Efficacy of Removal of Residual Dental Cement by Laser, Ultrasonic Scalers, and Titanium Curette: An In Vitro Study
Paul Fletcher, DDS; Eric Linden, DMD; Charles Cobb, DDS, MS, PhD; Donggao Zhao, PhD; Jamie Rubin, BFA, RG; and Penny Planzos, DMD, MS
Abstract
The purpose of this study was to conduct an in vitro evaluation of the efficacy of six dental lasers, two ultrasonic scalers, and a titanium curette in the removal of dental cement from the surface of an implant. The study used a total of 39 dental implants, representing three different surface textures. The implants were divided into 13 groups with one of each of the three surface textures in a group. A standardized amount of modified resin dental cement was applied to the implant surface. Each test instrument was used as a monotherapy. Additionally, three of the lasers were used as part of a dual therapy in conjunction with the piezo ultrasonic scaler. Laser irradiation was limited to 2 minutes. Following treatment, implants were graded visually and by scanning electron microscopy (SEM) for the presence of unremoved cement and concomitant damage, if any, to the implant surface. The results showed that no treatment removed all residual cement from any of the three implant surfaces, although specific protocols appeared more effective than others. Implant surface damage was frequently observed, both visually and by SEM, and appeared to result from laser irradiation and the use of ultrasonic scaling instruments.
Bränemark's original design for a final implant restoration called for the restoration to be screw retained to underlying abutments.1 As the demand for single implant restorations has increased over the years, implant companies have fostered the use of a cement-retained protocol in an effort to make implant restorative techniques similar to conventional fixed prosthetics. One study reported that in general dental practices 93% of the restorations were cement-retained.2 Although there are advantages to cemented restorations, they have proven to have significant limitations.
Because the margin of an implant crown often ends 3 mm or more below the gingival margin, residual cement, expressed from the margin of a cemented restoration, frequently remains. Excess cement has been shown to contribute to peri-implant bone loss when it is left subgingivally,3-5 with resin-modified cements being most frequently implicated.6 Additionally, patients with a prior history of periodontal disease who have restorations cemented with resin-modified cement have an increased tendency to develop peri-implant disease compared with patients without a prior history of periodontitis.7
While cementation protocols call for the use of temporary cement, a wide variety of luting agents are used in dentistry, and implant restorations are often cemented permanently. With resin-modified glass-ionomer cement protocols being taught as a procedure of choice in many dental schools,8 this potential problem may likely increase with time. In contrast, screw-retained restorations can be removed, allowing for their repair or modification as needed, and the underlying implant can be accessed for treatment if peri-implantitis develops.
A primary objective in the treatment of peri-implantitis is decontamination of the implant surface, with the goals of therapy being elimination of inflammation, reduction in pocket probing depth, stabilization of bone levels, and, ideally, regeneration of lost bone.9 There is no established and universally accepted protocol for achieving these objectives.10,11 The use of a dental laser, either as a monotherapy or in conjunction with other types of instrumentation, has been suggested as part of a peri-implantitis treatment protocol.12,13
While no dental lasers are inherently suited to decontaminate titanium without inflicting heat-related damage, each wavelength can be adjusted to use parameters that will achieve a clean surface while minimizing the potential for hard- and soft-tissue or implant surface damage. Although the photothermal effects of lasers have been shown to kill bacteria on an implant surface by evaporation, denaturization, and ablation,14-16 no study has assessed a laser's ability to remove subgingival residual dental cement. The goal of this investigation was to evaluate the efficacy of six different lasers, a titanium curette, a magnetostrictive elliptical ultrasonic scaler, and a piezo linear ultrasonic scaler in the removal of residual subgingival dental cement. Concomitant damage to the implant surface was also assessed.
Materials and Methods
Thirty-nine implants with three different commercially available surface textures were used in the study. Thirteen implants had a dual acid-etched surface (DAE) (Osseotite®, Zimmer Biomet, zimmerbiometdental.com), 13 had a resorbable blast media surface with a laser machined collar (RBM) (Laser-Lok®, Biohorizons, biohorizons.com), and 13 had a sandblasted, large-particle, acid-etched surface (SLA) (Straumann® SLA®, Straumann, straumann.com).
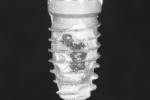
The fixture mount of each implant was placed in a stone base to facilitate its manipulation during instrumentation. A paper mask with a 4-mm diameter circular opening in the center was placed around the circumference of the implant, and a urethane resin-modified temporary cement (Premier® Implant Cement™, Premier Dental, premierdentalco.com), developed specifically for use with dental implants, was placed in the opening and smoothed with a beaver-tail instrument to keep it level with the outer diameter of the implant threads (Figure 1). The test implants were then divided into 13 groups, with each group having one implant representing each type of surface.
