Treatment of Obstructive Sleep Apnea Using Maxillomandibular Advancement Surgery
R. John Tannyhill III, DDS, MD
Abstract: Maxillomandibular advancement (MMA) is an invasive but potentially effective surgical option in the treatment of obstructive sleep apnea (OSA) for patients who have difficulty tolerating continuous positive airway pressure and whose OSA has been refractory to other surgical modalities. By advancing the maxillomandibular skeletal framework, the nasopharyngeal, retropalatal, and hypopharyngeal airways are enlarged, resulting in reduced pharyngeal collapsibility during negative-pressure inspiration. Meta-analysis of existing literature has shown a surgical success rate of 86% and an OSA cure rate of 43.2%. This article describes the MMA procedure and demonstrates successful results of the treatment.
Obstructive sleep apnea (OSA) is a sleep-related breathing disorder caused by repetitive airway obstruction during sleep that affects 9% to 38% of the adult population. It is associated with long-term health risks, such as cardiovascular disease, hypertension, type 2 diabetes mellitus, and depression.1 Additionally, OSA can take a severe toll on people's quality of life, job performance, and mental well-being.
Continuous positive airway pressure (CPAP) is accepted as the first-line therapy for moderate to severe OSA, but it requires nightly use to be effective. CPAP adherence is defined as nightly use of more than 4 hours; however, 46% to 83% of patients with OSA have been reported to be nonadherent to the relatively inconvenient and cumbersome CPAP treatment.2 This poor compliance and suboptimal use of CPAP often leads OSA patients to seek alternative solutions, including other non-invasive therapies and surgical treatment.3 These options could involve oral repositioning appliances, devices such as hypoglossal nerve stimulators, soft-tissue surgery, skeletal surgery or maxillomandibular advancement (MMA), and tracheostomy.
MMA is an invasive yet potentially effective surgical option in the treatment of OSA for patients who have difficulty tolerating CPAP and whose OSA has been refractory to other surgical modalities.4 By advancing the maxillomandibular skeletal framework, the nasopharyngeal, retropalatal, and hypopharyngeal airways are enlarged, resulting in reduced pharyngeal collapsibility during negative-pressure inspiration. Meta-analysis of existing literature has demonstrated a surgical success rate of 86%, defined as an apnea-hypopnea index(AHI) of <20 and a ≥50% reduction in the AHI post-MMA, and an OSA cure rate of 43.2%, defined as an AHI of <5.5
In an MMA procedure, the skeletal advancement is achieved via a maxillary LeFort osteotomy, mandibular bilateral sagittal split osteotomies, and, commonly, a genial tubercle advancement or advancement genioplasty that includes the genial tubercles. These osteotomies are usually planned virtually and stabilized with rigid internal fixation, frequently with 3D-printed titanium plates. Intermaxillary fixation is not required postoperatively.
The following sections describe this procedure, with a case presented that depicts key aspects of the treatment.
Patient Selection
Patients seeking MMA surgery often pursue consultation because of intolerance of CPAP and previously unsuccessful attempts at soft-tissue remediation, such as uvulopalatopharyngoplasty. A work-up should include consultations with sleep dentists, orthodontists for those patients with a pre-existing malocclusion, prosthodontists for patients who are completely or partially edentulous, sleep medicine specialists, and ear/nose/throat specialists. The indications for MMA surgery include CPAP intolerance, an AHI of >15 (considered moderate OSA) determined by a polysomnography study, and concomitant medical conditions such as hypertension and cardiovascular disease. Although the operative procedures are similar to those associated with orthodontic treatment and subsequent correction of a skeletal dentofacial deformity with orthognathic surgery, OSA patients tend to be older with comorbidities. As a result, the surgical work-up includes several additional pieces of data.
