Direct and Indirect Pulp Capping: A Brief History, Material Innovations, and Clinical Case Report
Gary Alex, DMD
Abstract: Among the goals of pulp capping are to manage bacteria, arrest caries progression, stimulate pulp cells to form new dentin, and produce a durable seal that protects the pulp complex. This article will provide a general discussion of direct and indirect pulp capping procedures, offering practitioners a pragmatic and science-based clinical protocol for treatment of vital pulp exposures. A clinical case will be presented in which a novel light-cured resin-modified mineral trioxide aggregate hybrid material was used to manage a mechanical vital pulp exposure that occurred during deep caries excavation.
Direct and indirect pulp capping, employing various materials and clinical protocols, has been used for many years to preserve the health and vitality of the pulp complex and induce pulp cells to form hard tissue (reparative/tertiary dentin). Direct pulp capping is used when the pulp is visibly exposed (vital pulp exposure) due to caries, trauma, or iatrogenic insult such as accidental exposure during tooth preparation or caries removal. Indirect pulp capping is generally used in deep cavity preparations, with or without caries remaining, that are in close proximity to the pulp but with no visible exposure. The ultimate objectives of any pulp capping procedure should be to manage bacteria, arrest any residual caries progression, stimulate pulp cells to form new dentin, and provide a biocompatible and durable seal that protects the pulp complex from bacteria and noxious agents.
The success of both direct and indirect pulp capping procedures is, of course, contingent on the health and vitality of the pulp complex to begin with. Teeth that have a history of unprovoked spontaneous pain, necrotic or partially necrotic pulps, radiographic pathology, or excessive hyperemia on direct pulp exposure due to irreversible pulpitis have a poor prognosis and often require endodontic intervention or extraction at some point. Conversely, pulps that are vital and healthy are viable candidates for pulp capping procedures. Indeed, the pulp may be a far more resilient tissue then many believe, provided it is healthy to begin with, bacteria is managed, and an environment and durable seal can be created using materials conducive to the pulp's continued health.
This article will provide a general discussion of direct and indirect pulp capping procedures and highlight a clinical case using a novel light-cured resin-modified mineral trioxide aggregate (MTA) hybrid material to manage a mechanical vital pulp exposure that occurred during deep caries excavation. The article aims to provide a pragmatic, practical, and science-based clinical protocol for dealing with vital pulp exposures.
Indirect Pulp Capping: Two-Stage Approach
When used appropriately, both direct and indirect pulp capping procedures have the potential to preserve pulp health, function, and vitality.1 In the case of indirect pulp capping, where the cavity preparation is in close proximity to the pulp but with no visible exposure, various one- and two-stage protocols have been advocated.2 With two-stage or stepwise caries removal techniques all carious dentin typically is removed from the walls and dentino-enamel junction of the cavity preparation. A layer of deep carious dentin, which is usually discolored but firm, may be left on the floor of the preparation if its removal might cause a pulp exposure.3 Typically, a liner such as calcium hydroxide [Ca(OH)2] is then placed and overlaid by a provisional restoration such as zinc oxide and eugenol or glass ionomer.
Of vital importance with this technique is the placement of a well-sealing provisional restoration for several months that isolates any remaining caries and bacteria from the oral environment. In fact, in terms of caries arrestment and dentin remineralization/reorganization, several studies suggest the provision of a seal and entombment of residual bacteria to arrest caries progression is more important than any specific base or liner placed initially.4-6 As an example, a recent clinical study restoring deep carious lesions using a two-stage indirect pulp capping protocol that used a resin-modified glass ionomer (RMGI) provisional with and without first placing a calcium hydroxide liner found no clinical benefit to using said liner.7 Although this was a short-term study (3 months) it supports the view held by some that the provision of a seal (in this case by the RMGI provisional) is more important than any specific indirect pulp capping liner or base that is placed initially.
After several months, and assuming all goes well during the provisional trial period (ie, no signs or symptoms of pain or pathology), the patient returns for the second step of the two-stage indirect pulp capping procedure. While there are variations in materials and technique, the provisional is typically removed, remaining caries is removed to hard tissue, and a final restoration is placed. The hope is that some degree of dentin remineralization, along with formation of reparative dentin and dentin bridging, occurred during the time interval between the first and second appointments allowing for residual caries removal during the second appointment without exposing the pulp.
