Utilization of Bioactive Dentin for Guided Bone Regeneration as Part of Implant Treatment
Robert A. Horowitz, DDS; Gregori M. Kurtzman, DDS, MAGD; and José Luis Calvo-Guirado, DDS, PhD, Eu PhD, MS
Abstract: When a tooth is to be extracted and the site utilized for implant placement, whether delayed or immediate, the extracted tooth may be processed and used as an autologous graft material for placement into the site for guided bone regeneration. This approach eliminates potential immunological issues that may arise from utilization of an allograft, xenograft, or synthetic materials, plus it provides host stimulation for conversion to host bone at the intended site. Periodontal repair of bone loss at and adjacent to sites that will undergo extraction is also an ideal application for autologous dentin grafting. This article reviews several different clinical situations utilizing autologous dentin grafting at the time of extraction for guided bone regeneration.
At the time of tooth extraction there are often objectives more important to the patient's final treatment plan than just allowing natural fill of the extraction socket. Loss of the tooth in its alveolus triggers a cascade of biological events that typically results in significant local anatomic changes, which when implants are planned may hamper their placement.1 This can be related to planned placement of an implant either at the time of extraction, following site healing (ie, delayed approach), or following repair of adjacent periodontal bone loss. Instances may occur where periodontal issues that result in the need to extract a tooth spread to adjacent teeth or implants resulting in loss of bone and a need to graft those areas to maintain the remaining dentition. These deficits may be horizontal, vertical, or combined. In the absence of predictable periodontal regenerative therapies, these teeth or implants could be sacrificed and replaced by fixed or removable prostheses. Preservation of the extraction socket and gingiva when an implant is being placed in a delayed manner should always be considered to prevent the loss of crestal bone that could compromise subsequent implant placement.2
Conventionally, to obtain predictable esthetic results after immediate socket therapy, a multitude of steps have been required.3,4 A simpler approach that allows the surgeon/restorative team to obtain excellent esthetic results may be possible. The patients presented in this case series were treated with a combination of a bioactive dentin bone replacement graft and a bioactive amnion-chorion barrier. Several types of cases are shown with follow-ups of up to 51 months. Maintenance of alveolar volume has been consistent when postoperative cone-beam computed tomography (CBCT) scans have been taken, and postoperative radiographs have demonstrated clinical bone maintenance or gain of up to 6 mm and more depending on the indication treated.
Bioactive Dentin Grafting
Various osseous graft materials have been documented in the literature for treatment of extraction sockets.5 These include autografts, typically sourced from the tuberosity region of the maxilla, ramus, or anterior mandible, allografts, xenografts, and synthetic materials, with each demonstrating clinical success as well as potential drawbacks.6-17
An alternative to these graft materials for use as an aid in socket preservation or augmentation at the time of immediate implant placement is utilization of the extracted tooth as the graft material. This autologous dentin graft, which following preparation results in either mineralized or partially demineralized dentin matrix (DDM), has been shown histologically to be well tolerated by the host, with no reported inflammatory reactions, and is highly biocompatible and osteogenic in nature with full conversion to host bone over 4 to 6 months.18-20 DDM has demonstrated great potential for osteoinductivity with low antigenicity without provoking an immunologic reaction.21 Histologically, DDM has been shown to work well in association with the adjacent implant surface with conversion of the dentin particles over time and no reported clinical issues.22 Autogenous dentin graft has been reported to have the capacity for bone formation during the early period of the site.23,24 DDM acts as a scaffold for osteoblasts, providing osteoconductive properties to the material.25 Additionally, its osteoinductive properties have been documented to improve graft conversion and replacement by host bone, providing a healed site that can accommodate implant placement. At the time of graft augmentation it is also possible to repair exposed threads on an implant that were affected by periodontal issues.26,27
Preparation of Autologous Dentin Graft
To prepare the autologous dentin graft, any restorative materials on the extracted tooth are removed with a high-speed handpiece. Enamel does not need to be removed from the extracted tooth prior to processing. The tooth is dried with a sterile gauze and placed into the dentin grinding device (Smart Dentin Grinder®, KometaBio, kometabio.com) to be processed. Should the tooth be nonvital it may be utilized in this manner. However, if an endodontic filling material has been placed in the tooth, no published studies on how to completely remove the filling material, sterilizing liquids, or cement prior to grinding have been published yet; therefore, the present authors do not incorporate such teeth in their surgical protocols.
