Comprehensive Oral Rehabilitation of a Patient with Aplastic Anemia by Periodontal and Prosthesis Treatments
Somayeh Ansari Moghadam, MSD; Sharie Faghihi, MSD; Reza Amid, MSD; Mahdi Kadkhodazadeh, MSD; and Shabnam Khaje Hosseini, MSD
Abstract
Aplastic anemia (AA) is a rare but extremely serious disorder that results from an unexplained failure of the bone marrow to produce blood cells. Patients with AA are at high risk for infections, including periodontitis. Treatment modalities are complicated by bleeding disorders. This is a case report of the successful periodontal and prosthesis treatment in a patient with AA, chronic diffuse gingivitis, excessive tooth caries that interfered with mastication, painful oral lesions, and limited mouth opening. Physiotherapy was performed to improve the mouth opening, and the patient was examined for HBsAg, HBsAb, and HIV; white blood cell and platelet count and coagulation tests were given before every periodontal treatment. Caries removal and root canal therapy were done, hopeless teeth were extracted, and full-mouth crown lengthening was performed. Free gingival grafts were placed in areas of inadequate keratinized tissue, and full-mouth rehabilitation with fixed prosthesis was completed. RESULTS: The mouth opening was improved to 31 mm, inflammation was significantly controlled especially after comprehensive periodontal treatment, the redness significantly decreased, and an acceptable condition for prosthesis reconstruction was obtained. DISCUSSION: The treatment modalities used in this case provide a new ray of hope for patients who are severely medically compromised.
Aplastic anemia (AA) is a rare disorder with the potential for serious complications. Failure of the bone marrow to produce blood cells is considered the main problem.1 The disorder can be inherited, idiopathic, or acquired. One of the well-known causal factors is the autoimmune disorder, in which the white blood cells attack bone marrow.2 Autoimmune diseases arise from an overactive immune response of the body against substances and tissues normally present in the body. In other words, the body actually attacks its own cells. The immune system mistakes some part of the body as a pathogen and attacks it. The current understanding about the exact etiology is limited in many cases. However, present data indicate an association between the disorder and exposure to substances such as benzene, radiation, or the use of certain drugs such as phenytoin.3
Severe AA is diagnosed according to the following criteria:
In contrast to severe cases, moderate aplastic anemia (MAA) is the classification for AA patients with moderately depressed cells—those who have significantly reduced blood counts but not as reduced as that which occurs in severe or very severe anemia. In many cases of MAA, doctors will simply monitor blood counts, which may remain unchanged for many years.
Moderate cases have an acceptable prognosis and their condition can be managed as a self-limited disease without any specific treatment. However, the differentiation between moderate and severe cases is difficult. In other words, there are some kinds of moderate cases that actually represent a transition stage to severe disease.5 Another problem that should be mentioned is a type of hepatitis that is associated with AA and most often affects adolescent and young males.6 The type of hepatitis is non-A, non-B, and most patients are seronegative for hepatitis A, B, and C.7 In some situations, immunosuppressive therapy may be appropriate; however, more complicated procedures such as bone marrow transplantation (BMT) is still the treatment of choice.8 The immunosuppressive approach is used in patients who do not meet the criteria for transplantation. Antithymocyte globulin (ATG) is approved for use in the United States.9 In patients with severe AA, the addition of cyclosporine to ATG has improved survival rates.10 Cyclosporine is a potent immunosuppressive agent used in the prevention of organ transplant rejection and several diseases of autoimmune organs. The exact mechanism of action is not well known. However, it is thought to selectively and reversibly inhibit T helper cells, which play a role in cellular and humoral immune responses.
