Digital Technology Redefining Implant Therapies: Two Case Reports
Sundeep Rawal, DMD
Abstract: Providing clinicians with comprehensive tools to transform the smiles of their patients, digital applications are wholly changing the dental profession. The impact of technology on the delivery of care has enabled clinicians to be increasingly more analytical and meticulous in planning and rigidly focused on the predictability of outcomes. Tools are available that allow clinicians to better understand their patients, not only anatomically and biomedically, but with regard to their personal goals for their oral care. This article highlights some of the more profound technological advancements clinicians have at their disposal to both plan and deliver streamlined minimally invasive treatment that equals or exceeds historically cutting-edge esthetic dentistry. The case studies presented indicate how professional collaboration and prosthetically driven planning may be utilized to achieve lofty goals for patient satisfaction, even in highly complex and challenging cases.
Traditionally, clinical restorative excellence required the practitioner to be a veritable blend of "Aquinas" and "Fra Angelico." Trained to achieve precise anatomy with dexterous manual technique, the best clinicians factored substantial logic and reason into the science; however, the creation of prostheses that were functional and esthetically natural additionally demanded the artist's eye for color and visual form. While there will always be a niche for superlative clinicians who possess these myriad talents, thanks to digital dental applications, more practitioners can achieve pristine results with the aid of sophisticated technology, and therefore more patients can receive optimal and predictable outcomes.
By no means has the multitude of software systems competing for the clinician's attention in today's dental practice caused the prosthetic smile design to be compromised. In fact, the traditional treatment plan now commences with extremely comprehensive analyses of the patient's face, converting multi-angular photographs into computer-generated results that consider midline, lip lines, and even the distance between the eyes and mouth.1 In addition to drawing very specific guidelines into the restorative plan for the clinician, contemporary software affords the patient a preview of the result, which often may be the chief motivational factor for acceptance of treatment.
Guided Implant Placement
Once the proposed smile design is approved by the patient, the same data can be imported into dental implant planning software to determine the position of dental implants. This is beneficial because digitally planning the case for the placement of dental implants is most effective when the planning is prosthetically driven,2 thus allowing for the angulation of the implants to anchor the prostheses with maximum precision. The implant-placing clinician then incorporates the appropriate files from the planning software into the guided modality of choice. Three types of guided implant placement are currently used in dentistry: static guided surgery with a printed resin or laser sintered metal guide, dynamic camera-based navigation applications, and robot-assisted dental implant placement. The latter combines the advantages of a physical boundary associated with a printed guide and the flexibility of interoperative changes associated with camera-based navigation, fostering the emergence of a modality that results in highly predictable functional and esthetic outcomes.3
Surgical robotics have been implemented in multiple disciplines for sophisticated medical treatment, not only to ensure precision and achieve predictable results but also to reduce recovery time for the patient. Guided dental implant placement has been evolving since the development of physical static guides as far back as 2004 (which are still viable in practice today although with less flexibility than the current navigational and robotic options).4 Dynamic camera navigation for implant placement came about a decade later and significantly influenced the art of precision. According to Block and Emery, who are pioneers in the use of dynamic navigation, control of the depth and angulation of implants became more predictably accurate, surgeons could more consistently avoid the inferior alveolar nerve, and flap mobilization could be minimized to promote a less invasive procedure for the patient.5 The use of navigation assists the case collaboration between the surgical and restorative clinicians in marrying the virtual plan to the orchestration of the treatment, thereby enabling the achievement of a high level of patient-specific results. Dynamic navigation has been widely adopted because it is a flexible, time- and cost-effective workflow; however, as Block and Emery further indicate, the clinician must undergo a learning curve to gain proficiency and will need to factor in training and simulation.5
Robotic surgical guidance has been in a state of continual refinement since its introduction in 2017. Because it is still in its infancy, numerous multicenter evidence-based studies will be required to produce the anticipated superior data outcomes. Robotic guidance brought the term "haptic" into the surgical vocabulary. Haptic refers to physical guidance in addition to visual and auditory guidance during implant surgery; the robotic system's software program utilizes a CBCT scan of the patient and allows the 3D planning of ideal implant positioning based on bone availability, biomechanical load, and the design of the definitive prostheses.The robotic assistance then provides the surgeon with physical guidance of the drills to the desired position, angulation, and depth. When the orientation is accurate to the plan, there is no robotic (haptic) resistance; if the drills deviate, the robot will constrain the tool axis to the planned orientation. Haptic refers to the surgeon experiencing a vibrating resistance to the normal sensations of drilling or implanting.6
While static, navigational, and robotic guides all provide valuable digital assistance in achieving esthetic, functional outcomes, the future of robotic guidance promises to achieve the highest degree of accuracy. Static guides run the risk of fracture and can impede the clinician's visibility to the osteotomy site; moreover, it is impossible to adjust the actual plan during the surgical procedure and still perform a guided surgery. Intraoperative navigation allows for clinician adjustments and provides real-time visual information through a display, although there tends to be a more rigid adherence to the digital plan. Furthermore, the procedure that is performed is essentially freehand with no physical boundary. Haptic guidance currently aims at providing the best adherence to the plan as it originates in the minds of the clinicians. The following case studies, using an entirely digital workflow, demonstrate dramatically reduced deviations from plan to execution using robotic guidance.