Following processing in the dentin grinder, graft particles in the range of 250 µm to 1,200 µm are produced. A cleanser (Dentin Cleanser, KometaBio) of sodium hydroxide in 20% ethanol with a high pH (very basic) is added to the particles to eliminate any bacteria and remaining organic material. After 5 minutes the cleanser is removed by using sterile gauze to wick away the moisture. As an additional step, ethylenediaminetetraacetic acid (EDTA) can be used to partially demineralize the particles to reveal more of the collagen and thereby gain more site activation sooner in the healing process. Next, a dentin wash (Dentin Wash, KometaBio) consisting of phosphate-buffered saline is added to the mixture and allowed to sit for a few seconds. Following this second soaking, the liquid wash is removed with a sterile gauze.28
The graft is now ready for use. To improve handling of the material, the autologous dentin graft particles may be combined with leukocyte platelet-rich fibrin (L-PRF) to form a gelatinous mass prior to site placement.23 The type of graft and the particle sizes are chosen based on the specific use of the graft and how long it will need to remain in situ. During processing of the tooth, the grinding and sorting protocols may be varied, so that smaller or larger particles may be obtained, thus altering the overall rate of graft resorption and new bone incorporation in the treated site. For small defects like infrabony periodontal pockets or immediate implant-socket gaps, small particles may be beneficial to obtain new bone quicker. In cases where there are large sockets, possibly missing bony walls, having a higher proportion of large particles should enable the material to maintain its scaffolding properties longer.
Cases
Case 1: Delayed Implant Placement Following Socket Preservation
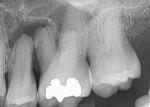
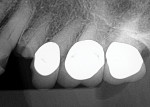
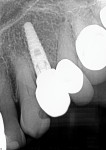
A 52-year-old man presented with purulent exudate draining between the maxillary left second premolar and first molar (teeth Nos. 13 and 14). Examination noted class 2+ mobility on both teeth and periodontal probing between these teeth to the apices at ≥10 mm. The patient reported some sinus congestion on the left side. A periapical radiograph was taken that confirmed loss of interproximal bone with a large apical lesion involving the second premolar and mesiobuccal root of the first molar (Figure 1). Upon CBCT evaluation, a lack of buccal and lingual bone associated with the two teeth was noted, as was sinus involvement of the dental infection resulting in a gray appearance on the scan signifying sinus congestion. Additionally, an absence of bone was noted at the trifurcation of the first molar.
Clinical findings were discussed with the patient, who was informed that the prognosis for the two teeth was hopeless due to the bone loss and that they would require extraction. A treatment plan was formulated to extract the affected teeth, utilize them as an autologous dentin graft material, graft the sites, and allow healing and incorporation of the graft material over several months. Then, implants would be placed and allowed to integrate prior to restoration. The patient accepted the treatment as outlined and reviewed and signed consent forms.
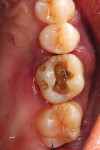
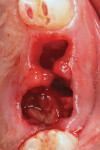
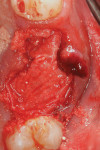
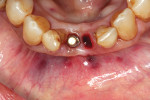
Local anesthetic was administered. Blood was drawn to create L-PRF to be combined with the autogenous graft material. Restorative material was removed from the two teeth to be extracted with a carbide in a high-speed handpiece (Figure 2). The teeth were extracted atraumatically with periotomes, straight elevators, and forceps. They were debrided of soft tissue and calculus and then processed into particulate material for grafting in the dentin grinder (Smart Dentin Grinder). The extracted teeth were processed into an autogenous dentin graft material that was sterilized in the unit and partially demineralized. The graft material was then mixed with pieces of L-PRF membranes created from the patient's drawn blood, which hydrated the dentin graft particles to create an L-PRF block ("bone block"). The adjacent teeth were scaled and root planed, and the extraction sockets were carefully debrided to avoid tearing the sinus (Schneiderian) membrane that was exposed (Figure 3). A radiograph was taken to evaluate the extraction sockets in relation to the maxillary sinus and determine if a perforation may have resulted. The adjacent exposed root surfaces were treated with citric acid for 2 minutes and rinsed thoroughly to aid in reattachment during site healing after graft placement.29
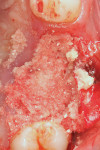
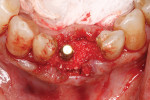
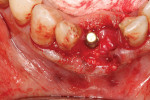
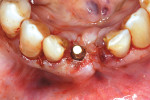
An L-PRF membrane was placed over the exposed Schneiderian membrane within the extraction sockets, and the sockets were filled with the L-PRF block of autologous dentin graft material (Figure 4). An amnion-chorion barrier (BioXclude®, Snoasis Medical, snoasismedical.com) was placed over the graft material, and a PRF membrane was added as a barrier over the amnion-chorion membrane (Figure 5). Because primary closure would not be possible and, thus, a large area of the crest would be exposed to the oral environment, a dense polytetrafluoroethylene (PTFE) barrier (Cytoplast™ TXT, Osteogenics, osteogenics.com) was placed over the site in the first molar region to decrease the potential of the patient disrupting early healing with mastication or brushing. Sutures were placed without tension to stabilize the soft tissues but not to obtain primary closure or compress the graft material. The patient was placed on antibiotics (Augmentin® 875 mg twice a day for 7 days) and over-the-counter pain medication for 2 days. A periapical radiograph was taken to document graft fill of the extraction sockets.
