Socket Preparation for Delayed Implant Placement Using a Mineralized Cancellous Allograft
Robert A. Horowitz, DDS; and Gregori M. Kurtzman, DDS
Abstract: Various materials have been used for extraction socket preservation in anticipation of implant placement or to limit resorption of socket walls. Materials have ranged from autogenous bone, allografts, and xenografts to synthetics. Ideally, the graft material being placed into the socket at the time of extraction should fully convert to host bone to yield vital bone that will permit implant osseointegration with as much contact between bone and the implant surface as possible. This article reviews different grafting materials and presents two cases in which extraction of a molar was required and future implant placement planned. One site closure needed a resorbable membrane, and the other used a nonresorbable membrane due to the size of the crestal opening. In each case the clinician utilized a different particle size of mineralized cancellous allograft material. Particle size is a factor with respect to how long the graft remains prior to conversion by the surrounding host tissue to vital bone.
Often when loss of a tooth that is deemed non-salvageable due to periodontal, endodontic, or restorative issues is planned, delayed implant placement after extraction is the course taken to allow site healing. Reasons for this approach may be the presence of insufficient bone to house the implant to provide initial stability or residual pathology that led to the failure of the natural tooth.1,2 This is especially common with molar sites, because typically the interseptal bone is the only bone present for implant placement. Also, prosthetic planning requires the implant to be placed at the center of the site where inadequate bone will initially house the implant.3 In these situations, socket grafting is indicated to develop a more ideal site to house the implant and achieve better initial stability after implant placement.4,5 Socket grafting followed by delayed implant placement allows for a prosthetically driven implant placement in an optimally developed bony site.
The tooth serves as support to the buccal and lingual osseous walls of the socket. Under function the tooth stimulates the surrounding bone to maintain and preserve its osseous contours in a stable position. Upon extraction of the tooth, the stimulating effects that occur during function are lost along with the support provided by the tooth, and the result is collapse of the socket, leading to resorption.6,7 Socket preservation, thus, is required to preserve the crestal dimensions to ensure adequate volume to house the implant that will be placed following site healing.8
Socket Preservation Materials
Various materials have been advocated for socket preservation whether in anticipation of implant placement or to limit resorption of socket walls at the crest for the site to serve as a pontic area or under removable prosthetics.9 These products range from autogenous bone, allografts, and xenografts to synthetic materials. Ideally, the graft material being placed into the socket at the time of extraction should fully convert to host bone to yield vital bone that will permit implant osseointegration with as much contact between bone and the implant surface as possible. Conversion of the graft to host bone is volume dependent, with a larger volume of graft material requiring longer periods to fully convert. Typically, a period of 3 to 5 months for conversion is ideal. Xenograft materials and some synthetic grafts either slowly convert or do not convert, leaving residual particles that can decrease the bone-to-implant contact. This process can affect long-term loading of implants placed within these materials by potentially decreasing implant stability and/or increasing susceptibility to peri-implant bone loss.10,11 These residual particles are space fillers. Ideally, vital bone that is able to functionally manage loads on the implant over time should be in contact with the implant surface.
Human bone particles, whether autogenous (from the same patient) or allograft (from a tissue bank), are preferred for socket preservation because they are similar to the surrounding host bone and can act as a scaffold. In this role, they enable ingrowth of osteoblast cells and the release of host and/or exogenous growth factors leading to full conversion of the graft to host bone. Autogenous bone has the advantage over other graft materials of having no additional material cost, but a secondary site is needed to harvest from the patient during treatment, and limited volumes can be obtained.12 Allograft materials offer unlimited availability in the volume accessible to fill a surgical defect (ie, extraction socket or sinus or ridge augmentation), but there is an additional cost for the material. This bone is available in mineralized or demineralized forms, and the particles are cortical, cancellous, or in combination. The mineralized form is advantageous in that it retains volume longer than the demineralized form, allowing host ingrowth to convert the graft material without a potential collapse in the area being grafted.13 Particle size also is a factor in regard to how long the graft remains prior to conversion by the surrounding host tissue to vital bone. Smaller particles (0.25 mm to 1 mm) are well suited for smaller extraction sockets, filling of defects or thread exposure at implant placement, periodontal defects, and apicoectomies. Larger particles (1 mm to 2 mm) are better suited for larger extraction sockets and sinus and ridge augmentation.
