Circumferential Occlusal Access Procedure (COAP): Novel Minimally Invasive Surgical Approach for Treatment of Peri-Implantitis—A Case Series
Paul Fletcher, DDS; and Dennis P. Tarnow, DDS
Though the use of dental implants has become a highly predictable treatment modality, as the number of implants placed annually rises and the number of years implants have been in place increases, problems are becoming evident.1,2 The number of cases of peri-implantitis, defined as a destructive inflammatory reaction affecting the soft and hard tissues around an implant in function, 3 is rising.
While peri-implantitis is multifactorial,4 the migration of bacterial biofilm down the surface of the implant and the inflammatory response that follows is thought to be a primary cause of the problem.5 Treatment of inflammatory peri-implantitis is directed toward reducing the bacterial load to a sub-inflammatory concentration by removing the biofilm from the implant surface and the associated bony defect.6,7
Peri-implant mucositis, defined as an inflammatory reaction in the soft tissue surrounding a functioning implant,3 can be successfully treated nonsurgically through the space between the soft tissue and the head of the implant.7 With peri-implantitis the nonsurgical decontamination of the implant and associated osseous defect is unpredictable due to the macro-anatomy of the implant threads and micro-anatomy of the implant surface, as well as the contour of the restoration, the limited access for an instrument to clean around the implant, and lack of direct visibility.7 Accordingly, open-flap surgical debridement followed by detoxification of the implant surface in conjunction with osseous recontouring and/or guided bone regeneration (GBR) is the recommended treatment.7-10
This approach, however, has drawbacks. The soft tissue around an implant may be thin, friable, and easily perforated, or bound down and difficult to reflect. Buccal and lingual flap elevation often extends to adjacent healthy teeth and implants and can lead to flap dehiscence and soft-tissue recession.11,12 In addition to the potential for cosmetic problems in the esthetic zone, these sequelae, along with soft-tissue fistulas and membrane exposure, can lead to contamination and loss of the bone graft.13-15
This article presents a procedure that enables access to an implant for surface decontamination and facilitates bone repair or regeneration while reducing the potential for membrane exposure and recession. This novel, minimally invasive surgical approach, called the circumferential occlusal access procedure (COAP), is designed for the treatment of peri-implantitis.
Overview of Circumferential Occlusal Access Procedure
COAP has two phases: a decontamination phase, which is used in cases of incipient peri-implantitis and around implants that have not responded to conventional treatments for mucositis; and a regenerative phase, which is used in addition to the decontamination phase in more advanced situations where bone grafting is necessary. COAP is indicated for use with a retrievable implant restoration.
In the decontamination phase of treatment the restoration and its abutment are removed, exposing the implant-abutment connection. A collar of soft tissue adjacent to the affected implant surface is then removed using a large curette by moving it circularly around the implant down to the base of the bone, or by using a scalpel to circumferentially resect the tissue and then remove it. Once the inflamed tissue has been removed the contaminated supracrestal section of the implant and the associated bony defect, along with the underside of the implant restoration, become accessible for thorough mechanical and chemical debridement and detoxification through the gap that's been created. In cases of incipient peri-implantitis the restoration is replaced and finger pressure is applied to the soft tissue-implant restoration interface to optimize the potential for soft-tissue readherence.
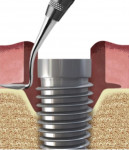
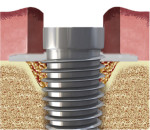
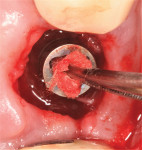
If bone grafting is indicated, the second phase of treatment is incorporated into the procedure. An interproximal knife is used to lift the inner aspect of the soft tissue surrounding the implant off the crest of bone creating a circumferential pouch around the defect (Figure 1). Bone substitute is placed in the defect and a sized collagen membrane with a punch hole in the center is placed over the head of the implant and manipulated apically to cover the bone graft material. To exclude epithelium the edges of the membrane are then tucked into the space that has been created between the crest of bone and the internal aspect of the soft tissue surrounding the peri-implant defect (Figure 2), and the restoration is replaced. Sutures are usually not required.
The authors present a series of cases. Case 1 illustrates the management of an incipient peri-implantitis lesion, and Cases 2 and 3 demonstrate the use of COAP with more complex defects.