Cyclosporine A is administered intravenously or orally, and dosages greater than 500 mg daily have been reported to induce gingival overgrowth. The occurrence of enlargement varies from 25% to 70% according to different studies. Children are affected more frequently, and its magnitude appears to be related more to the plasma concentration than to the patient’s periodontal status.11
Neutropenia is caused by the disorder itself, and its treatment leads to an increased susceptibility to infection; thrombocytopenia causes bruising and mucosal bleeding. Both of these complications correspond with sepsis and hemorrhage, the main cause of death in these patients.10,12 The oral cavity is a common site of these complications. Therefore, early diagnosis and treatment of oral lesions are important. A dental team’s insufficient knowledge regarding this condition could increase the risks of morbidity and mortality in patients with AA. Melanin pigmentation on oral mucosa, traumatic lesions, gingival bleeding, dental biofilm and gingival alterations are the main oral manifestations of Aplastic Anemia patients.13
Patients with AA are more susceptible to infection, so they should avoid situations that would put them at risk of infection or bleeding. These activities, including dental treatment, should be postponed until the patient’s white blood cell count rises to a normal level.14 Dentists should consider prescribing antibacterial mouthwash and oral antibiotics before dental procedures. To reduce the risk of uncontrolled bleeding during major dental treatments, the patients should be taking antifibrinolytics. These agents may decrease bleeding, particularly oral mucosal bleeding, in patients with thrombocytopenia by stabilization of thrombi.15
Chronic periodontitis is a severe focal infection and considered a potential risk of systemic infection in patients with AA. Therefore, some clinicians advocate using antibiotic prophylaxis in periodontal treatments in order to reduce the risk of systemic infection. The existence of bleeding disorders, which are common in AA, significantly complicate the treatments of periodontitis.16 The authors present a case report of successful periodontal and prosthesis reconstruction of a patient with AA, diffuse gingivitis, limited mouth opening, extensive caries, and spontaneous gingival bleeding.
Case Presentation
The patient was an 18-year-old man with AA who was diagnosed when he was 6 years old. A BMT was performed at age 14, and the subsequent treatment was cyclosporine (500 mg/day), CellCept® (Genentech, www.gene.com) (750 mg/day), and prednisone (5 mg/day).
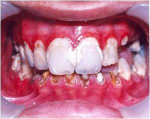
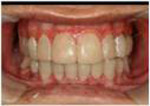
He was referred to the dental school of Shiraz University because of ulcerative lesions produced by severe caries and bleeding after brushing that interfered with oral hygiene (Figure 1). However, no treatment was performed at that time because of the patient’s history with BMT and AA. At the first visit, the neutrophil, platelet, and white blood cell counts were 40,000/µL, 219,000/µL, and 10,500/µL, respectively. The patient was seronegative for hepatitis and HIV infection. However, his serum glutamic-oxaloacetic transaminase (SGOT) and serum glutamic-pyretic transaminase (SGPT) levels were 154 and 164, respectively. The normal ratio of these blood values should be 5 Iu/I to 40 Iu/I for SGOT and 3 Iu/I to 35 Iu/I for SGPT. In this case, because of a drug hepatotoxicity risk (cyclosporin), side effects of bone marrow transplantation (GVHD), and hepatitis risk due to a previous blood transfusion, liver function tests were necessary.
Clinical Examination
A comprehensive clinical examination was performed to determine the number of remaining teeth, probing depth at six points around each tooth, mucogingival junction level, and width of keratinized gingiva for each tooth. A full-mouth periapical radiographic evaluation was performed to determine alveolar bone loss. The diagnosis of generalized diffuse chronic gingivitis modified by systemic disease was made based on the history and clinical findings. Radiographs determined no alveolar bone loss and furcation involvement for rejection of periodontitis.
Due to the limited facial muscles activity as a result of extensive caries and lack of masticatory function, maximum jaw opening was only 15 mm. Therefore, dental therapy was not possible. Also, the bleeding and plaque indices were 100% due to the limited opening.