Treatment for extraction socket preservation is ideally accomplished with a mineralized cancellous allograft, as the particle surface area and porosity allows for immediate, stable clot formation to retain the graft in the socket during the initial healing phase. The mineralized bone provides an osteoconductive scaffold for efficient turnover to vital host bone within a reasonable period (3 to 6 months) before the site can accept implant placement. Following extraction of the tooth and curettage of any residual tissue or pathology within the socket, there should be bleeding into the socket, which allows host bone cells and growth factors from blood vessels into the graft placed. If available, autogenous blood concentrates, such as platelet-rich fibrin (PRF) or platelet-rich plasma (PRP), can be mixed into the mineralized cancellous bone particles by the practitioner to form a gelatinous mass.14,15
Placing a membrane over the underlying graft material is advised, as containment within the socket during the initial healing phase will help prevent disruption of the graft material. Because achieving primary closure at an extraction site is often not possible or desired and is not attempted in flapless cases, the membrane helps prevent oral fluids, food particles, and bacteria from contacting the graft material. Such contact could lead to breakdown of the graft and subsequent loss in volume in the site.16
The type of membrane selected is dependent on the size of the crestal opening over the extraction site, number of bony walls, and whether the soft-tissue margins are to be approximated over the site. Larger crestal openings may require a membrane that is slowly resorbable or nonresorbable, because it will need to maintain its integrity longer so that soft tissue will be able to grow across it to seal the site or enable osteoid to form beneath it. When the size of the crestal opening is minimal (3 mm to 4 mm or less) a faster resorbing or short-term removable membrane may be used. Long-term collagen membranes are fully absorbable over a 3- to 4-month period; are cell occlusive, preventing soft-tissue cellular ingrowth into the graft material before it can organize; and have excellent biocompatibility with surrounding tissue encouraging soft-tissue growth over the membrane from the flap margins.17
Two cases are presented in which extraction of a molar was required and future implant placement planned. One site closure needed a resorbable membrane; in the other, a nonresorbable membrane was indicated due to the size of the crestal opening. In each case the clinician utilized a different particle size of mineralized cancellous allograft material.
Case 1
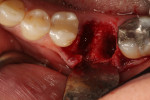
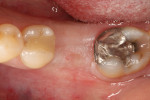
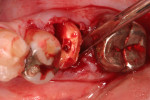
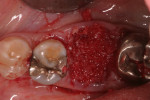
A 56-year-old woman presented with pain on the mandibular left first molar. The tooth had had prior endodontic treatment and the referring doctor had identified a fracture in the mesial root, leading to a hopeless prognosis. After administration of local anesthetic, venous blood was drawn and placed into a centrifuge to create leukocyte- and platelet-rich fibrin (L-PRF) (IntraSpin®, BioHorizons, biohorizons.com). To preserve blood supply to the buccal plate of bone, no flap was elevated. After the tooth was extracted, the sockets were curetted to remove any residual pathologic tissue remaining at the site, yielding a clean extraction socket ready for placement of osseous graft material (Figure 1).
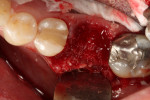
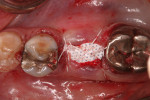
The socket was filled to the crest with mineralized cancellous bone allograft with a particle size of 1 mm to 2 mm (Surgical Esthetics, surgicalesthetics.com) that had been hydrated with liquid expressed from formation of the L-PRF barriers (Figure 2). A resorbable collagen membrane (CollaShield™, Surgical Esthetics) that was hydrated in liquid expressed from the L-PRF process was placed over the graft and tucked under the buccal and lingual gingival margins (Figure 3). The collagen membrane would remain intact for approximately 16 weeks, as per the manufacturer, before being fully absorbed into the patient's adjacent soft tissue as it grows over the healing socket. The marginal gingival tissues were stabilized with a 5-0 polyglycolic acid (PGA) suture (Hu-Friedy, hu-friedy.com) in a figure-8 fashion to contain the resorbable membrane (Figure 4).