While a number of studies and case reports support or advocate for various two-stage indirect pulp capping procedures,8-12 many dentists are not comfortable leaving residual caries in their cavity preparations and prefer to remove all caries at the initial appointment even at the risk of pulp exposure.13 Indeed, a PEARL (Practitioners Engaged in Applied Research and Learning) study conducted in 2007 indicated that most dentists prefer to remove all decay initially, while only 20% favored partial caries removal.14 These findings are corroborated in a more recent study that also found the vast majority of surveyed dentists, when restoring deep carious lesions, preferred complete carious removal to hard dentin at the initial appointment even at the risk of pulp exposure.15
Indirect Pulp Capping: One-Step Approach
With one-stage indirect pulp capping techniques typically all or most of the caries is removed at the initial appointment, some type of indirect pulp capping material is placed in close approximation to but not in direct contact with the pulp, and the final restoration is placed, all in the same appointment. One common technique is to remove only the "infected dentin" (dentin that is demineralized with denatured collagen, infiltrated with bacteria, and irreparably damaged) while leaving the "affected dentin" in place (dentin that is also demineralized but with the collagen structure still largely intact, is bacteria free, and still has the potential for remineralization). Typically, the affected dentin is then covered with a base and/or liner in the hope that over time it will remineralize, forming hard bacteria-free dentin. While this seems reasonable in theory, the clinical reality is that it can be extremely difficult to differentiate between infected and affected dentin. Caries detecting solutions (typically propylene glycol mixed with various dyes) that in principle stain only the denatured collagen of the infected dentin (Figure 1 and Figure 2) may be useful adjuncts in this regard, but their accuracy is questionable and it is doubtful they indicate with certainty that all active caries has or has not been removed.16,17
The technique the author prefers is the judicious use of caries detecting solutions in conjunction with careful and thorough use of tactile and visual criteria to assess the caries status of the dentin during cavity excavation and preparation. Also, dentists must be aware that it is more difficult to predictably bond directly to deep caries-affected dentin than normal dentin, because caries-affected dentin is different in morphological, chemical, and physical characteristics.18,19 Many dentists prefer to place some type of base or liner in deep cavity preparations prior to the use of an adhesive system and placement of the final restoration. One technique that has worked well for the author when dealing with deep caries-affected dentin is to first disinfect the substrate with a 2% aqueous solution of chlorhexidine digluconate (eg, Cavity Cleanser™, BISCO, bisco.com; Concepsis®, Ultradent Products, ultradent.com) followed by the placement of a RMGI liner (eg, Vitrebond™, 3M, 3m.com; Fuji Lining™ LC, GC America, gcamerica.com) (Figure 3 through Figure 6). The RMGI liner is placed in a thin layer (≤1 mm) before placing a dentin bonding agent and composite restorative. In vivo clinical studies support this general protocol.20,21
RMGI liners have several positive attributes, including good adhesive and sealing properties via micromechanical and chemical interaction with dentin.22 They are simple to mix and place, release high sustained levels of fluoride,23 have significant antimicrobial properties24,25 and low solubility,26,27 and exhibit a favorable modulus of elasticity and coefficient of thermal expansion and contraction (similar to that of dentin).28,29 In addition, RMGI liners have been shown in many studies to help reduce gap formation and microleakage.30-34 While there is scientific and anecdotal35,36 evidence supporting the use of RMGI liners in close proximity to (but not in direct contact with) pulp, their use as direct pulp capping agents is generally contraindicated in the literature.1,29,37,38 Clearly, the remaining dentin thickness, which is very difficult to access clinically, has an effect on the pulpal response to any indirect pulp capping material.38 The author has previously discussed in detail the use of RMGI liners as indirect pulp capping agents.39,40
Direct Pulp Capping
Direct pulp capping is used when the pulp is visibly exposed (vital pulp exposure) due to caries, trauma, or iatrogenic insult such as accidental exposure during tooth preparation or caries removal. The procedure typically involves arresting any pulpal hemorrhage followed by covering and sealing exposed pulp tissue in some fashion to preserve its health, function, and viability. Calcium hydroxide has traditionally been considered the "gold standard" and is the most commonly used material in this regard.41 This is partly because of its ability to dissociate into calcium and hydroxyl ions, its high pH, antibacterial properties, and apparent ability to stimulate odontoblasts and other pulp cells in various ways to form reparative dentin.1,41-43 Studies have also shown that the high pH of calcium hydroxide causes superficial coagulation necrosis where it contacts the pulp. This provides a degree of hemostasis and stimulates mineralized tissue and dentin bridge formation.44,45
Most likely, another reason for the widespread use of calcium hydroxide as a direct pulp capping agent on vital exposures is because this is the technique most dentists were taught in dental school. By and large, dentists use what they know, are comfortable with, and have had a reasonable degree of success with. Even when newer and potentially superior techniques and materials emerge, change can be slow to come. "Science and technology revolutionize our lives, but memory, tradition, and myth frame our response."46 Although successful outcomes are sometimes achieved with calcium hydroxide direct pulp caps, calcium hydroxide does have significant disadvantages, including lack of innate adhesive and sealing abilities, poor physical properties, and dissolution over time.47 In addition, some studies show that the dentin bridge formed under calcium hydroxide pulp caps contains multiple "tunnel" defects and porosities.48-50
Although it has long been accepted that calcium hydroxide has significant antimicrobial properties (most studies support this), at least one study questions this assumption.51 Long-term clinical studies show success rates with calcium hydroxide pulp caps on carious exposures to be highly variable, generally unpredictable, and often unsuccessful.52 It makes sense to develop and test medicaments for direct pulp capping that overcome some of the deficiencies of calcium hydroxide and have the potential to provide more consistent and predictable clinical outcomes.