Based on the clinical and radiographic examinations, the treatment plan was devised to include the following phases:
1. Motivation of the patient for better oral hygiene control
2. Scaling and root planing (SRP) and enhancement of mouth opening via physiotherapy
3. Full-mouth caries removal
4. Extraction of the hopeless molars with furcation involvement, poor access for surgery, and severe caries next to the furcation areas.
5. Root canal therapy for the remaining teeth
6. Full-mouth crown lengthening and free gingival graft for the areas of inadequate keratinized tissue
7. Increase in vertical dimension and full-mouth rehabilitation with fixed prosthesis.
Treatment Phases
First, the authors consulted with the patient’s physician about the treatment plan and the necessity of using a prophylactic antibiotic. Laboratory tests were: platelet, activated partial thromboplastin time, blood count, and white blood cell counts. HBsAg, HBsAb, and HIV screening was also performed. Partial Thromboplastin Time (PTT) can be used to detect the presence of a clotting disorder; Prothrombin Time (PT) was done to evaluate the blood for its ability to clot. This is often done before surgery to evaluate how likely the patient is to have a bleeding or clotting problem during or after surgery. The international normalized ratio (INR) is used to make sure the results from a PT test are the same at one laboratory as they are at another laboratory. The “normal” range of PT for one lab might be different than a “normal” value from another lab, creating problems for patients who were being treated in several locations. In order to standardize the results between labs, the INR was created. As a routine check-up of patients at risk of uncontrolled bleeding the authors check PT, PTT, INR. In aplastic anemia patients, platelet count is disturbed, not platelet function, thus BT screening was not necessary
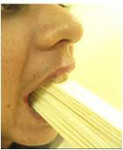
For increasing the mouth opening, the patient was referred to a physiotherapist. After physiotherapy sessions, the mouth opening was increased to 22 mm. Then, the authors recommended using tongue blades and continuing the physiotherapy (Figure 2 and Figure 3).
Tongue blades were placed along occlusal surfaces of the posterior teeth. The vertical opening was increased as an additional blade was added slowly and deliberately to the original stack. The patient was instructed to wait a few minutes between each additional blade placement. By increasing the number of tongue blades daily, the ability of the mouth opening increased slowly to 31 mm, which was sufficient for dental procedures.
Because of several painful intraoral lesions, dry mouth, and extensive caries, the patient was unable to perform dental home care properly. As a result, the plaque index was higher than 80%. After the SRP, extractions, caries removal, and root canal therapies, synthetic saliva products were prescribed to improve salivary quality. Biotene products were prescribed for development of salivary quality. Biotene® is a dental hygiene product manufactured by GlaxoSmithKline (www.gsk.com). It comes in a number of forms, including toothpaste, mouthwash, and cream. In addition to the active ingredient sodium monofluorophosphate in its toothpastes, all the products contain enzymes including glucose oxidase, lactoferrin, lactoperoxidase, and lysozyme. The PBF (plaque biofilm) products also contain the enzymes mutanase and dextranase. Patients with xerostomia (dry mouth) may use Biotene to reduce the rate of recurrence of dental plaque because these products improve saliva quality.17
Fluoride therapy was performed using special trays at least once a day. The patient was instructed to place a sufficient amount of fluoride gel onto the trays for 5 minutes immediately following brushing and to continue this preventive method for life. Also, he was strongly urged to eat foods with less sugar. The authors provided a tray as a carrier that simplified the fluoride application. He was monitored weekly for gingival inflammation. After acceptable inflammation control, surgical procedures were started.