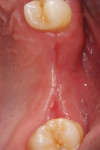
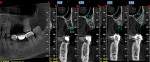
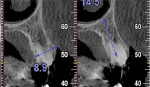
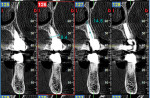
One year postoperatively, clinical examination noted healthy keratinized soft tissue covering the grafted site (Figure 6). A CBCT was taken to evaluate the osseous graft healing and the graft's incorporation to the host bone and determine if adequate volume of bone was present to allow implant placement (Figure 7). The scan demonstrated a fully clear sinus and sufficient bone to permit placement of two implants. Following local anesthetic administration a crestal incision was made and the crest was exposed to reveal dense bone with no visibly identifiable graft particles. An initial osteotomy was created in the second premolar and first molar sites with guided surgery. To achieve desired increased implant length, a crestal sinus augmentation was planned, and to improve bone quality surrounding the implant to be placed, an osseodensification protocol was used.30 Crestal sinus augmentation was performed with a combination of piezosurgery (Mectron, dental.mectron.us) and osseodensification (Versah, versah.com). These proprietary drills were also used to increase the density of the bone and improve osseointegration simultaneously.31
Two implants (Touareg™, Adin, adin-implants.com), 4.2 mm diameter in site No. 13 and 5 mm diameter in site No. 14, were placed with crestal grafting of dental putty (NovaBone® Dental Putty, Osteogenics). Secondary grafting with putty (SynOss™, Collagen Matrix, collagenmatrix.com) was performed to widen the alveolus on the facial aspect of the ridge. The graft was hydrated in the liquid from the preparation of the L-PRF as was a collagen membrane barrier (MatrixDerm®, Collagen Matrix), which was placed over the crestal and facial aspects of the graft. The site was closed with sutures achieving primary closure allowing the implants to heal in a submerged manner.
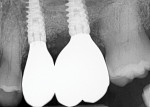
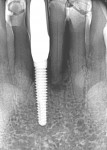
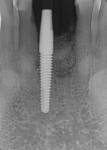
The patient presented at 7 months post-implant insertion to initiate the restorative phase of treatment. Following local anesthetic administration the implants were uncovered. Implant stability was tested using implant stability quotient (ISQ) (Osstell, osstell.com), recording ISQ measurements of 72 for implant No. 14 and 75 for implant No. 13, confirming stability to allow restoration to be initiated. The implants were prosthetically loaded with screw-retained restorations. At 2.5 years post-restoration a radiograph demonstrated bone and graft stability at the crestal aspect of the implants with a blend of the autogenous dentin graft and host bone (Figure 8).
Case 2: Immediate Implant Placement With Socket Grating
A 97.5-year-old healthy female patient presented for an emergency appointment with a fractured maxillary left first premolar (tooth No. 12). Examination noted the tooth was missing the coronal portion and was level with the gingival margin. A periapical radiograph was taken, and the tooth was deemed nonrestorable due to the degree of missing tooth structure (Figure 9). A CBCT cross-section view of the affected tooth demonstrated the dimensions of the space and an intact buccal aspect of the ridge (Figure 10). Treatment options were discussed with the patient that included extraction of the remaining root followed by either preparation of the teeth mesial and distal to the space and placement of a fixed partial denture or placement of an implant and restoration with a crown. The patient said she did not want her "teeth cut down" and selected the implant treatment option.
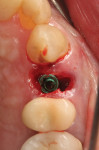
The patient reviewed and signed consent forms. Local anesthetic was administered and the residual root was extracted with the assistance of piezosurgery. Anticipating the need for graft placement around the implant to fill any socket void, the clinician used the piezosurgery unit scaler tip to remove soft tissue from the root surfaces. The root was then processed with the Smart Dentin Grinder following the manufacturer's instructions to create the autologous graft material. The socket was debrided and an osteotomy was created to accommodate a 3.7 mm x 13 mm implant (Intrahex, BioHorizons, biohorizons.com), which was placed toward the palatal wall of the extraction socket (Figure 11). An ISQ measurement was taken to check initial stability and presented a reading of 58. A gap was present buccally creating a void between the implant and socket wall. The gap was grafted with partially demineralized dentin created earlier in the appointment; it was mixed with L-PRF from blood drawn at the start of the appointment to create "sticky bone." Additional graft material was placed over the implant's shoulder to cover the alveolar crest and extend partly to the height of the connective tissue. An amnion-chorion barrier (BioXclude) was placed over the site and tucked under the flap margin on the buccal and palatal aspects, and then covered with a PRF plug (Figure 12). Polyglycolic acid sutures were placed to secure the underlying materials. While the PRF was not needed for healing, it was placed so that the patient would avoid chewing on the left side and not brush the area postoperatively during the initial healing period. The patient was seen 1 month postoperatively, and full gingival coverage over the surgical site was noted.
The patient returned 3 months post-implant placement and a periapical radiograph was taken to check the implant and graft healing. The graft that had been placed in the buccal gap to fill the void had blended with the surrounding host bone, and it was determined that restoration could be initiated and the implant was uncovered with a small surgical flap. An ISQ measurement of 78 was noted and a healing abutment was placed. Following gingival healing, the restorative phase was initiated and an impression was taken. A screw-retained zirconia-to-titanium-base crown was fabricated and delivered.