The workflow for both OSA patients and those with dentofacial deformity includes an orthognathic work-up, cone-beam computed tomography (CBCT) versus medical-grade CT, software planning (eg, Dolphin 3D, Dolphin, dolphinimaging.com; Quick Ceph Studio, Quick Ceph Systems, quickceph.com; IPS® CaseDesigner, KLS Martin, klsmartin.com), review of the orthognathic work-up with the patient, and finally, virtual surgical planning and fabrication of the implants (ie, plates and screws). This process may take several weeks. Additional components for the work-up of an OSA patient may include OSA severity, sleep-specific questionnaires such as the Epworth Sleepiness Scale, body mass index, airway anatomy, cephalometric analysis, fiberoptic nasopharyngoscopy, magnetic resonance imaging, fluoroscopy, and drug-induced sleep endoscopy. Whereas dentofacial deformities are often corrected surgically only after many months of orthodontic treatment, OSA patients can often be operated on within weeks. Those patients choosing to also undergo orthodontic treatment may elect to have surgery first to correct the OSA problem with the occlusion agreed upon prior to surgery in consultation with the orthodontist. Orthodontic options may include standard appliances or clear retainers, depending on the preference of the orthodontist and patient. However, many OSA patients elect not to proceed with orthodontics and to accept their current occlusion.
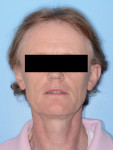
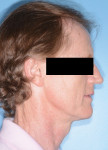
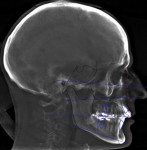
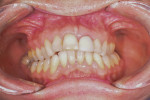
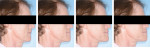
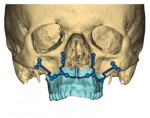
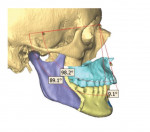
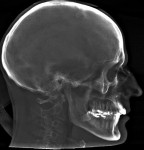
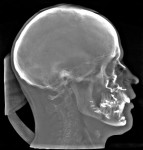
In the case presented, a 59-year-old male patient had severe OSA (AHI 45), decreased posterior airway space, and concomitant maxillofacial skeletal deformities (Angle class II malocclusion, maxillary and mandibular retrognathia, transverse maxillary hypoplasia, and transverse mandibular hypoplasia) (Figure 1 through Figure 4). Orthognathic surgical planning included tracings of a reconstructed lateral cephalogram to estimate postsurgical esthetic changes after advancements of 8 mm, 10 mm, and 12 mm (Figure 5). It is important to note that facial esthetics may change significantly, and this must be discussed in detail with the patient.
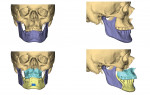
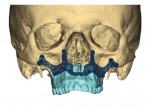
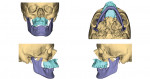
Virtual surgical planning in this case is demonstrated in Figure 6 through Figure 9: preoperative 3D CT imaging and planned surgical bony changes (Figure 6), preoperative 3D CT imaging and planned surgical soft-tissue changes (Figure 7), and examples of prefabricated cutting guides (Figure 8) and plates/screws for the maxilla (Figure 9). Careful attention must be paid to the root apices of the upper posterior and lower anterior teeth, the position of the inferior alveolar nerve and mental foramen, and any potential bony interferences after the reposition of the maxilla and mandible.
Surgical Procedure and Postoperative Course
OSA patients undergoing MMA are normally prescreened by the anesthesia personnel, and all comorbidities are considered. Nasal intubation and general anesthesia are administered, and hypotensive anesthesia during maxillary surgery is performed to decrease blood loss, which averages 300 ml to 500 ml. The operation typically takes 4 to 6 hours.
Either the maxilla or the mandible may be operated on and relocated first, and then the opposing jaw is moved into the final occlusion. The typical amount of movement is 10 mm, although this may be modified depending on the pre-existing occlusion and facial esthetics.6In the present case, Figure 10 shows the position of the maxilla after advancement, relative to the native position of the mandible; this is also known as the intermediate position. Figure 11 depicts the position of the mandible after advancement relative to the previously advanced maxilla, ie, the final planned surgical occlusion.