Case 1
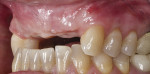
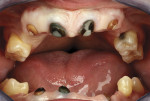
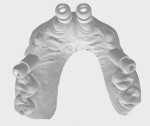
A middle-aged female patient presented with a severely damaged maxillary dentition due to traumatic injury (Figure 1). Ordinarily, this treatment would be planned to splint multiple adjacent implants for optimal mechanical strength and load distribution.7 However, in this case the patient specifically requested an independent implant for each single tooth (sites Nos. 6 through 10), as she previously had had poor experiences with bridged restorations and strongly desired to be able to maintain the restorations in a manner that was as close to natural, single teeth as possible without needing any adjunctive maintenance protocols due to splinting of restorations. Although this option is not typically recommended because of the challenges it poses in execution of therapy from an esthetic perspective, the clinician agreed to this approach to satisfy the patient's desires. The treatment was thus digitally planned accordingly.
A completely digital workflow was created allowing for collaborative treatment with the oral surgeon. The diagnostic phase included prolific intraoral photography and CBCT scanning, with concentration focused on the age-appropriate interdental papillae display for the patient, which is a key esthetic element needed for the virtual planning of the treatment.8
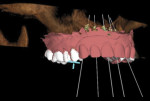
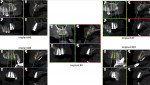
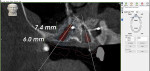
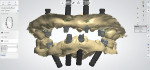
The all-digital treatment plan was prosthetically driven (Figure 2), with the clinician mindful of the critical urgency for optimal implant placement, with implants ideally 3 mm apart with most of the interproximal osseous crest maintained9 and with strict adherence to the design of the plan, based on Tarnow et al's esthetic guidelines for the maxillary anterior esthetic zone.10 The clinical team elected to use robotic guidance (Yomi® Dental Robotic System, Neocis, neocis.com), which on the day of surgery permits fiduciary markers to verify the accuracy of placement, angulation, and depth.
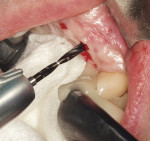
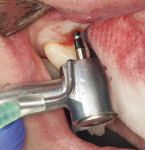
In preparation for the surgical procedure, modest bone grafting and soft-tissue augmentation was used to create more ideal oral parameters to receive the implants. Five implants (NobelActive®, Nobel Biocare, nobelbiocare.com) were placed in the maxillary anterior esthetic zone using haptic robotic guidance (Figure 3 and Figure 4). The procedure was entirely flapless, contributing to minimizing trauma for the patient. The surgeon, in full control of the robot, guided the arm to the planned implant positions while inspecting the soft-tissue type at the emergence, noting the bone quality, and monitoring adherence to the plan with the robotic system's intraoperative accuracy verification. The real-time audio and visual updates confirmed that the clinical parameters had been successfully achieved.
Postoperative CBCTs indicated a high level of accuracy adhering to the planned implant placement, with minimal compromise to the surrounding soft tissue (Figure 5). For the restorative dentist, the delivery of this highly accurate implant placement in ideal quality bone helped ensure that a robust foundation was securely in place for the definitive prostheses. During the healing period of 4 months, the patient wore a removable provisional prosthesis.