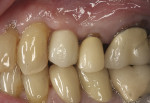
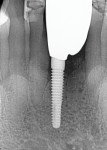
The patient remained on regular hygiene recall appointments. A periapical radiograph at 1 year post-restoration confirmed bone stability in relation to the implant and crestal bone (Figure 13). The area presented with healthy gingival tissue with no inflammation, minimal pocketing was noted, and acceptable esthetics had been maintained (Figure 14). At the time of this writing, the most recent follow-up was 4.5 years post-treatment, with the patient being 102.5 years old and in continued good health. CBCT cross-sections taken at that time demonstrated maintenance of the bucco-palatal ridge width by the autologous dentin graft placed in the buccal gap of the extraction socket (Figure 15).
Case 3: Extraction Socket Preservation With Adjacent Periodontal Tooth/Implant Grafting
A 75-year-old male patient who started smoking at age 72 but had no other contributing medical factors presented with issues associated with a one-stage implant that had been placed and restored in the mandibular right central incisor location 12 years prior. Examination noted a lack of papilla on the mesial and distal aspects of the implant restoration with intimate contact between the implant crown and the left central incisor from the cervical to the incisal edge with recession of the gingival margin on the two central incisors. A periapical radiograph noted a periapical lesion on the left central incisor with significant bone loss around that tooth and approximately 30% to 45% bone loss around the implant (Figure 16). Periodontal probing noted a depth of 6 mm between the implant crown and right lateral incisor, 5 mm between the implant crown and left central incisor, and 5 mm between the right lateral incisor and canine as measured from the facial with gingival recession noted on those teeth. The implant presented with no mobility and was deemed stable.
Based on the clinical findings the left central incisor was deemed hopeless, and it was recommended to the patient to extract the tooth, graft the extraction socket and exposed adjacent implant threads, and use a cantilever pontic off of the implant for restoration. The patient accepted treatment recommendations and signed and reviewed the consent form.
Following local anesthetic administration, tooth No. 9 was extracted (Figure 17), any soft tissue adherent to the root was curetted off the tooth, and the tooth was then ground and processed with the Smart Dentin Grinder. While the autologous dentin was being processed, a flap was elevated on the facial and lingual aspects of No. 8 to expose the implant threads that were supracrestal. They were curetted and treated with a titanium brush (RotoBrush-Titanium® Thread Cleaning Brush Kit, Salvin, salvin.com) to remove surface accretions. The No. 9 socket and adjacent root surfaces were debrided and root planed in preparation for graft placement. All exposed areas were treated with citric acid.32
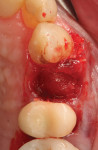
The autologous ground dentin was mixed with an equal portion of mineralized cancellous allograft (Ossif-i, Surgical Esthetics, surgicalesthetics.com) and hydrated with saline to increase the graft volume. The graft mixture was then placed in the extraction socket and around the implant and adjacent teeth to the level of the ideal alveolar crest (Figure 18). A barrier of amnion-chorion membrane (BioXclude) was placed around the implant, allowing the head of the implant to emerge through the membrane, and over the adjacent grafted area and tucked under the flap margins (Figure 19). The flaps were closed around the exposed implant shoulder, and polyglycolic acid sutures were placed to secure them in place (Figure 20). A periapical radiograph was taken to document the level of graft placement (Figure 21). A cantilever provisional restoration was placed.
The areas healed well and uneventfully. At the 18-month follow-up the restoration was removed demonstrating thick, healthy gingival tissues. Probing depths of 2 mm with no bleeding on probing were noted around the implant and healthy soft tissue was in contact with the restoration. Radiographically, 3 mm to 5 mm of vertical bone height was gained on the adjacent teeth, and most of the previously exposed threads on the existing implant were covered with bone (Figure 22). The autologous dentin graft had completely blended with the surrounding host bone, and no differentiation could be noted radiologically between the bone and previously placed graft.
Discussion
Dental graft material should have osteoconductive properties and, ideally, be osteoinductive. It should have osteostimulatory properties with a mineralized matrix and native collagenous and non-collagenous proteins. A key aspect in selection of graft material should be the osteoconductive properties acting as a scaffold for bone regeneration but also, ideally, the osteoinductive properties instigating accelerated bone regeneration and healing dynamics. Autologous graft material such as dentin presents a mineralized matrix with native collagenous and non-collagenous proteins.
Autogenic extracted teeth utilized as block grafts for lateral augmentation of the ridge33,34 or processed into a particulate dentin35-37 have been shown to undergo gradual resorption replacement similar to autogenous bone. Teeth require extraction for a variety of reasons, including advanced periodontal disease, nonrestorable coronal tooth structure, vertical root fracture, and others. Following tooth extraction, the alveolar ridge around the extraction socket undergoes remodeling during healing, specifically on the buccal aspect of the ridge.38 This leads to a decrease in bone volume at the site such that when implants are planned, implant stability, esthetics, and functionality may be compromised.39 During the initial 3 months following tooth extraction, the occurrence of up to 50% reduction in alveolar ridge volume has been reported.40,41 Studies have shown differing results regarding whether bone loss is higher with a delayed implant placement approach versus an immediate placement approach.42,43 Thus, when delayed placement is indicated, socket preservation will aid in preserving the ridges' volume and limit, if not prevent, resorption during site healing following extraction. When immediate placement is feasible, graft placement has been shown to improve implant stability and marginal bone levels, filling the void between the extraction socket walls and the implant.44
Alveolar bone and teeth develop from cells of the same macromolecular structure, containing type 1 collagen, hydroxyapatite (HA) mineral, and various non-collagenous proteins that are common to bone, dentin, and cementum. Teeth themselves are composed of three mineralized structures: enamel (a HA crystalline structure with no organic matrix), dentin, and cementum; the latter two are less mineralized than enamel and have similar composition to membranous bone.45 Dentin, therefore, which comprises 90% of tooth structure, can serve as a native bone grafting material similar to allograft or autograft bone utilized for the same purpose.