Surgical Procedure
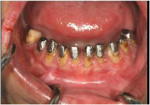
Based on the physician’s recommendations, amoxicillin (2 g, 1 hour before every nonsurgical and surgical procedure) was prescribed. SRP was performed for inflammation control, and then the patient was referred to the endodontist for root canal therapy of all the teeth that were included in the treatment plan. At first, the authors consulted with hematologists. There was no need for antifibrinolytic medications, because platelet and WBC count were normal before surgery. After caries removal, SRP was repeated, and oral hygiene methods were reviewed weekly with the patient. The patient was monitored for inflammation control. Three months later, when acceptable inflammation control was achieved, an apically positioned flap was done to lengthen the crowns in six sessions as a full-mouth surgery. For the anterior sextant of the maxilla, the remaining caries were removed and the teeth were filled by glass ionomer as temporary restorations. Finally, two free gingival grafts harvested from the palate at different times were placed in the areas of canines and premolars of both sides of the mandible, where inadequate keratinized gingiva was present. The patient was controlled via recall visits and supportive maintenance therapy.
Prosthetic Procedure
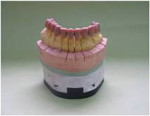
Through the use of physiotherapy and daily exercises with tongue blades, the increase in the patient’s mouth opening was enough to enable impression-taking to be performed. First, according to the facial landmarks and the patient’s vertical dimension at rest position, vertical dimension of occlusion (VDO) was determined and maintained during the time that the patient used temporary crowns on the upper and lower premolars. In the adjusted VDO, casting posts were made and temporary crowns were fabricated according to the diagnostic wax-up (Figure 4). According to Dawson’s guidelines, lower anterior temporary crowns were fabricated first with regard to the esthetics, phonetics, and harmony with the lower lip.18 Because of the extensive cervical and palatal carious lesions, upper anterior teeth were also prepared and covered with temporary crowns. The acrylic index was made and transferred to the articulator in order to customize the upper and lower anterior teeth and establish the canine rise occlusion. Then, temporary crowns were made for the posterior teeth and the occlusion was adjusted. The final impression was made, and definitive restorations were fabricated according to the existing indices. A soft nightguard was indicated because of disuse atrophy of the masticatory muscles and temporomandibular joints.
Results
On the first visit, it was noted that the patient had recently received a BMT and was taking medication. Due to the limitation of facial muscles activity, the maximum jaw opening was only 15 mm, making dental treatment impossible. Because of the intraoral mucosal lesions and special conditions, the patient was unable to perform plaque control procedures efficiently and the plaque index was significantly high (100%). After caries removal and SRP, the plaque index decreased to 91% but was still unacceptable. However, by completing the surgery and fabrication of 16 casting posts, plaque retention sites were reduced. The patient could perform oral hygiene procedure more efficiently, and the plaque index decreased significantly to 18%.
Using synthetic products such as previously mentioned Biotene products and fluoride therapy, gradually the saliva quality improved. Biotene dry-mouth products contain the patented Biotene triple enzyme system consisting of three enzymes and one protein that are found naturally in human saliva. Gradually, saliva quality increased and the oral lesions were reduced. After completing the surgical phase, inflammation and redness decreased significantly and the improved condition allowed for prosthesis fabrication. Finally, making a series of good quality prosthesis improved the patient’s ability in achieving optimum oral hygiene (Figure 5).
Discussion
In this case, the authors showed a full periodontal and prosthetic reconstruction in a patient with AA, a history of BMT, and immunosuppressive drug treatment, including cyclosporine (5 mg/day), CellCept (750 mg/day), and prednisone. As previously stated, cyclosporine dosages greater than 500 mg/day have been reported to induce gingival overgrowth; its magnitude appears to be related to the plasma concentration rather than to the patient’s periodontal status. Gingival enlargement, however, has not been observed in all patients taking the medication. The exact reason of more susceptibility to cyclosporine-induced enlargement is still unknown but could be related to the gingival cells possessing either unique receptors on their cell surfaces or more receptors than other cells. Special subpopulations of cells may also exist in these susceptible individuals. Alternatively, cells may possess specific internal pathways that respond to medications or hormones in a unique way in affected patients.11 In this patient, the consuming dosage and low susceptibility can be considered the main cause of the lack of gingival enlargement.