In 1824 Joseph Aspdin, a British stone mason, obtained a patent for a cement he formulated in his kitchen. The inventor heated a mixture of finely ground limestone and clay and ground the mixture into a powder creating a hydraulic cement (one that hardens with the addition of water). Aspdin named the product Portland cement because the set cement resembled a stone quarried on the Isle of Portland off the British coast.53 Today, various versions and formulations of Aspdin's Portland cement are used to create everything from concrete blocks, support beams, grout, mortar, and foundation slabs for roadways, buildings, and backyard patios. Aspdin had no way of knowing that roughly 170 years after his discovery this same product would form the backbone of a new class of calcium and alumina silicate based so-called "bioactive" dental materials, one of which was mineral trioxide aggregate (MTA).
Emergence of MTA and MTA/Hybrid Materials
In the early 1990s Torabinejad and colleagues began testing and experimenting with an MTA compound they developed at Loma Linda University that was essentially a modified Portland cement.54 They found it had significantly better sealing abilities than conventional endodontic materials when used as a root end sealer and in the repair of furcation and lateral root perforations.55-58 This was due, in part, to the fact that MTA sets and functions well in the inherently moist environment in and around a tooth in an in vivo situation. In fact, just as water is an essential component in the setting of Portland cement, it is also required in and enhances the setting of MTA.
While the chemical reactions that occur when MTA is mixed with water are actually quite complex and beyond the scope of this article, the main reactants of MTA (tricalcium and dicalcium silicate and tricalcium aluminate) break down into a number of reaction products, including calcium silicate and the familiar calcium hydroxide.59 The calcium hydroxide dissociates into hydroxyl ions (-OH) resulting in a high pH local environment. Unlike regular calcium hydroxide, which has solubility issues over time, this conglomeration of reaction products has very low solubility and maintains its physical integrity long after placement.
In addition to its low solubility, MTA has several other positive attributes, including high biocompatibility, bioactivity, hydrophilicity, radiopacity, less toxicity than calcium hydroxide, and good sealing ability.60-62 MTA has been shown to promote regeneration of the periodontal ligament, dental pulp, and peri-radicular tissues.61,63 As with calcium hydroxide, there is equivocation in the literature regarding the antibacterial abilities of MTA.64
Although MTA was originally developed primarily for endodontic purposes, such as a root end sealant and for sealing inadvertent furcation and root canal perforations, its use as a potential direct pulp capping agent was quickly recognized.61 Indeed, there is a growing body of scientific evidence, including controlled clinical studies, that has found MTA typically has better clinical outcomes and histologic responses than calcium hydroxide when both are compared head to head in direct pulp capping studies.65-70 While studies indicate MTA is a viable, and perhaps better, option than calcium hydroxide for direct pulp capping procedures, traditional powder/liquid formulations of MTA have significant drawbacks and are not used in most dental offices.36 For example, they are not especially user friendly, require very precise mixing, can cause tooth discoloration, and manipulation and placement can be challenging. In addition, traditional MTA formulations require long setting times (typically 2 to 4 hours), with one study that simulated intraoral conditions finding that the MTA formulation tested (ProRoot® MTA) was still not set after 6 hours.71 Hydration accelerators, such as citric acid, lactic acid, calcium chloride, and calcium lactate gluconate, have been added to some modified MTA formulations (eg, Biodentine®, Septodont, septodontusa.com; MTA Angelus®, Angelus Soluções Odontológicas, angelus.ind.br). This, along with incorporation of the powder and liquid components into capsules that can be triturated (ie, Biodentine) and the manipulation of particle sizes and powder/liquid ratios, as well as other modifications, has significantly decreased the setting time of some MTA formulations to a much more clinically manageable, though still long, 10 to 15 minutes.