The test instruments were used as a monotherapy or as part of a dual therapy (ie, laser + piezo ultrasonic scaler) on the cement of each of the three implant surfaces for a maximum of 2 minutes. Manufacturers' recommended settings, published treatment protocols, or laser settings assumed unlikely to damage an implant surface or bone were followed.12-16
Twelve instruments and instrument combinations were tested: (1) titanium curette (Ti curet); (2) magnetostrictive elliptical ultrasonic scaler with a metal tip (magnetostrictive scaler); (3) piezo linear ultrasonic scaler with a metal tip (piezo scaler); (4) diode laser (980 nm) (diode laser); (5) diode laser (980 nm) + piezo scaler (diode + piezo scaler); (6) Nd:YAG laser (1,064 nm) (Nd:YAG laser); (7) Nd:YAG laser (1,064 nm) + piezo scaler (Nd:YAG + piezo scaler); (8) Er,Cr:YSGG laser (2,780 nm) (Er,Cr:YSGG laser); (9) Er:YAG laser (2,940 nm) (Er:YAG laser); (10) CO2 laser (9,300 nm) (CO2 9300 nm); (11) CO2 laser (10,600 nm) (CO2 10,600 nm); (12) CO2 laser (10,600 nm) + piezo scaler (CO2 10,600 nm + piezo scaler); plus untreated controls (control) (Figure 1).
The following laser treatment protocols were used:
(1) diode, 980 nm: 2-3 watts, pulsed, non-focused, non-initiated, non-contact for 20 seconds, followed by piezo scaler for 100 seconds.
(2) Nd:YAG, 1,064 nm: 4 watts, 20 Hz, 100 μsec, 180 mJ, 360 μm diameter optical fiber. Irradiation for 20 seconds, non-contact mode, followed by piezo scaler for 100 seconds.
(3) Er,Cr:YSGG, 2,780 nm: 1.5 watts, 30-40 Hz variable pulse rate, H mode, PFTPS-5 tip, 40%/50% air/water, 120 seconds duration of irradiation.
(4) Er:YAG, 2,940 nm: 40-50 mJ, 20-30 Hz, PS600T tip, water/air 7/10 ratio, 120 seconds duration of irradiation.
(5) CO2, 9,300 nm: Ultraguide, 15/600 tip, 1.25 mm spot size, 20% governor speed, 1% mist, 120 seconds duration of irradiation.
(6) CO2, 10,600 nm: 2 watts, superpulse, 0.25 ceramic tip, high air purge, laser for 25 seconds followed by piezo scaler for 95 seconds.
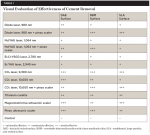
At the completion of treatment each implant surface was examined visually, and using a three-point scale, the effectiveness of the treatment for the removal of the cement was estimated (Table 1). The three-point assessment scale was as follows:
+ Minimal treatment effect = some cement removal from crest of implant threads, no cement removal from the concavities between threads (Figure 2 and Figure 3).
++ Moderate treatment effect = removal of cement from crest of implant threads, some cement removal between threads, but minimal exposure of the subjacent implant surface (Figure 4 and Figure 5).
+++ Mostly effective treatment = removal of cement from crest of implant threads and most of the cement between threads, with exposure of most of the subjacent implant surface (Figure 6 and Figure 7).
After visual assessment, the specimens were evaluated by scanning electron microscopy (SEM) for the presence of residual cement and for damage to the titanium surface (Table 2).
Results
From a visual standpoint, none of the treatments was completely successful at removing all of the cement from the implant surfaces (Table 1). The SEM images supported the visual observations with respect to effectiveness of cement removal and expanded on these findings from the perspective of surface alterations.
The CO2 9300 nm laser visibly removed most of the cement on all three implant surfaces and showed minimal surface changes under SEM (Figure 6 and Figure 7), while the Er,Cr:YSGG and Er:YAG lasers and the Nd:YAG laser alone had a minimal effect on the cement and implant surface (Figure 2 and Figure 3). Cement removal by the CO2 10,600 nm laser as a monotherapy was variable depending on the surface tested.
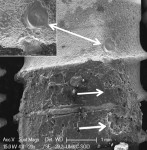
The Ti curet, magnetostrictive scaler, piezo scaler, Nd:YAG + piezo scaler, and the CO2 10,600 nm + piezo scaler showed moderate to mostly effective treatment results, although scratches on the implant surfaces were observable. A flattening and smearing of the crests of the implant threads was apparent under SEM (Figure 4, Figure 5, Figure 8, and Figure 9). These effects were not evident when the two lasers were applied as a monotherapy.
Damage to the cement layer and implant surfaces was visually apparent between the threads of the implants treated with the diode laser (Figure 10 and Figure 12). Additionally, SEM revealed areas of heat-induced titanium surface melting and cavitational defects with melting of residual cement (Figure 11 and Figure 13). SEM examination of implants subjected to the diode + piezo scaler protocol showed chipping, smearing, and flattening of the implant threads.