Case 1
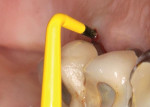
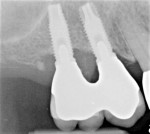
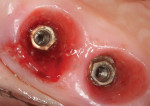
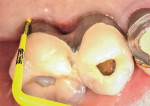
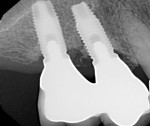
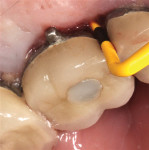
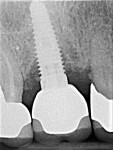
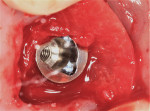
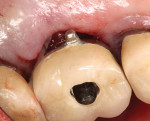
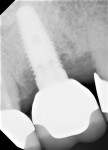
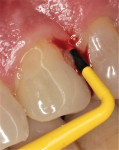
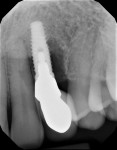
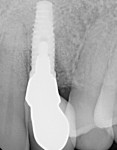
A 56-year-old female patient had implants placed in the upper right first molar and second bicuspid positions. She returned for periodic maintenance visits at 3- to 6-month intervals. Bone levels remained at the first thread and probing depths were 3 mm to 4 mm for 10 years. At a 12-year maintenance visit, 8 mm to 9 mm pocketing and bleeding on probing (BOP) were evident around the upper right first molar implant restoration (Figure 3), and a radiograph showed bone loss to the third implant thread (Figure 4).
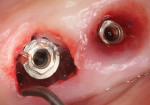
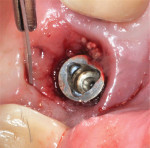
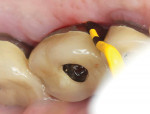
After providing informed consent, the patient was anesthetized and the two splinted crowns were removed. The soft tissue around the first molar implant was spongy and inflamed and had a gelatinous consistency (Figure 5). This tissue was easily removed by using a large, sharp Prichard curette (Hu-Friedy, hu-friedy.com) in a circular motion. The same instrument was used to debride the granulomatous tissue in the bony defect. This created a gap between the soft-tissue margin and implant and exposed the implant platform screw-access opening and supra- and subosseous threads, making them accessible for both mechanical and chemical decontamination (Figure 6).
The threads of the implant were decontaminated mechanically with the use of a titanium curette (Figure 6). The implant surface, implant connection, and underside of the restoration were detoxified chemically with firm burnishing with a 1.5% solution of hydrogen peroxide on a cotton pellet, formed by mixing equal amounts of 3% hydrogen peroxide and sterile water, on all surfaces for 1 minute. The same protocol was repeated using sterile saline, and the entire defect was thoroughly irrigated with sterile saline from an irrigating syringe. The goal was to disrupt, detoxify, and remove the subgingival biofilm, diluting it to a sub-inflammatory concentration.
The implant restoration was replaced. Finger pressure was applied for 1 minute at the point where the gingival margin met the restoration or implant abutment to maximize the chances for connective tissue readherence and to minimize epithelial downgrowth. Amoxicillin 250 mg and metronidazole 250 mg were prescribed, three times per day for 8 days.16 The patient was advised to rinse with an essential oil mouthwash (Listerine®, Johnson & Johnson, listerine.com) twice per day for 30 seconds for 2 weeks, but not to brush the area.
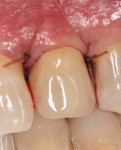
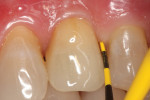
The patient was seen 2, 4, and 8 weeks postoperatively and then at periodic 3-month maintenance visits. A 28-month post-treatment follow-up showed pocket depths reduced from 8 mm to 3 mm with no BOP (Figure 7), and though the preoperative and postoperative radiographs were not standardized there appeared to be evidence of bone fill (Figure 8).
Case 2
An 85-year-old female patient presented with BOP and 6 mm to 9 mm pocketing around an implant-supported screw-retained restoration placed 3 years prior in the upper right first molar position (Figure 9). A radiograph showed vertical bone loss on both the mesial and distal aspects (Figure 10). The patient was advised it would be necessary to remove the restoration to detoxify the implant surface and to attempt to regrow the bone that had been lost.
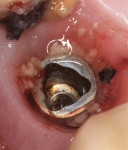
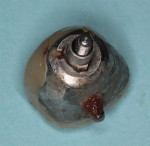
After the patient provided informed consent she was anesthetized and the crown was removed. Heavy accumulations of plaque and debris were evident on both the internal connection of the implant (Figure 11) and the underside of the restoration (Figure 12). After the gross debris was removed, an internal circumferential incision with a #15C scalpel was made to remove the diseased fibrous tissue adjacent to the implant (Figure 13) and to provide adequate access for mechanical and chemical decontamination. Plastic and titanium curettes were then used to mechanically decontaminate the smooth collar and threads of the implant and to thoroughly debride the associated infrabony defect.