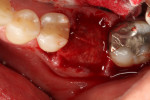
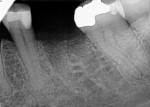
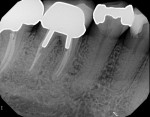
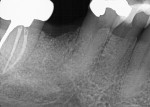
The patient was seen at monthly intervals, and at 5 months post-socket grafting the quality of bone present was evaluated radiographically to determine if the site was ready for implant placement. The examination noted healthy non-inflamed keratinized tissue covering the site and a preservation of width (Figure 5). A periapical radiograph was taken and revealed that the graft blended with the adjacent host bone with no crestal loss noted (Figure 6).
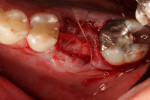
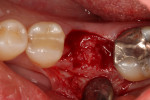
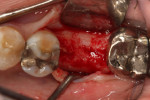
Local anesthetic was administered and an incision was made to the lingual aspect of the site to permit the keratinized tissue to be mobilized to the facial, thus increasing the zone of keratinized tissue that results following healing. This provides a larger zone of tissue than what a crestal or "punch" approach would provide (Figure 7).
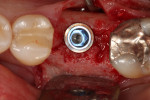
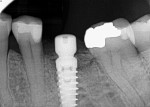
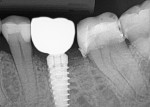
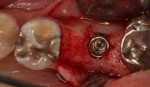
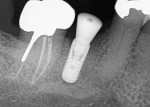
Upon flap elevation it was noted that a majority of the former socket was filled with bone that appeared vital with no discernable graft particles (Figure 7). A core sample was removed at the center of the site of the planned osteotomy using a trephine and sent for histological evaluation. Due to the size of the mesial root, the core included primarily grafted, regenerated bone. The osteotomy was completed and a 5 mm x 11.5 mm Hahn™ implant (Glidewell Dental, glidewelldental.com) was inserted into the site flush with the crestal bone (Figure 8). A healing abutment was placed and the soft tissue was adapted with sutures (Figure 9). Three months after this, impressions were made for the final restoration, which was inserted 2 weeks later. A radiograph was taken of the completed restoration to verify mating of the abutment to the implant (Figure 10).
The osseous core sample was processed by non-demineralized methods to preserve all of the original structure and stained with Stevenel's blue and van Gieson's picrofuchsin. It is shown in Figure 11 in conventional and polarized versions. The histological report noted that 47% of the sample was bone with 80% of that being vital (Figure 11, left). The polarized version of the specimen image demonstrated some remnants of the allograft that had been placed (pink) and immature bone (blue) (Figure 11, right). Collagen fibers were noted within the osseous sample under polarizing light.
Case 2
A 62-year-old woman presented with pain on the endodontically treated mandibular right first molar. A deep class 2 furcation was noted with a suspected fracture in the furcation. Examination clinically and radiographically determined that the tooth was not salvageable, and extraction and site preservation with bone grafting was recommended (Figure 12).
At the time of surgery, local anesthetic was administered and blood was drawn for preparation of L-PRF. The tooth was sectioned at the furcation to decrease trauma to the alveolar housing. The distal root was elevated to the mesial and extracted atraumatically (Figure 13).
Mineralized cancellous bone allograft with a particle size of 0.25 mm to 1 mm (Surgical Esthetics) was hydrated with L-PRF to form a gelatinous mass and the socket was filled to the crest (Figure 14). Due to the large area uncovered at the crest a nonresorbable membrane was utilized. The buccal and lingual gingival margins were elevated 2 mm to 3 mm. A nonresorbable high-density polytetrafluoroethylene (PTFE) membrane (Cytoplast™ TXT-200, Osteogenics Biomedical, osteogenics.com) was placed over the mineralized cancellous allograft and tucked under the buccal and gingival margins. It was secured with 4-0 PTFE sutures (Cytoplast™, Osteogenics Biomedical) in an interrupted fashion to contain the membrane and underlying graft material during the initial healing phase (Figure 15).
The barrier was removed 3 weeks postoperatively. Six months after the extraction the patient was seen for a postoperative appointment to evaluate the site. A periapical radiograph was taken to assess graft healing (Figure 16). The graft had blended with the surrounding host bone and no graft particles were noted radiographically. The site was determined to be ready for implant placement.