Dentin is composed of 40% to 45% HA mineral and a similar volume of organic matrix. The organic matrix consists of 90% type 1 collagen fibers, with non-collagenous molecules attached to those fibers. This makes the organic matrix an attractant for osteogenic cells to induce growth and differentiation of bone.46 Dentin phosphoprotein, a regulatory protein expressed in dentin and to a lesser extent in bone, has been implicated in bone mineralization and has an ability to regulate both initial mineralization and remodeling. During mineralization of the bone matrix, HA minerals are deposited around and between collagen fibers, protecting the organic dentin matrix from enzymatic degradation. As it has been reported that growth factors are conserved in the tooth, dentin particulate can be recognized by osteoclasts and resorbed in a similar manner to membranous cortical bone.47 These dentin particles are then fully or partially resorbed, being replaced by new bone, as non-collagenous molecules like dentin phosphoprotein, fibroblast growth factor, and bone morphogenetic protein are released during dentin resorption. The resorption of the dentin particles during the remodeling process occurs at a much slower pace than that of allograft materials. This preserves the site and the volume grafted allowing the host to convert the dentin graft into viable bone without loss of volume as has been observed with other graft materials in use clinically.
Utilization of the demineralized dentin form of processed autogenous dentin graft has proven to attract more osteogenic cells, producing a greater amount of woven bone during site healing and maturation of the graft.48,49 Although small particles of demineralized dentin may resorb faster than mineralized dentin graft particles, using a combination with large particles will help preserve volume over time. It has been suggested that the dentin particulate graft be treated with EDTA for 2 minutes to create a thin 10 μm surface layer of demineralized dentin to induce the release of attractant molecules,50 thus providing a thin surface of demineralized dentin to attract osteogenic cells with concomitant deposition of bone during the early regenerative phase of bone remodeling. This process would leave most of the mineralized dentin intact extending the time the graft will remain prior to resorption and conversion to host bone.
Autologous dentin particles with a size of 250 μm to 1,200 μm prepared with the Smart Dentin Grinder protocol were used as a grafted biomaterial in socket site preservation procedures. Postoperative CT scans documented that the particulated dentin maintained the 3-dimensional volume of the socket sites. Histologically, new bone formation was reported filling the entire grafted volume previously occupied by the roots of the extracted tooth, and the grafted dentin particles were replaced by viable host bone.51
Whether or not socket preservation is utilized, alveolar resorption can be limited by sealing the coronal aspect of the socket.52 Limiting soft-tissue ingrowth of the socket and/or graft material allows the slower process of osteoblastic activity and angiogenesis to progress, maintaining the ridges' dimensions. Various barrier membranes are available for application with extraction socket preservation whether implants are being placed simultaneously with the graft or not. Resorbable membranes of biological origin have been documented to provide the best results with minimal potential for complications that have been reported with other resorbable or nonresorbable membranes commonly used in the past.53
Bioactive barriers, some using processed human amnion and chorion tissue as their structure, are available.54 Additionally, the growth-stimulating effects of amnion-chorion membranes and amnion-chorion's increased mechanical strength with a lack of immunogenicity make it ideal for delivering allogeneic cells and for tissue engineering applications. One such membrane (BioXclude), which was used in the cases documented in this report, contains various growth factors and biologic mediators important for bone and keratinized tissue formation.55 These stimulatory factors include platelet-derived growth factor, vascular endothelial growth factor, laminin 5, extracellular matrix proteins, and various types of collagen.56 Of benefit in extraction socket preservation, the amnion-chorion membrane does not require primary closure, as some exposure is to be expected when treating an extraction site. The membrane has been demonstrated to decrease inflammation.57 Also, antibacterial properties have been reported; these properties, especially with regard to extraction sites where primary closure is not achievable, provide an advantage to the membrane's use to combat bacterial accumulation that may result upon completion of the surgical treatment, leading to compromised tissue healing and a decrease in regenerative potential.57,58 Clinical studies have demonstrated accelerated periodontal healing, increased rapid flap reattachment, and decreased postoperative recession.59,60 These membranes may be used in combination with other barriers such as those fabricated from autogenous blood concentrates like PRF.