Because the laboratory tests showed that the platelet and white blood cell counts were always normal, platelet transfusion was not indicated. However, prophylactic antibiotic (amoxicillin 2 g 1 hour before every debridement and surgery) was prescribed due to the physician’s recommendations.
The gingiva showed severe bleeding even after mild irritation. Two main objectives of periodontal treatment for a patient with AA are to prevent infection and manage the hemorrhage—bleeding exaggerated locally or systemically by infection or due to bone marrow depression. Accordingly, oral bacteria must be removed in order to reduce the infection and hemorrhage. However, depending on the AA severity, periodontal treatment can be life threatening. Therefore, the clinician should evaluate a complete hematologic test in coordination with the patient’s physician to determine white blood cell, neutrophil, and platelet counts.18
The normal range of platelet count 150,000/µL to 450,000/µL is required before invasive procedures such as subgingival scaling or oral surgery. Leukopenia is often subsequent to steroid or other immunosuppressive agent administration, which might increase susceptibility to infection. Therefore, the practitioner who encounters a patient with AA should examine not only the platelet counts but also other leukocyte counts, particularly neutrophil.
The type of hepatitis-associated aphasic anemia is non A, non B, and most patients are seronegative for hepatitis A, B, and C.5 At the patient’s first visit, HBsAg, HBsAb, and HIV tests were checked. The findings were negative. His physician attributed the patient’s jaundice and paleness to BMT reaction. However, the authors thought it may have been related to the type of hepatitis that cannot usually be recognized. Although the hepatitis tests were negative, because the hepatitis type in these patients may not be recognized with usual tests, the patient could still be at risk of hepatitis. As a precaution, the authors used complete infection control techniques.
All members of the dental team play a role in the AA diagnosis not only because oral manifestations are common but also due to a chance of initial diagnosis. When oral (gingival or mucosal) bleeding without a local cause is detected in patients, a comprehensive test of blood hemostasis must be done. In a previous study in children with Fanconi anemia, the inherited type of AA, the prevalence of poor oral hygiene and dental caries was 35% and gingivitis prevalence 30%.19 It would not be unusual for dentists to see an AA patient who has a complaint of gingival or mucosal bleeding.19
At the patient’s first appointment, limited mouth opening (15 mm) was one of the chief complaints. This problem would hinder any dental treatment and potentially result in a compromise in nutrition. The condition was improved through a combination of daily exercise and physiotherapy; the more frequent the exercise regimen, the more beneficial the result. The simplest and least expensive method of exercising is using tongue blades.20 While the patient found the exercises painful, it was recommended that he perform them several times each day.
After these recommendations, the mouth opening was improved from 15 mm to 31 mm. The most effective method of treating carious conditions is through the daily use of topical applications of fluoride. Both stannous or sodium fluoride had been used in various forms (gel, rinse, and toothpaste) with significant success.20
In the authors’ patient, the gingival architecture was a thin biotype because the buccal marginal gingival was delicate and was also receded. There was also a thin buccal bone wall (Figure 6). In this kind of periodontium, flap reflection without laceration is difficult. Also, the keratinized gingiva width was inadequate in many locations. As a result, the authors tried to reflect the flap minutely and not only preserve the whole gingiva but also use the free soft-tissue graft for increasing the width of thin gingiva in the mandible.
Conclusion
The treatment modalities used for the present patient provides a new ray of hope for the treatment of medically comprised patient. This article also shows if these patients are well controlled, they can be treated similarly to a normal patient.
References
1. Goldman L, Ausiello D. Cecil Textbook of Medicine. 23 ed. Philadelphia, PA: Saunders Elsevier; 2007:1241-1246.
2. Rose NR, Bona C. Defining criteria for autoimmune diseases (Witebsky's postulates revisited). Immunol Today. 2007;14(9):426-430.
3. Marsh JC, Ball SE, Darbyshire P, et al; British Committee for Standards inHaematology. Guidelines for the diagnosis and management of acquired aplastic anaemia. Br J Haematol. 2003;123(5):782-801.