After administration of local anesthetic, a crestal incision with sulcular incisions on the buccal and lingual was made to minimally expose the crestal bone (Figure 17). Upon examination, it was noted that complete fill of the socket had been achieved. A significant preservation of alveolar ridge width of bone that was dense to instrument touch was present and no graft particles were observed.
A core sample was taken with a trephine at the planned osteotomy location slightly distal to the center of the crest to harvest more of the grafted and regenerated bone where the larger distal root had been located. The osseous core sample was processed as in Case 1. The osteotomy was completed and a 4.1 mm x 10 mm bone-level implant (Straumann® Bone Level Implant, Straumann, straumann.com) was placed (Figure 18). The implant was fully stable on insertion. A healing abutment was placed (Figure 19), and the flap was repositioned and sutured with 5-0 PGA sutures (Hu-Friedy).
Histologically, new bone formation was seen in the core sample (Figure 20). Overall, the core was filled with 70% bone, all of which was vital. In the high-power view shown in Figure 20, immature bone in the sample was noted in orange and more mature bone presented in red. No graft particles were noted in the histological evaluation of the core sample, confirming full conversion of the allograft to vital host bone.
Discussion
Various surgical techniques are available for the removal of a tooth and the replacement of lost alveolar structure. Eliminating flap elevation and placing a graft and/or barrier reduces site collapse.5 When graft materials are utilized, particle size can influence conversion rate and consideration should be given based on the volume of the defect being filled, number of bony walls, and anticipated time before re-entry is planned.
In a dog model, Artzi and colleagues showed that quicker bone formation occurred in a barrier-protected socket or one where a resorbable alloplast was placed rather than a xenograft.10 In their study, in the equivalent timeframe of 9 months in a human, at the center of the preformed socket most of the xenograft would have been embedded in avascular connective tissue, which is not ideal for implant placement.
Kadkhodazadeh and colleagues compared synthetic hydroxyapatite and a xenograft in dogs.11 After allowing 12 weeks of healing, they found new bone of 35% to 41%, residual graft of 14% to 15%, and connective tissue of 45% to 50% in grafted extraction sockets. Correlating this to humans, it would be weak type III bone after 9 months of healing. In the present article, the cases shown with the specific allograft used in this study were re-entered at much earlier timeframes than 9 months. The vital bone percentage in the two core biopsies shown was greater in the present human study than in the aforementioned dog study. Also, there was significantly less residual graft and very little connective tissue. All of this indicates that there is a probability of greater primary implant stability and a higher initial bone-to-implant contact with these materials. This should provide patients treated with these techniques and biomaterials both a higher osseointegration success rate and greater ability to prevent peri-implant disease.
Conclusion
When implant placement cannot be performed at the time of extraction, the treatment goal should be socket preservation to maintain ridge contours and volume and provide an optimal site for subsequent implant placement. Use of mineralized cancellous bone particles provides a scaffold for ingrowth of surrounding cells and allows angiogenesis for the conversion of the graft to vital host bone, typically in 4 to 6 months depending on socket size, number of bony walls, and other factors. When longer periods of time are anticipated before implant placement into the grafted site, mineralized cortical bone offers a slower conversion rate and is able to maintain volume longer than mineralized cancellous particles. When larger defects are being grafted, use of a mixture of cancellous/cortical particles should be considered, as this will maintain the volume better, limiting potential site collapse during the healing phase and allowing host conversion.18 Particle size also influences conversion rate and should be considered; determining factors are the volume of the defect being filled and the anticipated time before re-entry is planned. The authors have found that histological studies of different graft materials support a higher percentage of vital bone at equivalent periods of healing with the materials used in this study, which they have observed in their clinical use.
Disclosure
This article was commercially supported by Surgical Esthetics.
About the Authors
Robert A. Horowitz, DDS
Adjunct Clinical Assistant Professor, Department of Periodontology and Implant Dentistry, New York University College of Dentistry, New York, New York
Gregori M. Kurtzman, DDS
Master, Academy of General Dentistry; Diplomate, International Congress of Oral Implantologists; Private Practice, Silver Spring, Maryland
References
1. Cosyn J, De Lat L, Seyssens L, et al. The effectiveness of immediate implant placement for single tooth replacement compared to delayed implant placement: a systematic review and meta-analysis. J Clin Periodontol. 2019;46 suppl 21:224-241.