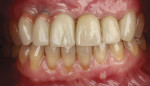
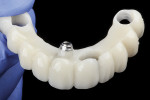
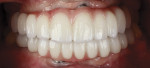
Five individual screw-retained milled ceramic crowns completed the all-digital treatment used to restore the patient without complication. As indicated in photographs taken at delivery of the final prostheses, the wholly digitally planned and executed restoration achieved a completely technological, natural-appearing outcome, and the patient was extremely satisfied (Figure 6).
Case 2
A male patient presented with classic ectodermal dysplasia, the congenital anomaly caused by a single abnormal gene or pair of abnormal genes.11 The typical malformation of the alveolar ridge, bone deficiency, and absence of tooth buds were evident (Figure 7).
Maxillofacial rehabilitation of adults inflicted with ectodermal dysplasia is most successfully accomplished through therapeutic protocols utilizing osseointegrated dental implants and advanced ceramic prosthodontics to provide esthetically pleasing smiles.12 The treatment plan for this patient was for implementation of a definitive prosthesis in the maxillary arch built on four implants-two in the anterior esthetic zone and two posterior tilted implants placed adjacent to the anterior wall of the sinus (Figure 8).
A fully integrated digital design was created focusing on the patient's desired outcomes of having a highly esthetic, natural-looking smile with ideal form and function that was as close to a natural dentition as possible. The smile design created using design software (3Shape Dental System, 3Shape, 3shape.com) and surface scanning images were merged with STL and CBCT files to engineer a virtual surgical plan, and then a pre-manufactured screw-retained fixed provisional restoration was fabricated-milled polymethyl methacrylate manufactured on a five-axis mill (M5 Heavy Metal Milling Unit, Zirkonzahn, zirkonzahn.com)-that would function as the basis for the desired result (Figure 9 and Figure 10). The hypoplastic nature of the tissue and congenital deformity necessitated strict precision in the placement of the implants. Robotic guidance was employed to ensure the surgeon could make real-time plan adjustments if the bone or soft tissue contraindicated the treatment plan.
Predictability governed every aspect of this plan. Prior to the surgical phase of treatment, the clinical team scrutinized their bank of statistical analysis from 40 previously treated patients indicating coronal, apical, and angular deviations using robotic guidance. Each consideration of tissue augmentation, bone remodeling, implant placement, and prosthesis adjustment created "confidence intervals" for the success of this case. Knowing the predictability of the technology, the surgeon could proceed to extract the necessary teeth and place the four implants. The provisional was already prepared with one of the anterior abutments embedded to facilitate alignment with the remaining implants (Figure 11). The patient eventually was restored in both the maxilla and mandible with four implants in each arch to create an esthetic smile and functional restoration that was anticipated to be long-lasting (Figure 12).
Discussion
The delivery of precise and artistic implant and prosthodontic care has experienced a paradigm shift due to technology. Over the past decade aggressive progress has been achieved in honoring Dr. Per-Ingvar Bränemark's mandate: the success of implant reconstruction lies in the precision of the placement of the implants themselves.13 The onset of virtual planning has significantly advanced freehand surgical procedures, but the concept of guided surgery, which led to camera navigation and eventually robotic guidance, has enhanced the processes utilized for precise placement of implants. It has also significantly reduced the risk of damage to nerves and neighboring teeth.14 Additionally, navigated flapless implant placement optimizes implant position in relation to the prosthetic requirements by engaging more bone, leading to primary stability for immediate loading.14
Digital data acquisition has allowed clinicians to learn more about their patients' oral physiology than ever before. Intraoral scanners have all but eliminated conventional impressions and stone casts, serving as a reliable basis for accurate STL files, which when married to CBCT imaging gives clinicians a volume of diagnostic information to virtually plan even the most complex case.15 Assessment of bone quality and tissue integrity with digital insight greatly contributes to the veracity of the plan.
The most astutely planned and precisely executed implant surgery will still require a prosthodontic finish that functions biomechanically and achieves natural esthetics ideally suited to each individual patient. If there is one aspect of prosthodontic treatment that might find challenges in the digital age, it would be in the painstaking artistic labors of the ceramist, shaping and painting teeth to mimic nature; however, with the continual refinement of digitally manufactured prostheses, both provisional and definitive, equality with handmade products has seemingly been achieved.