The internal connection and threads were then chemically detoxified by first burnishing the surfaces for 1 minute with a cotton pellet saturated in a 0.25% solution of sodium hypochlorite, which was formed by mixing 5 ml (one teaspoon) of 5% to 6% household bleach with 125 ml (4 oz.) of sterile water (Figure 14). This was followed by burnishing with a 1.5% hydrogen peroxide solution using the same protocol, and then irrigating with copious amounts of sterile saline. The underside of the restoration (Figure 15) and screw were similarly cleaned.
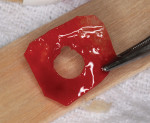
An interproximal knife was inserted into the occlusal access opening and placed between the soft tissue and crest of bone to separate and lift the internal surface of the soft tissue from the bone surrounding the defect as shown in Figure 1, similar to the technique used during a mucogingival tunneling procedure.17 Small increments of bovine bone (Bio-Oss®, Geistlich Biomaterials, geistlich-na.com) were placed within the confines of the osseous defect. A porcine collagen membrane (DynaMatrix®, Keystone Dental, keystonedental.com) with a 4-mm diameter opening in the center was created using a 4-mm diameter tissue punch (Figure 16). The collagen membrane was sized and placed over the head of the implant and manipulated so it covered the bone graft material (Figure 17). The edges of the membrane were tucked into the circumferential space that had been created between the soft tissue and bone surrounding the peri-implant defect, as shown in Figure 2.
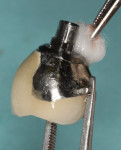
The restoration was replaced and pressure was applied at the gingival margin for 1 minute. No sutures were necessary (Figure 18). The crown removal button was removed. Amoxicillin 250 mg and metronidazole 250 mg were prescribed, three times per day for 8 days, and the patient was told not to brush the area for 2 weeks but to rinse with an essential oil mouthwash twice per day for 30 seconds. The patient was seen postoperatively at 2, 4, and 8 weeks and then at periodic 3-month maintenance visits. At the 8-week follow-up visit, it was noted that the implant screw had loosened and the crown had become mobile. Tightening the screw resolved the problem. An 8-month clinical follow-up showed 6 mm residual buccal pocketing and 5 mm mesio-palatal pocketing (Figure 19) but no BOP, and bone graft material was still evident radiographically (Figure 20).
Case 3
A 57-year-old male patient presented with 9 mm pocketing (Figure 21) and advanced radiographic bone loss (Figure 22) around an implant that had been placed in the maxillary left cuspid position 7 years prior. After the patient provided informed consent, he was anesthetized, the crown was removed, and the inflamed crevicular tissue and debris in the internal connection were visualized.
A circumferential incision with a #15C scalpel was made to remove the inflamed tissue; this exposed the implant surface as well as a deep, wide infrabony peri-implant defect on the distal and palatal aspects. The implant threads and the defect were mechanically debrided with a titanium curette, and the implant surface and underside of the restoration were chemically detoxified by burnishing with a 0.25% sodium hypochlorite solution on a cotton pellet for 1 minute followed by burnishing with solutions of 1.5% hydrogen peroxide and sterile saline for similar amounts of time. The bony defect and implant were then irrigated with copious amounts of sterile saline.
The defect was filled with a 50:50 mix of freeze-dried cortico-cancellous bone (Puros®, Zimmer Biomet, zimmerbiomet.com) and bovine bone and, similar to Case 2, covered with a collagen membrane with a 4-mm diameter punch hole in the center that was slipped over the head of the implant. The edges of the membrane were tucked into a circumferential space between the bone and soft tissue that had been created with an interproximal knife (Figure 2). The restoration was reinserted, two interrupted sutures were placed (Figure 23), and pressure was applied for 1 minute at the gingival margin. The patient was placed on amoxicillin 250 mg and metronidazole 250 mg, three times per day for 8 days, and was told not to brush the area for 2 weeks but to rinse with an essential oil mouthwash twice per day for 30 seconds.
The patient was seen 2, 4, and 8 weeks postoperatively and then for 3-month maintenance visits. At a 14-month follow-up visit, the upper left cuspid probed 3 mm (Figure 24), and though the preoperative and postoperative radiographs were not standardized bone fill appeared to be evident radiographically (Figure 25).