2. Kim YK, Ku JK. Extraction socket preservation. J Korean Assoc Oral Maxillofac Surg. 2020;46(6):435-439.
3. Tettamanti L, Andrisani C, Bassi MA, et al. Post extractive implant: evaluation of the critical aspects. Oral Implantol (Rome). 2017;10(2):119-128.
4. Dos Santos Canellas JV, D'Albuquerque Medeiros PJ, da Silva Figueredo CM, et al. Which is the best choice after tooth extraction, immediate implant placement or delayed placement with alveolar ridge preservation? A systematic review and meta-analysis. J Craniomaxillofac Surg. 2019;47(11):1793-1802.
5. Horowitz R, Holtzclaw D, Rosen PS. A review on alveolar ridge preservation following tooth extraction. J Evid Based Dent Pract. 2012;12(3 suppl):149-160.
6. Ben Amara H, Kim JJ, Kim HY, et al. Is ridge preservation effective in the extraction sockets of periodontally compromised teeth? A randomized controlled trial. J Clin Periodontol. 2021;48(3):464-477.
7. Couso-Queiruga E, Stuhr S, Tattan M, et al. Post-extraction dimensional changes: a systematic review and meta-analysis. J Clin Periodontol. 2021;48(1):126-144.
8. Avila-Ortiz G, Chambrone L, Vignoletti F. Effect of alveolar ridge preservation interventions following tooth extraction: a systematic review and meta-analysis [erratum appears in J Clin Periodontol. 2020;47(1):129]. J Clin Periodontol. 2019;46 suppl 21:195-223.
9. Horowitz RA, Leventis MD, Rohrer MD, Prasad HS. Bone grafting: history, rationale, and selection of materials and techniques. Compend Contin Educ Dent. 2014;35(4 suppl):1-6.
10. Artzi Z, Weinreb M, Givol N, et al. Biomaterial resorption rate and healing site morphology of inorganic bovine bone and beta-tricalcium phosphate in the canine: a 24-month longitudinal histologic study and morphometric analysis. Int J Oral Maxillofac Implants. 2004;19(3):357-368.
11. Kadkhodazadeh M, Sohrabi Z, Abrishami MR, et al. Histologic and histomorphometric assessment of xenograft bone substitute versus synthetic nonceramic hydroxyapatite for canine tooth-socket preservation. J Long Term Eff Med Implants. 2019;29(4):281-288.
12. Kim YK, Yun PY, Um IW, et al. Alveolar ridge preservation of an extraction socket using autogenous tooth bone graft material for implant site development: prospective case series. J Adv Prosthodont. 2014;6(6):521-527.
13. Wen SC, Barootchi S, Huang WX, Wang HL. Time analysis of alveolar ridge preservation using a combination of mineralized bone-plug and dense-polytetrafluoroethylene membrane: a histomorphometric study. J Periodontol. 2020;91(2):215-222.
14. Mogharehabed A, Birang R, Torabinia N, et al. Socket preservation using demineralized freezed dried bone allograft with and without plasma rich in growth factor: a canine study. Dent Res J (Isfahan). 2014;11(4):460-468.
15. Solakoglu Ö, Heydecke G, Amiri N, Anitua E. The use of plasma rich in growth factors (PRGF) in guided tissue regeneration and guided bone regeneration. A review of histological, immunohistochemical, histomorphometrical, radiological and clinical results in humans. Ann Anat. 2020;231:151528. doi: 10.1016/j.aanat.2020.151528.
16. Duong M, Mealey BL, Walker C, et al. Evaluation of healing at molar extraction sites with and without ridge preservation: a three-arm histologic analysis. J Periodontol. 2020;91(1):74-82.
17. Troiano G, Zhurakivska K, Lo Muzio L, et al. Combination of bone graft and resorbable membrane for alveolar ridge preservation: a systematic review, meta-analysis, and trial sequential analysis. J Periodontol. 2018;89(1):46-57.
18. Demetter RS, Calahan BG, Mealey BL. Histologic evaluation of wound healing after ridge preservation with cortical, cancellous, and combined cortico-cancellous freeze-dried bone allograft: a randomized controlled clinical trial. J Periodontol. 2017;88(9):860-868.