Biodentine is designed as a dentin replacement (base) and, according to the manufacturer, has physical properties similar to dentin. It can be used as a dentin substitute under composites for direct and indirect pulp capping, or as an endodontic repair material. One study showed Biodentine induced odontoblast differentiation from pulp progenitor cells.72 Another in vivo study showed complete dentinal bridge formation, as well as an absence of an inflammatory pulp response, in the majority of teeth whose pulps were purposely exposed, pulp capped with Biodentine, and subsequently extracted after 6 weeks for histologic examination.73 Positive clinical outcomes have been reported using Biodentine as a direct pulp capping material.74 While the setting time of some MTA-based materials such as Biodentine has been significantly improved, the still relatively long setting times of 10 to 15 minutes can be problematic. MTA-based resin-modified materials that set on demand via light polymerization may offer an attractive alternative, as having the ability to cure quickly offers a significant clinical advantage. One important consideration regarding such a hybrid product is whether or not the incorporation of light-curable resin components and chemistry into an MTA matrix would in any way compromise the bioactive capabilities and desirable properties of the MTA. While there are promising studies and anecdotal evidence (discussed below) that support such a product, further research is warranted to fully address this important issue.
Development of a Light-Curable Resin-Modified MTA-Based Material
In the early 2000s, chemists Dr. Byoung Suh, Dr. Rui Yin, and David Martin, along with dentist and researcher Dr. Mark Cannon, began developing and experimenting with a single-component hydrophilic resin-modified light-curable MTA-based material. In November 2011, after nearly 10 years of research, the finished product (TheraCal LC®, BISCO) was introduced into the marketplace (according to a personal communication the author had with Dr. Liang Chen, head chemist at BISCO, in July 2017). The company markets this product as a "light-cured resin-modified calcium silicate" (RMCS) formulated to be used as a liner for both direct and indirect pulp capping procedures. The chemistry of the product includes, but is not limited to, approximately 45% by weight MTA (type III Portland cement), calcium oxide, barium zirconate (for radiopacity), fumed silica (thicken agent), strontium glass, bisphenol A-glycidyl methacrylate (bis-GMA), photoinitiators, and polyethylene glycol dimethacrylate (PEGDMA, hydrophilic monomer).
The material is designed to be placed in a thin layer (1-mm increment) and light-polymerized for 20 seconds. Once the resin components of TheraCal LC are light-polymerized the MTA is essentially trapped in the polymer matrix. It is logical to assume that the presence of the resin matrix modifies both the setting mechanism and ion leaching abilities and characteristics of the MTA in TheraCal LC. It is important to remember water is needed for the hydraulic setting reactions and ion releasing abilities of MTA. Because there is no water inherent in TheraCal LC's formulation this water must come from another source. Instructions for the application of TheraCal LC state it should be placed on "visibly moist" dentin. It is possible this water source assists in the initial setting reaction of the MTA. In addition, due to the hydrophilic nature of polymerized TheraCal LC, it is postulated that water/fluids that are inherent in intertubular dentin, dentin tubules, and (in the case of direct pulp capping) the pulp itself diffuse into the polymer matrix over time, reacting with the MTA-releasing calcium and hydroxide ions in the process.
A study by Gandolfi and colleagues found TheraCal LC had higher calcium releasing ability and lower solubility than either ProRoot MTA or Dycal® (Dentsply Sirona) throughout the testing period of 28 days when the samples were stored in water.75 Another study found that samples of TheraCal LC and Dycal (again stored in water) tested for calcium-ion concentration at different time intervals (7, 14, and 21 days) showed TheraCal LC had greater ion release at all time intervals.76 However, there is equivocation in the literature as another comprehensive study, in which a highly sensitive ion detecting technique (ion chromatography) was used, reported significantly higher calcium-ion levels leached from Biodentine compared to that of TheraCal LC (samples stored in both water and physiologic saline solution).77
It is important to note that measurement of ion release from MTA and MTA/hybrid materials such as TheraCal LC typically occurs with samples submerged in water and/or simulated physiologic solutions. While this technique enables comparative ion values for different materials to be ascertained under those conditions, such an abundant water source is not the clinical reality for pulp capping materials that are typically sealed deep inside a cavity preparation and cut off from an external water supply. Other authors and researchers have also raised this critical issue and found ion release for some products to be significantly less than reported when tested under more realistic clinical conditions.78
The ultimate test for any restorative material, of course, is clinical performance-ie, does it work? TheraCal LC has demonstrated the ability to form apatite similar to that of ProRoot MTA when immersed in simulated phosphate-containing body fluid,79 and has also been shown to induce odontoblastic differentiation and mineralization.80 Two in vivo studies have shown that TheraCal LC has the potential to induce reparative dentin and dentin bridge formation when used as a direct and indirect pulp capping agent.81,82 However, one study found TheraCal LC did not have as favorable a pulpal response as ProRoot MTA when used to cap pulpotomies in dogs' teeth.83 It should be pointed out that the company does not advocate the use of Theracal LC for this procedure. One researcher reported an impressive 20 out of 20 teeth with exposed pulps capped with TheraCal LC remained vital after 2 years.84 That same researcher reported 19 out of 20 of those same teeth remained vital after 4 years (according to a personal communication the author had with that researcher, Dr. Scotti Nicola, in January 2016). While positive studies and anecdotal reports of clinical success using MTA/resin hybrid products such as TheraCal LC exist, caution is urged before they can be unequivocally endorsed.