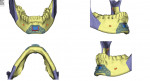
Once a stable, reproducible final occlusion is confirmed, attention is turned to the genial tubercle advancement. This is a small, 1 cm x 2 cm bony block extending from just inferior to the anterior mandibular tooth roots that allows for the advancement of the genioglossus musculature and thus the tongue, resulting in greater posterior airway space (usually 6 mm to 8 mm). In the present case, the advancement of the genioglossal attachments was 6 mm (Figure 12).
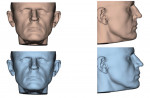
The patient is then awakened and sent to either the intensive care unit overnight or the surgical floor, depending on any preoperative comorbidities and the operative course. Hospital stay is generally 1 to 2 nights. A postoperative CBCT is taken on postoperative day 1. In the present case, the postoperative CBCT, as compared to a preoperative CBCT, demonstrated the successful results of the treatment (Figure 13 and Figure 14).
Patients will typically have pain for 2 to 5 days postoperatively that is commonly managed with narcotic medications. Facial swelling peaks at 1 to 2 weeks and takes 6 to 8 weeks to largely resolve. Facial dynamism takes several weeks to resolve as well.
Up to 42% of patients have a permanent deficit of the inferior alveolar nerve distribution, resulting in a paresthesia or anesthesia of the lips and chin, with increasing likelihood if a full genioplasty is done.7 If present postoperatively, this numbness may continually improve for up to 18 months. In-office follow-up visits should be every 1 to 2 weeks for the first 6 to 8 weeks, and a follow-up polysomnography test should be done at 6 months post-procedure to confirm success of the surgery. A successful MMA is defined as achieving an AHI of <20 and a ≥50% reduction in the AHI post-procedure, and potential OSA cure, defined as an AHI of <5.5
Conclusion
Maxillomandibular advancement is second only to tracheostomy as the most successful treatment for severe obstructive sleep apnea.4 Patients are often able to maintain their oxygen saturation on room air on the night of their surgery. Many patients have concerns over their postoperative esthetic changes, especially younger patients, but such concerns usually resolve after several months as they grow accustomed to their new appearance. In the author's experience, almost all patients have a significant increase in their sleep quality and quality of life. Sleep practitioners should carefully consider this treatment option after appropriate work-up.
About the Author
R. John Tannyhill III, DDS, MD
Oral and Maxillofacial Surgery Residency Program Director, Massachusetts General Hospital, Boston, Massachusetts; Assistant Professor, Harvard School of Dental Medicine, Boston, Massachusetts; Fellow, American College of Surgeons
References
1. Senaratna CV, Perret JL, Lodge CJ, et al. Prevalence of obstructive sleep apnea in the general population: a systematic review. Sleep Med Rev. 2017;34:70-81.
2. Weaver TE, Grunstein RR. Adherence to continuous positive airway pressure therapy: the challenge to effective treatment. Proc Am Thorac Soc. 2008;5(2):173-178.
3. Gottlieb DJ, Punjabi NM. Diagnosis and management of obstructive sleep apnea: a review. JAMA. 2020;323(14):1389-1400.
4. Zaghi S, Holty JE, Certal V, et al. Maxillomandibular advancement for treatment of obstructive sleep apnea: a meta-analysis. JAMA OtolaryngolHead Neck Surg. 2016;142(1):58-66.
5. Holty JE, Guilleminault C. Maxillomandibular advancement for the treatment of obstructive sleep apnea: a systematic review and meta-analysis. Sleep Med Rev. 2010;14(5):287-297.
6. Giralt-Hernando M, Valls-Ontañón A, Guijarro-Martínez R, et al. Impact of surgical maxillomandibular advancement upon pharyngeal airway volume and the apnoea-hypopnoea index in the treatment of obstructive sleep apnoea: systematic review and meta-analysis. BMJ Open Respir Res. 2019;6(1):e000402.
7. McLeod NM, Bowe DC. Nerve injury associated with orthogna-thic surgery. Part 2: inferior alveolar nerve. Br J Oral Maxillofac Surg.2016;54 (4):366-371.