Conclusion
When tooth extraction is indicated, the tooth may be processed and utilized as a particulate graft material similar to other available graft materials. Coming from the patient who is receiving the graft, autologous dentin grafts have demonstrated conversion to host bone without any potential immunological issues, which although rare have been reported with some commercial graft products. The cases shown in this article demonstrated full regrowth of the height of alveolar bone and keratinized tissue in treated sites. This approach may offer the added benefit of reducing the risk of periodontal and peri-implant diseases through regeneration of the initial alveolar complex. By decreasing avascular spaces around graft particles and increasing vascular supply in the area, the treated site is able to return to a histological appearance of "native" bone. Physiologically, autologous dentin grafts should be well able to recruit the body's healing abilities to treat the disease process and reduce any potential inflammatory response to plaque, which causes bone loss and future periodontitis or peri-implantitis. Additionally, treatment cost is reduced as packaged graft materials are not required, and secondary surgical sites associated with autogenous grafts taken from elsewhere intraorally are not needed. The use of the bioactive graft and barrier shown in this case series can be considered when immediate or delayed implant placement protocols are indicated or in socket preservation and associated repair of periodontal bone loss on adjacent teeth or integrated implants.
Disclosure
Dr. Horowitz has lectured for, performed research sponsored by, and received honoraria from KometaBio and Snoasis Medical.
About the Authors
Robert A. Horowitz, DDS
Adjunct Clinical Assistant Professor, Departments of Oral and Maxillofacial Surgery and Periodontology and Implant Dentistry, New York University College of Dentistry, New York, New York
Gregori M. Kurtzman, DDS, MAGD
Former Assistant Clinical Professor, Department of Restorative Dentistry and Endodontics, University of Maryland School of Dentistry; Diplomate, International Congress of Oral Implantologists; Private Practice, Silver Spring, Maryland
José Luis Calvo-Guirado, DDS, PhD, Eu PhD, MS
Associate Researcher, Universidad Autonoma de Chile, Santiago, Chile;
Private Practice, Murcia, Spain
References
1. Avila-Ortiz G, Elangovan S, Kramer KWO, et al. Effect of alveolar ridge preservation after tooth extraction: a systematic review and meta-analysis. J Dent Res. 2014;93(10):950-958.
2. Horowitz R, Holtzclaw D, Rosen PS. A review on alveolar ridge preservation following tooth extraction. J Evid Based Dent Pract. 2012;12(3 suppl):149-160.
3. Levine RA, Ganeles J, Kan J, Fava PL. 10 keys for successful esthetic-zone single implants: importance of biotype conversion for lasting success. Compend Contin Educ Dent. 2018;39(8):522-529.
4. Chu SJ, Salama MA, Garber DA, et al. Flapless postextraction socket implant placement, Part 2: the effects of bone grafting and provisional restoration on peri-implant soft tissue height and thickness - a retrospective study. Int J Periodontics Restorative Dent. 2015;35(6):803-809.
5. Horowitz RA, Leventis MD, Rohrer MD, Prasad HS. Bone grafting: history, rationale, and selection of materials and techniques. Compend Contin Educ Dent. 2014;35(4 suppl):1-6.
6. da Rosa JC, de Oliveira Rosa AC, Fadanelli MA, Sotto-Maior BS. Immediate implant placement, reconstruction of compromised sockets, and repair of gingival recession with a triple graft from the maxillary tuberosity: a variation of the immediate dentoalveolar restoration technique. J Prosthet Dent. 2014;112(4):717-722.
7. Horowitz RA, Kurtzman GM. Socket preparation for delayed implant placement using a mineralized cancellous allograft. Compend Contin Educ Dent. 2021;42(4):f1-f4.
8. Solakoglu Ö, Götz W, Heydecke G, Schwarzenbach H. Histological and immunohistochemical comparison of two different allogeneic bone grafting materials for alveolar ridge reconstruction: a prospective randomized trial in humans. Clin Implant Dent Relat Res. 2019;21(5):1002-1016.
9. Chan HL, Lin GH, Fu JH, Wang HL. Alterations in bone quality after socket preservation with grafting materials: a systematic review. Int J Oral Maxillofac Implants. 2013;28(3):710-720.
10. Artzi Z, Tal H, Dayan D. Porous bovine bone mineral in healing of human extraction sockets. Part 1: histomorphometric evaluations at 9 months. J Periodontol. 2000;71(6):1015-1023.
11. Mordenfeld A, Albrektsson T, Hallman M. A 10-year clinical and radiographic study of implants placed after maxillary sinus floor augmentation with an 80:20 mixture of deproteinized bovine bone and autogenous bone. Clin Implant Dent Relat Res. 2014;16(3):435-446.
12. Ayna M, Açil Y, Gulses A. Fate of a bovine-derived xenograft in maxillary sinus floor elevation after 14 years: histologic and radiologic analysis. Int J Periodontics Restorative Dent. 2015;35(4):541-547.
13. Traini T, Piattelli A, Caputi S, et al. Regeneration of human bone using different bone substitute biomaterials. Clin Implant Dent Relat Res. 2015;17(1):150-162.
14. Rodriguez AE, Nowzari H. The long-term risks and complications of bovine-derived xenografts: a case series. J Indian Soc Periodontol. 2019;23(5):487-492.
15. Bannister SR, Powell CA. Foreign body reaction to anorganic bovine bone and autogenous bone with platelet-rich plasma in guided bone regeneration. J Periodontol. 2008;79(6):1116-1120.