Case Report
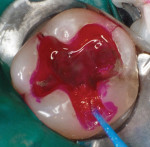
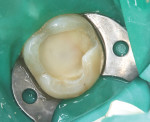
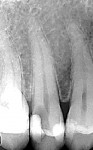
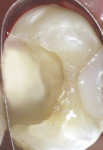
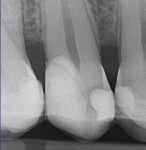
In September 2015 the patient, a 40-year-old man, presented to the office with the chief complaint of cold sensitivity in the upper right quadrant. A radiograph revealed extensive decay on the distal of tooth No. 4 (Figure 7). The tooth was not percussive sensitive but was sensitive when stimulated with cold. After discussion with the patient, the area was anesthetized with articaine, the tooth opened, and the decay visualized. Extensive soft decay was evident and removed with the aid of caries detecting solution, slow-speed round burs, and spoon excavators, until the clinician reached solid dentin that appeared to be caries-free.
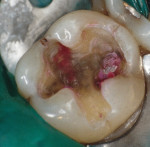
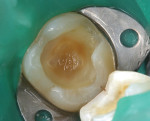
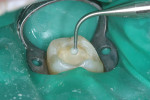
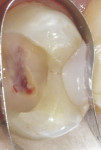
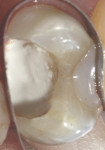
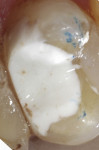
A bleeding pulp exposure was evident (Figure 8). The bleeding was stopped with direct pressure for approximately 60 seconds using a cotton pellet soaked in a 2% chlorhexidine solution (Figure 9). The chlorhexidine-soaked cotton pellet was removed but the dentin not dried. A thin layer of TheraCal LC was placed directly over the exposed pulp and onto the still-moist (with chlorhexidine) dentin surrounding the pulp. The TheraCal LC was then light-polymerized for 20 seconds using a light-curing unit with a power density of approximately 1,000 mW/cm2 (Figure 10). The preparation was then briefly air-dried and a thin layer of Vitrebond was placed over and beyond the borders of the set TheraCal LC and light-polymerized (Figure 11). The tooth was then built up for a future crown using a total-etch adhesive system and self-curing core build-up material (Figure 12).
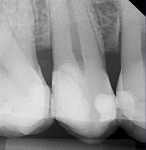
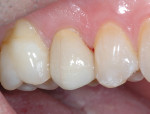
Comparative radiographs taken approximately 14 months apart showed what appeared to be the formation of tertiary (reparative) dentin (Figure 13 and Figure 14). The tooth remained asymptomatic throughout this time period and pulp-tested vital 14 and 22 months after the direct pulp cap was performed. After approximately 22 months the tooth was restored with a full-coverage lithium-disilicate restoration (Figure 15).
Discussion
The author has experienced good clinical success using a combination of products for direct pulp capping procedures that includes chlorhexidine, TheraCal LC, and Vitrebond RMGI liner. Chlorhexidine is a potent antimicrobial, has been shown to inhibit matrix metalloproteinases that may contribute to the breakdown of the adhesive interface over time, does not interfere with the bond strength of adhesive systems, and may have a synergistic effect with calcium hydroxide (one of the reaction products of MTA).39,85 Overlaying TheraCal LC with the RMGI liner and extending the RMGI liner onto dentin beyond the borders of the TheraCal LC offers several potential benefits, including preventing the TheraCal LC from accidently dislodging at some point in the restorative protocol (as some have reported).
In addition, Vitrebond may provide additional antimicrobial activity24,25 and improve the overall seal of the pulp and restoration in general.30-34 This RMGI liner also has a significant amount of water in its formulation. It is possible, though speculative, that some of this water could be absorbed by TheraCal LC (which is hydrophilic) and assist in the setting and ion exchange of the MTA in the material.
Conclusion
While the case presented appears to show dentin bridging and a positive clinical outcome after 22 months, it is premature to draw definitive conclusions as to the exact reason for this short-term success. It may be the combination of products used, and not any specific product, that led to success in this case. Moreover, it may also be that bacteria was managed and the pulp, which likely was reasonably healthy to begin with and still had the capacity for self-repair, was then well sealed allowing normal physiologic responses and healing to occur. Clearly, additional research, including controlled clinical studies, is required to better assess the long-term clinical ramifications of this exciting though relatively new class of dental materials.