Discussion
Treating peri-implantitis nonsurgically is unpredictable, as mechanical debridement alone has been shown to be inadequate.7 Flap elevation followed by open debridement in conjunction with mechanical and chemical decontamination, GBR, and superior patient home care optimizes the chances of reforming lost bone, reducing pocket depths, and arresting peri-implantitis.7-12 Full-thickness flap elevation can be problematic, however. Primary approximation of the flap can be challenging. Flap dehiscence, leading to soft-tissue recession and membrane and graft exposure, may occur13-15 and result in a compromised cosmetic or regenerative outcome.
COAP is a flapless, minimally invasive surgical technique that provides access to the contaminated implant surface, bony defect, and all of the components of the implant restoration. A retrievable restoration is a prerequisite for using COAP. After the implant crown and its abutment have been removed, the implant-abutment junction can be visualized. Studies show bacteria can migrate in and out of this connection.18,19 This is illustrated in Figure 5, Figure 11, and Figure 12; these images demonstrate the value of utilizing a screw-retained or retrievable cemented restoration.
After the inflamed soft tissue adjacent to the implant has been removed, the threads, screw-access opening, and implant connection become accessible for mechanical and chemical detoxification. In addition, the underside of the restoration (Figure 15), the abutment, and the implant screw (Figure 14) become accessible for thorough decontamination.
In cases of incipient peri-implantitis (eg, Case 1) the inflamed tissue often has a gelatinous consistency and can be scooped out using a sharp curette in a circular motion. This approach is also indicated in situations where the soft tissue surrounding the implant is thin or where a minimal zone of keratinized tissue exists. With more chronic, advanced lesions (eg, Case 2 and Case 3) the tissue abutting the implant tends to be dense and fibrous and requires a circumferential incision with a scalpel to remove it.
Numerous mechanical and chemical approaches have been presented for decontaminating an implant surface.20-22 The present authors used slightly varied protocols in each case. Titanium and plastic curettes were used for mechanical debridement, while sodium hypochlorite, hydrogen peroxide, and sterile saline were successfully used for chemical decontamination.Sodium hypochlorite is a highly cytotoxic antiseptic and oxidizing agent that kills bacteria by denaturing its organic component. Found naturally in macrophages and neutrophils, sodium hypochlorite does not corrode titanium and shows no visible effect on periodontal healing.16 While hydrogen peroxide duplicates the oxidative effect of sodium hypochlorite, in vitroresearch indicates it can enhance fibrinogen absorption, growth of the oxide layer, and deposition of calcium on the implant surface.23 Meanwhile, sterile saline has been shown in animal research to detoxify an implant surface to an extent that re-osseointegration occurred.24 The potential effectiveness of this low-cost decontamination protocol is illustrated by the elimination of BOP and the reduction in pocketing in the cases presented.
When GBR was indicated the technique illustrated in Figure 1 and Figure 2 was used to graft the bony lesion. In Case 2 bovine bone was employed as a regenerative material, and in Case 3 it was used in combination with freeze-dried cortico-cancellous bone. It should be noted that to prevent overfill of the infrabony defect, bone graft material should be placed with care in small increments within the confines of the walls of the defect. A porcine collagen membrane was utilized as a barrier in both Case 2 and Case 3. The use of a punch to create a circular opening in a tissue graft or membrane has been used for years25 and has applications in both soft- and hard-tissue grafting.
A recent human histologic case study explained the rationale for the above protocol and demonstrated this sequence of low-cost mechanical and chemical decontamination in conjunction with similar GBR materials to be successful in achieving new direct bone-to-implant contact around an implant previously affected by peri-implantitis.26
The decontaminated components of the implant restoration are replaced after grafting. The exposed occlusal portion of the membrane is covered and protected from contamination and premature resorption by the underside of the restoration as it is seated back into the implant connection (Figure 18). To optimize the chances for connective tissue readherence while limiting epithelial downgrowth finger pressure is applied for 1 minute at the point where the soft-tissue margin meets the restoration or abutment in an attempt to establish a stable blood clot. Sutures are usually not required, though in Case 3 two interrupted sutures were placed to better adapt the interproximal tissue. Amoxicillin and metronidazole are prescribed to eliminate any residual bacteria that may have permeated into the soft tissues surrounding the implant.27
COAP is designed to reduce the chances of incomplete wound closure and soft-tissue dehiscences. While soft-tissue contraction may occur, especially if the patient has a thin biotype, recession is not as likely because the circulation between the bone and soft tissue is not disrupted. The soft tissue surrounding the implant will remain nourished by the underlying bone, and the supracrestal connective tissue fibers associated with an adjacent tooth or implant will continue to support the soft-tissue height. This is an important advantage versus a full-thickness flap, especially in the esthetic zone.