16. Bartols A, Kasprzyk S, Walther W, Korsch M. Lateral alveolar ridge augmentation with autogenous block grafts fixed at a distance versus resorbable poly-d-l-lactide foil fixed at a distance: a single-blind, randomized, controlled trial. Clin Oral Implants Res. 2018;29(8):843-854.
17. Froum SJ, Tarnow DP, Wallace SS, et al. The use of a mineralized allograft for sinus augmentation: an interim histological case report from a prospective clinical study. Compend Contin Educ Dent. 2005;26(4):259-268.
18. Kuperschlag A, Keršytė G, Kurtzman GM, Horowitz RA. Autogenous dentin grafting of osseous defects distal to mandibular second molars after extraction of impacted third molars. Compend Contin Educ Dent. 2020;41(2):76-82.
19. Ku JK, Jeong YK, Um IW. Review of allogeneic dentin graft for maxillofacial bone defects. Tissue Eng Part C Methods. 2021;27(8):472-480.
20. Zhang S, Li X, Qi Y, et al. Comparison of autogenous tooth materials and other bone grafts. Tissue Eng Regen Med. 2021;18(3):327-341.
21. Um IW, Lee JK, Kim JY, et al. Allogeneic dentin graft: a review on its osteoinductivity and antigenicity. Materials (Basel). 2021;14(7):1713.
22. Flanagan D. Autogenous dentin with calcium sulfate as graft material: a case series. J Oral Implantol. 2021. doi: 10.1563/aaid-joi-D-20-00309.
23. Yüceer-Çetiner E, Özkan N, Önger ME. Effect of autogenous dentin graft on new bone formation. J Craniofac Surg. 2021;32(4):1354-1360.
24. Kızıldağ A, Tasdemir U, Arabacı T, et al. Effects of autogenous tooth bone graft and platelet-rich fibrin in peri-implant defects: an experimental study in an animal model. J Oral Implantol. 2020;46(3):221-226.
25. Umebayashi M, Ohba S, Kurogi T, et al. Full regeneration of maxillary alveolar bone using autogenous partially demineralized dentin matrix and particulate cancellous bone and marrow for implant-supported full arch rehabilitation. J Oral Implantol. 2020;46(2):122-127.
26. Elfana A, El-Kholy S, Saleh HA, El-Sayed KF. Alveolar ridge preservation using autogenous whole-tooth versus demineralized dentin grafts: a randomized controlled clinical trial. Clin Oral Implants Res. 2021;32(5):539-548.
27. Um IW, Kim YK, Mitsugi M. Demineralized dentin matrix scaffolds for alveolar bone engineering. J Indian Prosthodont Soc. 2017;17(2):120-127.
28. Binderman I, Hallel G, Nardy C, et al. A novel procedure to process extracted teeth for immediate grafting of autogenous dentin. J Interdisciplinary Medicine and Dental Science. 2014;2:1-5.
29. Stahl SS, Froum SJ. Human clinical and histologic repair responses following the use of citric acid in periodontal therapy. J Periodontol. 1977;48(5):261-266.
30. Inchingolo AD, Inchingolo AM, Bordea IR, et al. The effectiveness of osseodensification drilling protocol for implant site osteotomy: a systematic review of the literature and meta-analysis. Materials (Basel). 2021;14(5):1147.
31. Lahens B, Neiva R, Tovar N, et al. Biomechanical and histologic basis of osseodensification drilling for endosteal implant placement in low density bone. An experimental study in sheep. J Mech Behav Biomed Mater. 2016;63:56-65.
32. Suarez F, Monje A, Galindo-Moreno P, Wang HL. Implant surface detoxification: a comprehensive review. Implant Dent. 2013;22(5):465-473.
33. Schwarz F, Golubovic V, Becker K, Mihatovic I. Extracted tooth roots used for lateral alveolar ridge augmentation: a proof-of-concept study. J Clin Periodontol. 2016;43(4):345-353.
34. Schwarz F, Hazar D, Becker K, et al. Efficacy of autogenous tooth roots for lateral alveolar ridge augmentation and staged implant placement. A prospective controlled clinical study. J Clin Periodontol. 2018;45(8):996-1004.
35. Dentistry Journal Editorial Office. The use of tooth particles as a biomaterial in post-extraction sockets. Experimental Study in Dogs. [retraction of Calvo-Guirado JL, Maté-Sánchez de Val JE, Ramos-Oltra ML, et al. In: Dent J (Basel). 2018;6(2):12. doi: 10.3390/dj6020012]. Dent J (Basel). 2020;8(3):92. doi: 10.3390/dj8030092.
36. Del Canto-Díaz A, de Elío-Oliveros J, Del Canto-Díaz M, et al. Use of autologous tooth-derived graft material in the post-extraction dental socket. Pilot study. Med Oral Patol Oral Cir Bucal. 2019;24(1):e53-e60.
37. Netaneli E, Artzi Z, Renert U, Mimon-Gil T. The particulated dentin - an autogenous reliable bone graft substitute in socket site preservation. Clin Oral Implants Res. 2019;30(S19):259.