About the Author
Gary Alex, DMD
Private Practice, Huntington, New York
References
1. Hilton TJ. Keys to clinical success with pulp capping: a review of the literature. Oper Dent. 2009;34(5):615-625.
2. Thompson V, Craig RG, Curro FA, et al. Treatment of deep carious lesions by complete excavation or partial removal. J Am Dent Assoc. 2008;139(6):705-712.
3. Carrotte PV, Waterhouse PJ. A clinical guide to endodontics-update part 2. Br Dent J. 2009;206(3):133-139.
4. Kuhn E, Reis A, Chibinski AC, Wambier DS. The influence of the lining material on the repair of the infected dentin in young permanent molars after restoration: a randomized clinical trial. J Conserv Dent. 2016;19(6):516-521.
5. Dalplan DM, Casagrande L, Franzom R, et al. Dentin microhardness of primary teeth undergoing partial carious removal. J Clin Pediatr Dent. 2012;36(4):363-367.
6. Chibinski AC, Reis A, Kreich EM, et al. Evaluation of primary carious dentin after cavity sealing in deep lesions: a 10-13 month follow-up. Pediatr Dent. 2013;35(3):e107-e112.
7. Pereira MA, Santos-Junior RBD, Tavares JA, et al. No additional benefit of using a calcium hydroxide liner during stepwise caries removal. J Am Dent Assoc. 2017;148(6):369-376.
8. Pinto AS, de Araujo FB, Franzon R, et al. Clinical and microbiological effect of calcium hydroxide protection in indirect pulp capping in primary teeth. Am J Dent. 2006;19(6):382-386.
9. Marchi JJ, de Araujo FB, Froner AM, et al. Indirect pulp capping in the primary dentition: a four year follow-up study. J Clin Pediatr Dent. 2006;31(2):68-71.
10. Evidenced-based review of clinical studies on indirect pulp capping. J Endod. 2009;35(8):1147-1151.
11. Biorndal L, Reit C, Markvart M, et al. Treatment of deep caries lesions in adults: randomized clinical trials comparing stepwise vs. direct complete excavation, and direct pulp capping vs. partial pulpotomy. Eur J Oral Sci. 2010;118(3):290-297.
12. Fagundes TC, Barata TJ, Prakki A, et al. Indirect pulp treatment in a permanent molar: case report of a 4-year follow-up. J Appl Oral Sci. 2009;17(1):70-74.
13. Weber CM, Alves LS, Maltz M. Treatment decisions for deep carious lesions in the Public Health Service in Southern Brazil. J Public Health Dent. 2011;71(4):265-270.
14. Oen KT, Thompson VP, Vena D, et al. Attitudes and expectations of treating deep caries: a PEARL Network survey. Gen Dent. 2007;55(3):197-203.
15. Koopaeei MM, Inglehart MR, McDonald N, Fontana M. General dentists', pediatric dentists', and endodontists' diagnostic assessment and treatment strategies for deep carious lesions: a comparative analysis. J Am Dent Assoc. 2017;148(2):64-74.
16. MaComb D. Caries-detector dyes-how accurate and useful are they? J Can Dent Assoc. 2000;66(4):195-198.
17. Javaheri M, Maleki-kambakhsh S, Etemad-Moghadam S. Efficacy of two caries detector dyes in the diagnosis of dental caries. J Dent (Tehran). 2010;7(2):
18. Wei S, Sadr A, Shimada Y, Tagami J. Effect of caries-affected dentin hardness on the shear bond strength of current adhesives. J Adhes Dent. 2008;10(6):431-440.
19. Masatoshi N, Sitthikorn K, Taweesak P, Junji T. Bonding to caries-affected dentin. Jap Dent Sci Review. 2011;47(2):102-114.
20. Costa CA, Giro EM, Nascimento AB, et al. Short-term evaluation of the pulpo-dentin complex response to a resin-modified glass-ionomer cement and a bonding agent applied in deep cavities. Dent Mater. 2003;19(8):739-746.
21. de Souza Costa CA, Teixeira HM, Lopes do Nascimento AB, Hebling J. Biocompatibility of resin-based dental materials applied as liners in deep cavities prepared in human teeth. J Biomed Mater Res B Appl Biomater. 2007;81(1):175-184.
22. Yoshida Y, Van Meerbeek B, Nakayama Y, et al. Evidence of chemical bonding at biomaterial-hard tissue interfaces. J Dent Res. 2000;79(2):709-714.