While 8 mm to 9 mm pockets can be reduced to 2 mm to 3 mm as shown in Case 1 and Case 3, and radiographic evidence of bone fill without osseous grafting can be achieved as appeared to be demonstrated in Case 1, these are not routine occurrences. More common is the situation in Case 2 where 4 mm to 5 mm pocketing without BOP may remain, especially in instances where there is thick tissue in the maxillary molar region. While the residual pocketing and loss of some of the bone graft in Case 2 may have been compounded by the mobility of the restoration when the implant screw loosened, research shows that following open debridement, implants with moderate residual pocketing can be maintained with superior patient home care and periodic maintenance visits.28,29
COAP was initially used in patients with peri-implant bone loss in the esthetic zone,30 as in Case 3, where flap elevation might have led to significant recession. The procedure was subsequently utilized in the treatment of incipient peri-implantitis, as in Case 1, where it would have been difficult to elevate a full-thickness flap without involving healthy adjacent implants or teeth. The potential of COAP was more fully realized once it became apparent that GBR could also be accomplished through a minimally invasive occlusal access opening, as in Case 2 and Case 3.
This procedure has limitations in that the clinician must be able to access the entire implant surface and bony lesion for thorough debridement and decontamination. Narrow ridges are not conducive to removing a circumferential collar of soft tissue, and confined infrabony defects are more easily managed than wide osseous defects or adjacent peri-implantitis defects where open-flap debridement might provide better accessibility.
Conclusions
COAP is a novel approach for the treatment of peri-implantitis. It is flapless, minimally invasive, and often sutureless, yielding minimal postoperative pain, swelling, and associated morbidity. With COAP, all contaminated aspects of the implant, restoration, and bony defect become accessible for debridement and detoxification. The procedure can be used successfully in both grafting and nongrafting situations. While the decontamination phase alone is sufficient for the management of early peri-implantitis lesions, the addition of the regenerative phase is a useful treatment option when bone grafting is indicated. Additional cases with long-term follow-up are needed to determine the full potential of this procedure.
About the Authors
Paul Fletcher, DDS
Associate Clinical Professor, Division of Periodontics, Columbia University College of Dental Medicine, New York, New York; Private Practice, New York, New York
Dennis P. Tarnow, DDS
Clinical Professor, Director of Implant Education, Division of Periodontics, Columbia University College of Dental Medicine, New York, New York; Private Practice, New York, New York
References
1. Daubert DM, Weinstein BF, Bordin S, et al. Prevalence and predictive factors for peri-implant disease and implant failure: cross-sectional analysis. J Periodontol. 2015;86(3):337-347.
2. Derks J, Schaller D, Håkansson J, et al. Effectiveness of implant therapy analyzed in a Swedish population: prevalence of peri-implantitis. J Dent Res. 2016;95(1):43-49.
3. Zitzmann NU, Berglundh T. Definition and prevalence of peri-implant diseases. J Clin Periodontol. 2008;35(suppl 8):286-291.
4. Albrektsson T, Buser D, Sennerby L. Crestal bone loss and oral implants. Clin Implant Dent Relat Res. 2012;14(6):783-791.
5. Berglundh T, Zitzmann NU, Donati M. Are peri-implantitis lesions different from periodontitis lesions? J Clin Periodontol. 2011;38(suppl 11):188-202.
6. Mombelli A. Microbiology and antimicrobial therapy of peri-implantitis. Periodontol 2000. 2002;28:177-189.
7. Renvert S, Roos-Jansåker A, Claffey N. Non-surgical treatment of peri-implant mucositis and peri-implantitis: a literature review. J Clin Periodontol. 2008;35(suppl 8):305-315.
8. Claffey N, Clarke E, Polyzois I, Renvert S. Surgical treatment of peri-implantitis. J Clin Periodontol. 2008;35(suppl 8):316-332.
9. Schwarz F, Jepsen S, Herten M, et al. Influence of different treatment approaches on non-submerged and submerged healing of ligature induced peri-implantitis lesions: an experimental study in dogs. J Clin Periodontol. 2006;33(8):584-595.