Many patients today choose implant-supported dental solutions as, along with almost instant gratification, they provide psychological security, increased self-confidence, more secure chewing ability, and improved phonetics and esthetics.16 Both the milling and printing of prostheses have evolved, and current technology includes software that can simultaneously create the substructure, veneer, and soft-tissue replication in a single process. These manufactured prostheses, if based on accurate clinical recording and careful laboratory digitizing, are able to deliver a true-to-nature smile that may require no clinical adjustments.17
Conclusion
While there will always be a place for human wisdom and logic and the eyes of an artist in the dental profession,18 the tremendous advancements in digital technology have vastly improved the precision and endurance of restorative care. Using software for the entire treatment workflow from data acquisition to prosthetic delivery conserves substantial time, making quality care more widely available and broadening the spectrum of patients who may benefit from attaining a healthy smile.
About the Author
Sundeep Rawal, DMD
Private Practice, Merritt Island, Melbourne/Suntree, Lake Nona, and Winter Park, Florida; Co-founder, Digital Dentistry Institute
References
1. Coachman C, Calamita MA, Coachman FG, et al. Facially generated and cephalometric guided 3D digital design for complete mouth implant rehabilitation: a clinical report. J Prosthet Dent. 2017;117(5):577-586.
2. Horowitz J, Zuabi O, Machtei EE. Accuracy of a computerized tomography-guided template-assisted implant placement system: an in vitro study. Clin Oral Implants Res. 2009;20(10):1156-1162.
3. Bolding S, Reebye U. Accuracy of haptic robotic guidance of dental implant surgery for completely edentulous arches. J Prosthet Dent. 2021;S0022-3913(21)00056-1.
4. Marra R, Acocella A, Rispoli A, et al. Full-mouth rehabilitation with immediate loading of implants with computer-guided flap-less surgery: a 3-year multicenter clinical evaluation with oral health impact profile. Implant Dent. 2013;22(5):444-452.
5. Block MS, Emery RW. Static or dynamic navigation for implant placement - choosing the method of guidance. J Oral Maxillofac Surg. 2016;74(2):269-277.
6. Srivastava R, Jyoti B, Kushwaha S, Priyadarshi PK. Computer aided navigation for predictable dental implantology: a review. Natl J Integr Res Med. 2019;10(3):63-67.
7. Balshi SF, Wolfinger GJ, Balshi TJ. A protocol for immediate placement of a prefabricated screw-retained provisional prosthesis using computed tomography and guided surgery and incorporating planned alveoplasty. Int J Periodontics Restorative Dent. 2011;31(1):49-55.
8. Hochman MN, Chu SJ, Tarnow DP. Maxillary anterior papilla display during smiling: a clinical study of the interdental smile line. Int J Periodontics Restorative Dent. 2012;32(4):375-383.
9. Spear F. Implants or pontics: decision making for anterior tooth replacement. J Am Dent Assoc. 2009;140(9):1160-1166.
10. Tarnow DP, Cho SC, Wallace SS. The effect of inter-implant distance on the height of inter-implant bone crest. J Periodontal. 2000;71(4):546-549.
11. Freire-Maia N. Ectodermal dysplasia. Hum Hered. 1971;21:309-312.
12. Petropoulos VC, Balshi TJ, Wolfinger GJ, Balshi SF. Ectodermal dysplasia: an 11-year follow-up of siblings with 2 implant treatment approaches. Implant Dent. 2014;23(4):387-393.
13. Bränemark PI, Hansson BO, Adell R, et al. Osseointegrated implants in the treatment of the edentulous jaw. Experience from a 10-year period. Scand J Plast Reconstr Surg Suppl. 1977;16:1-132.
14. Witter G, Adeyemo WL, Schicho K, et al. Navigated flapless transmucosal implant placement in the mandible: a pilot study in 20 patients. Int J Oral Maxillofac Implants. 2007;22(5):801-807.
15. Arunyanak SP, Harris BT, Grant GT, et al. Digital approach to planning computer-guided surgery and immediate provisionalization in a partially edentulous patient. J Prosthet Dent. 2106;116(1):8-14.
16. Balshi T, Balshi S. A new digital solution for implant supported restorations. Inside Dental Technology. 2017:8(3):40-46.
17. Marchak CB. CAD/CAM-guided implant surgery and fabrication of an immediately loaded prosthesis for a partially edentulous patient. J Prosthet Dent. 2007;97(6):389-394.
18. Rawal S, Tillery DE, Brewer P. Robotic-assisted prosthetically driven planning and immediate placement of a dental implant. Compend Contin Educ Dent. 2020;41(1):26-30.