Discussion
Each of the 12 instrumentation protocols was tested for its effectiveness at removing a layer of resin-modified temporary cement from three different implant surfaces. The cement was an elastomeric polymer resin with light viscosity, developed specifically for use with implants, and is one of the more widely used products of its type in the United States.17 An attempt was made to smooth the cement onto the implant surface at a uniform thickness, within the confines of the threads (Figure 1). However, given that each implant body presented its own characteristic shape, pitch, angle, and depth of threads, and that each implant type varied in coronal-apical taper, it could not be assumed that this goal was routinely achieved. Use of a scanner in future studies would mitigate this potential inconsistency.
The 2-minute timeframe used for cement removal was an arbitrary choice. As all instrumentation was accomplished using direct vision of the cement and implant surface, it was thought that this length of time would suffice for visual quantification and provide the operator a sense of relative effectiveness of the various treatment protocols.
Two aspects of the cement removal process were assessed: the instrument's proficiency in removing cement from the implant surface, and the amount of damage, if any, resulting from the instrumentation. The composition of the cement seems to be a factor in determining a laser's effectiveness. A laser will remove cement from an implant surface if its wavelength is absorbed and heats the cement to a temperature sufficient for vaporization.13 Thus, depending on a specific cement's chemical composition and density, one laser may remove a specific cement more effectively than another.
Table 1 reveals that none of the treatment modalities was completely successful at removing all cement from the implant test surfaces. Post-treatment SEM evaluation was carried out to obtain magnified views of residual cement and assess damage to the implant surface that might have occurred as a result of treatment. The effectiveness of cement removal varied markedly, with the implants treated with the CO2 9,300 nm laser protocol being mostly free of cement (Figure 6 and Figure 7) and the Er,Cr:YSGG and Er:YAG lasers (Figure 2 and Figure 3) and Nd:YAG laser (as a monotherapy) being minimally effective. When used as part of a dual therapy in conjunction with the piezo scaler, the Nd:YAG, CO2 10,600 nm, and diode lasers were only marginally more effective than the piezo scaler alone.
The Ti curet's mechanical action, the magnetostrictive scaler's elliptical motion, and the piezo scaler's linear stroke pattern were moderately to mostly successful at removing cement on the three test implant surfaces. However, shedding of titanium particles from the implant surface into soft tissue has been reported to occur with ultrasonic scaling.18 Corrosion of these particles has been associated with the development of inflammatory peri-implant disease.19,20 The shedding caused by the ultrasonic scalers would seem to call into question their routine use in surface decontamination or cement removal protocols.
The post-treatment SEM images revealed that surface damage varied and included scratching, flattening, smearing, and chipping of the crests of the implant threads. Additionally, both visually and under SEM, the diode laser, when used as a monotherapy, showed localized areas of carbonization and cement surface damage due to excessive heat.When used as a dual therapy with the piezo scaler, less damage was evident as the diode laser was not used alone for the total decontamination time. Although the damages noted might preclude osseointegration or soft-tissue adherence at specific sites on an implant, extensive damage that might interfere with re-osseointegration of an entire implant surface was not observed using any of the test instruments at the prescribed settings (Table 2).
Because a patient with a prior history of periodontal disease is more susceptible to peri-implant disease,7 and the chemical composition of certain cements exacerbates the inflammatory response, it would seem prudent to use a mechanically retained restoration and avoid cemented restorations in susceptible patients. If the use of a cemented restoration is unavoidable, cement control techniques, such as cementing dyes and cement shield membranes, should be utilized.21,22
Conclusions
The following conclusions may be made as a result of this study:
None of the instrument protocols tested was completely effective for the removal of resin-modified temporary implant cement, even with direct vision of the surface. Consequently, the efficacy of protocols advocating a closed flap approach for implant cement removal must be questioned.
The CO2 9,300 nm laser was the most promising treatment tested and deserves further evaluation. It removed most of the cement on all three implant surfaces and inflicted little to no surface changes.
Surface scratching, smearing, and flattening of the crest of the implant threads were common findings using SEM, with localized areas of surface melting evident after the use the diode laser. While not substantial enough to preclude integration of an entire implant, whether these surface changes or corrosion from the shedding of titanium particles are sufficient to cause an adverse tissue reaction is unknown.
Additional research evaluating the effectiveness of the test instruments on a variety of cements is indicated, as different cement compositions may be more easily removed.
Acknowledgment
The authors acknowledge and thank David R. Scharf, DMD, for his technical assistance with this project.