10. Mombelli A, Moëne R, Décaillet F. Surgical treatments of peri-implantitis. Eur J Oral Implantol. 2012;5(suppl):S61-S70.
11. Heitz-Mayfield LJA, Salvi GE, Mombelli A, et al. Anti-infective surgical therapy of peri-implantitis: a 12-month prospective clinical study. Clin Oral Impl Res. 2012;23(2):205-210.
12. Romeo E, Ghisolfi M, Murgolo N, et al. Therapy of peri-implantitis with resective surgery. A 3-year clinical trial on rough screw-shaped oral implants. Part I: clinical outcome. Clin Oral Implants Res. 2005;16(1):9-18.
13. Khoury F, Buchmann R. Surgical therapy of peri-implant disease: a 3-year follow-up study of cases treated with 3 different techniques of bone regeneration. J Periodontol. 2001;72(11):1498-1508.
14. Aughtun M, Richter EJ, Hauptmann S, Yildrium M. Studies on the treatment of deep peri-implant bone pockets with E-PTFE membranes. J Dent Imp. 1992;8:246-250.
15. Jovanovic SA, Spiekermann H, Richter EJ, Koseoglu M. Guided tissue regeneration around dental implants. Integration in oral, orthopedic and maxillofacial reconstruction. In: Laney WR, Tolman DE, eds. Proceedings of the Second International Congress of Tissue Integration in Oral Orthopedic and Maxillofacial Reconstruction. Chicago, IL: Quintessence Publishing; 1992.
16. Slots J. Low-cost periodontal therapy. Periodontol 2000. 2012;60(1):110-137.
17. Allen AL. Use of the supraperiosteal envelope in soft tissue grafting for root coverage. I. rationale and technique. Int J Periodontics Restorative Dent.1994;14(3):216-227.
18. Teixeira W, Ribeiro RF, Sato S, Pedrazzi V. Microleakage into and from two-stage implants: an in vitro comparative study. Int J Oral Maxillofac Implants. 2011;26(1):56-62.
19. Assenza B, Tripodi D, Scarano A, et al. Bacterial leakage in implants with different implant-abutment connections: an in vitro study. J Periodontol. 2012;83(4):491-497.
20. Schmage P, Thielemann J, Nergiz I, et al. Effects of 10 cleaning instruments on four different implant surfaces. Int J Oral Maxillofac Implants. 2012;27(2):308-317.
21. Zablotsky MH, Diedrich DL, Meffert RM. Detoxification of endotoxin-contaminated titanium and hydroxyapatite-coated surfaces utilizing various chemotherapeutic and mechanical modalities. Implant Dent. 1992;1(2):154-158.
22. Gosau M, Hahnel S, Schwarz F, et al. Effect of six different peri-implantitis disinfection methods on in vivo human oral biofilm. Clin Oral Implants Res. 2010;21(8):866-872.
23. Wälivaara B, Lundström I, Tengvall P. An in-vitro study of H2O2-treated titanium surfaces in contact with blood plasma and a simulated body fluid. Clin Mater. 1993;12(3):141-148.
24. Alhag M, Renvert S, Polyzois I, Claffey N. Re-osseointegration on rough implant surfaces previously coated with bacterial biofilm: an experimental study in the dog. Clin Oral Implants Res. 2008;19(2):182-187.
25. Tarnow D. Periodontal concepts related to osseointegrated implants. Paper presented at: Academy of Osseointegration Annual Meeting; 1987; Chicago, IL.
26. Fletcher P, Deluiz D, Tinoco EM, et al. Human histologic evidence of reosseointegration around an implant affected with peri-implantitis following decontamination with sterile saline and antiseptics: a case history report. Int J Periodontics Restorative Dent. 2017;37(4):499-508.
27. Leonhardt A, Dahlén G, Renvert S. Five-year clinical, microbiological, and radiological outcome following treatment of peri-implantitis in man. J Periodontol. 2003;74(10):1415-1422.
28. Salvi GE, Zitzmann NU. The effects of anti-infective preventive measures on the occurrence of biologic implant complications and implant loss: a systematic review. Int J Oral Maxillofac Implants. 2014;29(suppl):292-307.
29. Serino G, Turri A, Lang NP. Maintenance therapy in patients following the surgical treatment of peri-implantitis: a 5-year follow-up study. Clin Oral Implants Res. 2015;26(8):950-956.
30. Constantinides C, Chang J, Fletcher P. Management of an ailing anterior implant using a minimally invasive flapless surgical technique: a case report. Clin Adv Periodontics. 2017;7(4):201-206.