4. Aplastic Anemia and Myelodysplasia Association of Canada. Understanding aplastic anemia. 2010. https://www.aamac.ca/#AP. Accessed March 30, 2011.
5. Maciejewski JP, Risitano A. Aplastic anemia: management of adult patients. Hematology Am Soc Hemalol Educ Program. 2005;(1):110-117.
6. Hibbs JR, Frickhofen N, Rosenfeld SJ, et al. Aplastic anemia and oral hepatitis. Non-A, non-B, non-C? JAMA. 1992;267(15):2051-2054.
7. Pol S, Driss F, Devergie A, et al. Is hepatitis C virus involved in hepatitis-associated aplastic anemia? Ann Intern Med. 1990;113(6):435-437.
8. Bacigalupo A, Brand R, Oneto R, et al. Treatment of acquired severe aplastic anemia: bone marrow transplantation compared with immune suppressive therapy— The European Group for Blood and Marrow Transplantation Experience. Semin Hematol. 2000;37(1):69-80.
9. Young NS, Barrett AJ. The treatment of severe acquired aplastic anemia. Blood. 1995;85:3367-3377.
10. Torres HA, Bodey GP, Rolston KV, et al. Infections in patients with aplastic anemia: experience at a tertiary care cancer center. Cancer. 2003;98(1):86-93.
11. Newman MG, Takei HH, Klokkevold PR, et al. Carranza's Clinical Periodontology. 10th ed. Philadelphia, PA: Saunders; 2006:378,379,924.
12. Kim I, Yoon SS, Park S, et al. The treatment of severe aplastic anemia: outcomes of bone marrow transplantation and immunosuppressive therapy in a single institution of Korea. J Korean Med Sci .2003;18:365–371.
13. de Araujo MR, de Oliveira Ribas M, Koubik AC, Mattioli T, de Lima AA, França BH. Fanconi's anemia: clinical and radiographic oral manifestations. Oral Dis. 2007 May;13(3):291-295.
14. Sepúlveda E. Brethauer U, Rojas J, et al. Oral manifestation of aplastic anemia in children. J Am Dent Assoc. 2006;137(4):474-478.
15. Medspace reference. https://emedicine.medscape.com/article/960401-medication#3. Accessed Nov. 8, 2011.
16. Oyaizu K, Mineshiba F, Mineshiba J, et al. Periodontal treatment in severe aplastic anemia. J Periodontol. 2005;76(7):1211-1216.
17. Biotene® Dry Mouth Products. https://www.biotene.com/Products.aspx. Accessed Nov. 8, 2011.
18. Nissen-Druey C. Pathophysiology of aplastic anaemia. Baillieres Clin Haematol. 1989;2(1):37-49.
19. Tekcicek M, Tavil B, Cakar A, et al. Oral and dental findings in children with Fanconi anemia. Pediatr Dent. 2007;29(3):248-252.
20. Taylor TD. Clinical and Maxillofacial Prosthetics. Chicago, IL: Quintessence; 2000:43-45.
About the Authors
Somayeh Ansari Moghadam, MSD
Assistant Professo
Dental School
Zahedan University of Medical Science
Zahedan, Iran
Sharie Faghihi, MSD
Associate Professor
Dental Facult
Shiraz University of Medical Sciences
Shiraz, Iran
Reza Amid, MSD
Assistant Professor
Dental School
Shahid Beheshti Medical Universit
Tehran, Iran
Mahdi Kadkhodazadeh, MSD
Assistant Professor
Dental School
Shahid Beheshti Medical University
Tehran, Iran
Shabnam Khaje Hosseini ,MSD
Prosthodontis
Private Practice
Tehran, Iran
- no other hematologic disease
- bone marrow cellularity < 30%
-
two of the following blood criteria:
- neutrophils < 500/mm4
- platelets < 20,000/mm4
- absolute reticulocyte count < 40,000/mm4