Int J Oral Maxillofac Implants. 1993;8:117-123.
35. Sindet-Pedersen S, Enemark H. Mandibular bone grafts for reconstruction of alveolar clefts. J Oral Maxillofac Surg. 1988;46(7):533-537.
36. Sindet-Pedersen S, Enemark H. Reconstruction of alveolar clefts with mandibular or iliac crest bone grafts: a comparative study. J Oral Maxillofac Surg. 1990;48(6):554-560.
37. Jensen J, Reiche-Fischel O, Sindet-Pedersen S. Autogenous mandibular bone grafts for molar augmentation. J Oral Maxillofac Surg. 1995;53(1):88-90.
38. Ganz SD. Mandibular tori as a source for onlay bone graft augmentation: a surgical procedure. Pract Periodontics Aesthet Dent. 1997;9(9):973-984.
39. Proussaefs P. Clinical and histologic evaluation of the use of mandibular tori as donor site for mandibular block autografts: report of three cases. Int J Periodontics Restorative Dent. 2006;26(1):43-51.
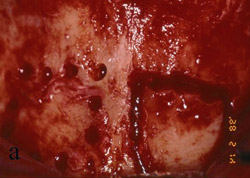 Figure 1 Outline of the block graft to be removed from the mandibular symphysis. | 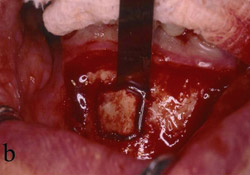 Figure 2 The block graft was harvested with a chisel. | ||||||
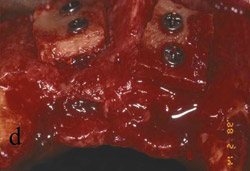 Figure 3 The symphysis after the blocks was removed. | 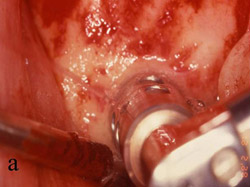 Figure 4 The block grafts were placed and secured with screws. | ||||||
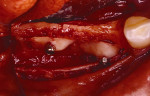
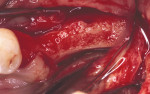
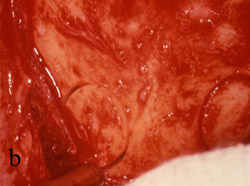 Figure 5 A trephine was used to begin the harvesting procedure. | 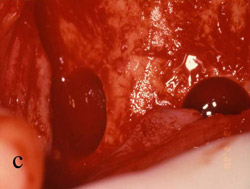 Figure 6 After the trephine was removed, outlines of the grafts were evident. | ||||||
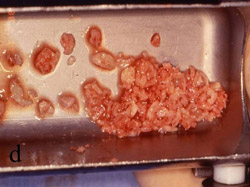 Figure 7 The bone was removed from the mandibular symphysis. | 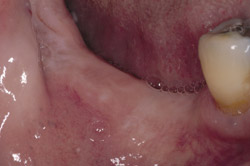 Figure 8 The bone then was particulated for vertical ridge augmentation with GBR. | ||||||
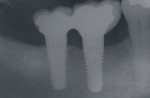
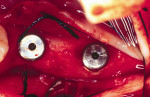
 Figure 9 Soft-tissue before vertical ridge augmentation. Note concavity of the edentulous ridge. | 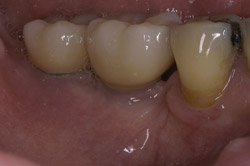 Figure 10 Radiograph after vertical ridge augmentation. Note the native bone, the titanium-reinforced membrane, and the grafted area underneath. | ||||||
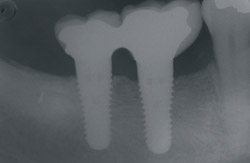 Figure 11 Final restoration in place. Note the height of the abutments is comparable with the clinical crowns of the contiguous teeth. | 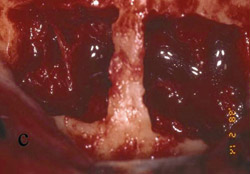 Figure 12 A radiograph confirmed stabilization of the augmentation around the implants after loading. With the vertical augmentation, the implants were positioned more coronally, with a better final crown-to-root ratio. | ||||||