23. Mitra SB. In vitro fluoride release from a light-cured glass-ionomer liner/base. J Dent Res. 1991;70(1):75-78.
24. DeSchepper EJ, Thrasher MR, Thurmond BA. Antibacterial effects of light-cured liners. Am J Dent. 1989;2(3):74-76.
25. Scherer W, Lippman N, Kaim J, et al. Antimicrobial properties of VLC liners. J Esthet Dent. 1990;2(2):31-32.
26. Powell LV, Gordon GE, Johnson GH. Clinical comparison of Class V composite and glass ionomer restorations. Am J Dent. 1992;5(5):249-252.
27. Croll TP. Visible light-hardened glass-ionomer cement base/liner as an interim restorative material. Quintessence Int. 1991;22(2):137-141.
28. Mitra SB, Conway WT. Coefficient of thermal expansion of some methacrylate modified glass ionomers. J Dent Res. 1994;73:218. Abstract 944.
29. 3M ESPE. Technical Product Profile: Vitrebond. https://multimedia.3m.com/mws/media/402237O/vitrebondtm-plus-light-cure-liner-base.pdf. Accessed February 1, 2018.
30. Wibowo G, Stockton L. Microleakage of Class II composite restorations. Am J Dent. 2001;14(3):177-185.
31. Gupta S, Khinda VIS, Grewal N. A comparative study of microleakage below cemento-enamel junction using light cure and chemically cured glass ionomer cement liners. J Indian Soc Pedod Prev Dent. 2002;20:(4)158-164.
32. Sampaio PC, de Almeida Júnior AA, Francisconi LF, et al. Effect of conventional and resin-modified glass-ionomer liner on dentin adhesive interface of Class I cavity walls after thermocycling. Oper Dent. 2011;36(4):403-412.
33. Aggarwal V, Singla M, Yadav S, Yadav H. Effect of flowable composite liner and glass ionomer liner on class II gingival marginal adaptation of direct composite restorations with different bonding strategies. J Dent. 2014;42(5):619-625.
34. Kasraei S, Azarsina M, Majidi S. In vitro comparison of microleakage of posterior resin composites with and without liner using two-step etch-and-rinse and self-etch dentin adhesive systems. Oper Dent. 2011;36(2):213-221.
35. Weston J. Use of a resin-modified glass-ionomer (RMGI) liner in conservative direct treatment of deep caries. Compend Contin Educ Dent. 2015;36(1):42-45.
36. Child PL, Cannon ML. Pulp capping: improving the prognosis. DentalTown. January 2015:52-56.
37. do Nascimento AB, Fontana UF, Teixeira HM, Costa CA. Biocompatibility of a resin-modified glass-ionomer cement applied as pulp capping in human teeth. Am J Dent. 2000;13(1):28-34.
38. Jiang RD, Lin H, Zheng G, et al. In vitro dentin barrier cytotoxicity testing of some dental restorative materials. J Dent. 2017;58:28-33.
39. Alex G. Is total-etch dead? Evidence suggests otherwise. Compend Contin Educ Dent. 2012;33(1):12-25.
40. Alex G. Adhesive considerations in the placement of direct composite restorations. Functional Esthetics and Restorative Dentistry. 2007;1(1):20-25.
41. Agnes A, Long A, Best S, Lobner D. Pulp capping materials alter the toxicity and oxidative stress induced by composite resins in dental pulp culture. European Endod J. 2017;2(1):11-16.
42. Athanassiadis B, Abbott PV, Walsh LJ. The use of calcium hydroxide, antibiotics and biocides as antimicrobial medicaments in endodontics. Aust Dent J. 2007;52(suppl 1):S64-S82.
43. Sawicki L, Pameijer CH, Emerich K, Adamowicz-Klepalska B. Histological evaluation of mineral trioxide aggregate and calcium hydroxide in direct pulp capping of human immature permanent teeth. Am J Dent. 2008;21(4):262-266.
44. Schröder U. Effects of calcium hydroxide-containing pulp-capping agents on pulp cell migration, proliferation, and differentiation. J Dent Res. 1985;64 spec no:541-548.
45. Aparecida-Giro EM, Gondim JO, Hebling J, de Souza Costa CA. Response of human dental pulp to calcium hydroxide paste preceded by a corticosteroid/antibiotic dressing agent. Braz J Oral Sci. 2010;9(3):337-344.
46. Arthur M. Schlesinger Quotes. BrainyQuote.com. Xplore Inc, 2018. https://www.brainyquote.com/quotes/arthur_m_schlesinger_109503. Accessed February 1, 2018.
47. Cox CF, Hafez AA, Akimoto N, et al. Biological basis for clinical success: pulp protection and the tooth-restoration interface. Pract Periodontics Aesthet Dent. 1999;11(7):819-826.