38. Covani U, Ricci M, Bozzolo G, et al. Analysis of the pattern of the alveolar ridge remodelling following single tooth extraction. Clin Oral Implants Res. 2011;22(8):820-825.
39. Binderman I, Duda M, Hallel G, Horowitz R. The use of autogenous dentin particulate graft for alveolar ridge preservation and augmentation following tooth extraction. In: Miron RJ, Zhang Y, eds. Next-Generation Biomaterials for Bone & Periodontal Regeneration. Batavia, IL: Quintessence Publishing; 2019:chap 8, 109-119.
40. Schropp L, Wenzel A, Kostopoulos L, Karring T. Bone healing and soft tissue contour changes following single-tooth extraction: a clinical and radiographic 12-month prospective study. Int J Periodontics Restorative Dent. 2003;23(4):313-323.
41. Araújo MG, Silva CO, Misawa M, Sukekava F. Alveolar socket healing: what can we learn? Periodontol 2000. 2015;68(1):122-134.
42. Menchini-Fabris GB, Toti P, Crespi G, et al. Effect of different timings of implant insertion on the bone remodeling volume around patients' maxillary single implants: a 2-3 years follow-up. Int J Environ Res Public Health. 2020;17(18):6790.
43. Botticelli D, Berglundh T, Lindhe J. Hard-tissue alterations following immediate implant placement in extraction sites. J Clin Periodontol. 2004;31(10):820-828.
44. Jalaluddin M, Sathe S, Thomas J, et al. Assessment of implant stability in immediate implant placement using different bone grafting materials: a clinical study. J Pharm Bioallied Sci. 2021;13(suppl 1):S612-S615.
45. Goldberg M, Kulkarni AB, Young M, Boskey A. Dentin: structure, composition and mineralization. Front Biosci (Elite Ed). 2011;3(2):711-735.
46. Lee SL, Veis A, Glonek T. Dentin phosphoprotein: an extracellular calcium-binding protein. Biochemistry. 1977;16(13):2971-2979.
47. Schmidt-Schultz TH, Schultz M. Bone protects proteins over thousands of years: extraction, analysis, and interpretation of extracellular matrix proteins in archeological skeletal remains. Am J Phys Anthropol. 2004;123(1):30-39.
48. Kim YK, Kim SG, Yun PY, et al. Autogenous teeth used for bone grafting: a comparison with traditional grafting materials. Oral Surg Oral Med Oral Pathol Oral Radiol. 2014;117(1):e39-e45.
49. Dhuvad JM, Mehta D. Does an autogenous demineralized dentin (ADDM) graft has the ability to form a new bone? Natl J Maxillofac Surg. 2021;12(2):181-187.
50. Silva AC, Gomes Moura CC, Ferreira JA, et al. Biological effects of a root conditioning treatment on periodontally affected teeth-an in vitro analysis. Braz Dent J. 2016;27(2):160-168.
51. Binderman I, Halperin-Sternfeld M, Netanely E, et al. Biomaterials selection - cellular interactions during regenerative and remodeling phases. In: Artzi Z, ed. Bone Augmentation by Anatomical Region: Techniques and Decision-Making. Wiley-Blackwell; 2020:43-59.
52. Di Girolamo M, Barlattani A Jr, Grazzini F, et al. Healing of the post extractive socket: technique for conservation of alveolar crest by a coronal seal. J Biol Regul Homeost Agents. 2019;33(6 suppl 1):125-135.
53. Horowitz RA. Preservation of alveolar dimensions at the time of tooth extraction. In: Karateew ED, ed. Implant Aesthetics: Keys to Diagnosis and Treatment. Cham, Switzerland: Springer International Publishing AG; 2017:chap 11, 171-188.
54. Mohan R, Bajaj A, Gundappa M. Human amnion membrane: potential applications in oral and periodontal field. J Int Soc Prev Community Dent. 2017;7(1):15-21.
55. Horowitz RA. Extraction environment enhancement: critical evaluation of early socket healing in long-term barrier-protected extraction sockets [erratum appears in Compend Contin Educ Dent. 2005;26(11):765]. Compend Contin Educ Dent. 2005;26(10):703-713.
56. Niknejad H, Peirovi H, Jorjani M, et al. Properties of the amniotic membrane for potential use in tissue engineering. Eur Cell Mater. 2008;
15:88-99.
57. Palanker ND, Lee CT, Weltman RL, et al.Antimicrobial efficacy assessment of human derived composite amnion-chorion membrane. Sci Rep. 2019;9(1):15600.
58. Ashraf H, Font K, Powell C, Schurr M. Antimicrobial activity of an amnion-chorion membrane to oral microbes. Int J Dent. 2019;2019:
1269534.
59. Holtzclaw D, Toscano N. Amnion chorion allograft barrier: indications and techniques update. J Imp Adv Clin Dent. 2012;4(2):25-38.
60. Holtzclaw DJ, Toscano NJ. Amnion-chorion allograft barrier used for guided tissue regeneration treatment of periodontal intrabony defects: a retrospective observational report. Clin Adv Perio. 2013;3(3):131-137.