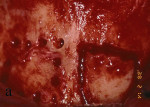
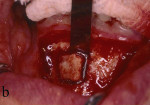
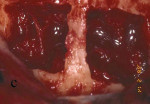
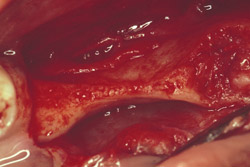 Figure 13 Occlusal view of the damaged ridge showing insufficient thickness for implant placement. | 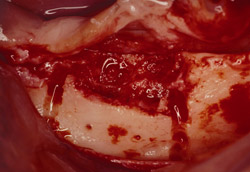 Figure 14 Donor graft is removed from the retromolar area. Fixation holes were prepared before removing the graft. | ||||||
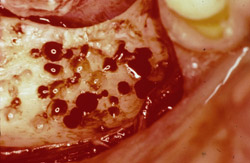 Figure 15 The recipient site then was prepared with a round No. 2 carbide bur to accomplish intramarrow penetration. | 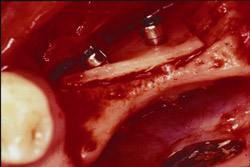 Figure 16 Occlusal view of the graft in place. | ||||||
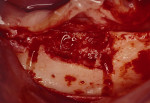
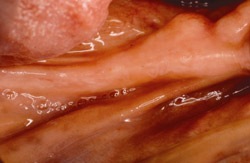 Figure 17 On the contralateral side, pedunculated tori were noted. | 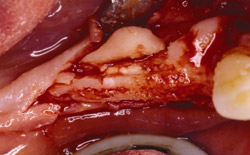 Figure 18 The tori were harvested. | ||||||
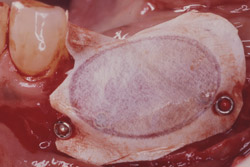 Figure 19 Occlusal view of the tori grafted into place. | 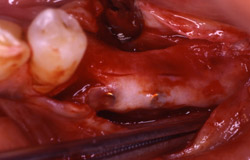 Figure 20 Gore-Tex nonresorbable membrane in place. | ||||||
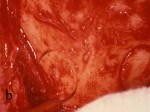
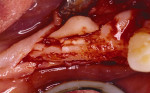
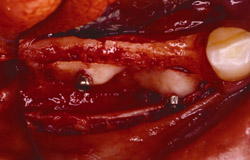 Figure 21 Gore-Tex nonresorbable membrane before removal at implant placement. | 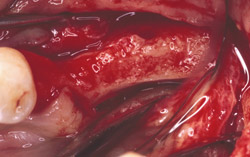 Figure 22 Augmented ridge before implant placement. | ||||||
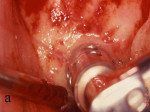
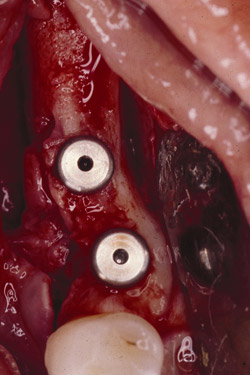 Figure 23 The implants were placed completely into the bone. | 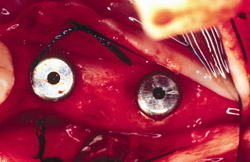 Figure 24 The implants were placed and a sample was harvested from the buccal for histology. | ||||||
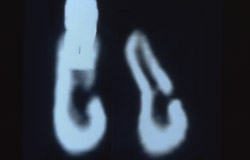 Figure 25 Pre- and postoperative CT scan showing augmented ridge at the level of the mental foramen. | |||||||
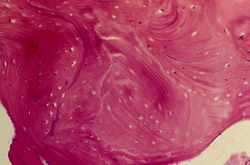 Figure 26 | 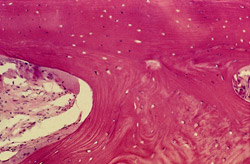 Figure 27 | ||||||
| Foci of bone with empty lacunae (consistent with the graft material) surrounded by intimately opposed woven and lamellar bone demarcated by distinct cement lines. The new bone marrow demonstrated a mild degree of fibrosis without inflammatory reaction. There was some evidence of remodeling (Original magnification x 30). | |||||||
| |||||||