About the Authors
Paul Fletcher, DDS
Associate Clinical Professor, Division of Periodontics, Stony Brook University School of Dental Medicine, Stony Brook, New York; Private Practice, New York, New York
Eric Linden, DMD
Assistant Clinical Professor, Columbia University College of Dental Medicine, New York, New York; Private Practice, Woodcliff Lake, New Jersey
Charles Cobb, DDS, MS, PhD
Professor Emeritus, Department of Periodontics, School of Dentistry, University of Missouri-Kansas City, Kansas City, Missouri
Donggao Zhao, PhD
Director of Electron Microscope Laboratory, School of Dentistry, University of Missouri-Kansas City, Kansas City, Missouri
Jamie Rubin, BFA, RG
New York City College of Technology, New York, New York
Penny Planzos, DMD, MS
Private Practice, Brooklyn, New York
References
1. Bränemark PI, Zarb, GA, Albrektsson, T. Tissue-Integrated Prostheses: Osseointegration in Clinical Dentistry. Chicago, IL: Quintessence Publishing; 1985:241-282.
2. Da Silva JD, Kazimiroff J, Papas A, et al. Outcomes of implants and restorations placed in general dental practices: a retrospective study by the Practitioners Engaged in Applied Research and Learning (PEARL) Network. J Am Dent Assoc. 2014;145(7):704-713.
3. Wilson TG Jr. The positive relationship between excess cement and peri-implant disease: a prospective clinical endoscopic study. J Periodontol. 2009;80(9):1388-1392.
4. Callan DP, Cobb CM. Excess cement and peri-implant disease. J Implant Adv Clin Dent. 2009;1(6):61-68.
5. Cobb CM, Satheesh KM, Salas ML, MacNeill SR. Biophysical factors affecting bacterial adhesion to dental implant surfaces: a focused review. J Implant Adv Clin Dent. 2015;7(5):61-67.
6. Linkevicius T, Vindasiute E, Puisys A, Peciuliene V. The influence of margin location on the amount of undetected cement excess after delivery of cement-retained implant restorations. Clin Oral Implants Res. 2011;22(12):1379-1384.
7. Linkevicius T, Puisys A, Vindasiute E, et al. Does residual cement around implant-supported restorations cause peri-implant disease? A retrospective case analysis. Clin Oral Implants Res. 2013;24(11):1179-1184.
8. Tarica DY, Alvarado VM, Truong ST. Survey of United States dental schools on cementation protocols for implant crown restorations. J Prosthet Dent. 2010;103(2):68-79.
9. Renvert S, Roos-Jansaker AM, Claffey N. Non-surgical treatment of peri-implant mucositis and peri-implantitis: a literature review. J Clin Periodontol. 2008;35(8 suppl):305-315.
10. Gosau M, Hahnel S, Schwarz F, et al. Effect of six different peri-implantitis disinfection methods on in vivo human oral biofilm. Clin Oral Implants Res. 2010;21(8):866-872.
11. Esposito M, Grusovin MG, Worthington HV. Treatment of peri-implantitis: what interventions are effective? A Cochrane systematic review. Euro J Oral Implantol. 2012;5(suppl):S21-S41.
12. Aoki A, Mizutani K, Schwarz F, et al. Periodontal and peri-implant wound healing following laser therapy. Periodontol 2000. 2015;68(1):217-269.
13. Ishikawa I, Aoki A, Takasaki AA, et al. Application of lasers in periodontics: true innovation or myth? Periodontol 2000. 2009;50:90-126.
14. Dederich DN, Pickard MA, Vaughn AS, et al. Comparative bactericidal exposures for selected oral bacteria using carbon dioxide laser radiation. Lasers Surg Med. 1990;10(6):591-594.
15. Rooney J, Midda M, Leeming J. A laboratory investigation of the bactericidal effect of a NdYAG laser. Br Dent J. 1994;176(2):61-64.
16. Linden E, Vitruk P. SuperPulse 10.6 μm CO2 laser-assisted, closed flap treatment of peri-implantitis. Implant Practice. 2015;8:30-34.
17. Premier Implant Cement, Product Review. Dental Advisor. 2005;22:20.
18. Harrel SK, Wilson TG Jr, Pandya M, Diekwisch TG. Titanium particles generated during ultrasonic scaling of implants. J Periodontol. 2019;90(3):241-246.
19. Noronha Oliveira M, Schunemann WVH, Mathew MT, et al. Can degradation products released from dental implants affect peri-implant tissues? J Periodontal Res. 2018;53(1):1-11.
20. Souza JGS, Oliveira BEC, Bertolini M, et al. Titanium particles and ions favor dysbiosis in oral biofilms. J Periodontal Res. 2020;55(2):258-266.
21. Wadhwani CPK. Peri-implant disease and cemented implant restorations: a multifactorial etiology. Compend Contin Educ Dent. 2013;34(spec no 7):32-37.
22. Haas RC, Haas SE. Cement shield membrane technique to minimize residual cement on implant crowns: a dental technique. J Prosthet Dent. 2020;123(2):223-227.