48. Cox CF, Sübay RK, Ostro E, et al. Tunnel defects in dentin bridges: their formation following direct pulp capping. Oper Dent. 1996;21(1):4-11.
49. Min KS, Park HJ, Lee SK, et al. Effect of mineral trioxide aggregate on dentin bridge formation and expression of dentin sialoprotein and heme oxygenase-1 in human dental pulp. J Endod. 2008;34(6):666-670.
50. Khayat A, Abbasi A, Tanideh N. A comparative study of dentin bridge formation following pulpotomy using calcium hydroxide and MTA in young dogs. Iranian J of Veterinary Res. 2004;5(2):47-54.
51. Yalcin M, Arslan U, Dundaar A. Evaluation of antibacterial effects of pulp capping agents with direct contact test method. Eur J Dent. 2014;8(1):95-99.
52. Barthel CR, Rosenkranz B, Leuenberg A, Roulet JF. Pulp capping of carious exposures: treatment outcome after 5 and 10 years: a retrospective study. J Endod. 2000;26(9):525-528.
53. History of Portland Cement. Portland Cement Association website. https://www.concretethinker.com/detail/History-Portland-Cement.aspx. Accessed February 1, 2018.
54. Oliveira MG, Xavier CB, Demarco FF, et al. Comparative chemical study of MTA and Portland cements. Braz Dent J. 2007;18(1):3-7.
55. Torabinejad M, Watson TF, Pitt Ford TR. Sealing ability of a mineral trioxide aggregate when used as a root end filling material. J Endod. 1993;19(12):591-595.
56. Lee SJ, Monsef M, Torabinejad M. Sealing ability of a mineral trioxide aggregate for repair of lateral root perforations. J Endod. 1993;19(11):541-544.
57. Pitt Ford TR, Torabinejad M, McKendry DJ, et al. Use of mineral trioxide aggregate for repair of furcal perforations. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1995;79(6):756-763.
58. Torabinejad M, Hong CU, McDonald F, Pitt Ford TR. Physical and chemical properties of a new root-end filling material. J Endod. 1995;21(7):349-553.
59. Camilleri J. The chemical composition of mineral trioxide aggregate. J Conserv Dent. 2008;11(4):141-143.
60. Agnes A, Long A, Best S, Lobner D. Pulp capping materials alter the toxicity and oxidative stress induced by composite resins in dental pulp culture. Eur Endod J. 2017;2(1):11.
61. Torabinejad M, Chivian N. Clinical applications of mineral trioxide aggregate. J Endod. 1999;25(3):197-205.
62. Tawil PZ, Duggan DJ, Galicia JC. MTA: a clinical review. Compend Contin Educ Dent. 2015;36(4):247-264.
63. Bartols A, Roussa E, Walther W, Dörfer CE. First evidence for regeneration of the periodontium to mineral trioxide aggregate in human teeth. J Endod. 2017;43(5):715-722.
64. Komabayashi T, Zhu Q, Eberhart R, Imai Y. Current status of direct pulp-capping materials for permanent teeth. Dent Mater J. 2016;35(1):1-12.
65. Leye Benoist F, Gaye Ndiaye F, Kane AW, et al. Evaluation of mineral trioxide aggregate (MTA) versus calcium hydroxide cement (Dycal) in the formation of a dentine bridge: a randomised controlled trial. Int Dent J. 2012;62:33-39.
66. Hilton TJ, Ferracane JL, Mancl L, et al. Comparison of CaOH with MTA for direct pulp capping—a PBRN randomized clinical trial. J Dent Res. 2013;92(suppl 7):16S-22S.
67. Mente J, Hufnagel S, Leo M, et al. Treatment outcome of mineral trioxide aggregate or calcium hydroxide direct pulp capping: long-term results. J Endod. 2014;40(11):1746-1751.
68. Dammaschke T, Wolff P, Sagheri D, et al. Mineral trioxide aggregate for direct pulp capping: a histologic comparison with calcium hydroxide in rat molars. Quintessence Int. 2010;41(2):e20-e30.
69. Zhu C, Ju B, Ni R. Clinical outcome of direct pulp capping with MTA or calcium hydroxide: a systematic review and meta-analysis. Int J Clin Exp Med. 2015;8(10):17055-17060.
70. Sawicki L, Pameijer CH, Emerich K, Adamowicz-Klepalska B. Histological evaluation of mineral trioxide aggregate and calcium hydroxide in direct pulp capping of human immature permanent teeth. Am J Dent. 2008;21(4):262-266.
71. Charland T, Hartwell GR, Hirschberg C, Patel R. An evaluation of setting time of mineral trioxide aggregate and EndoSequence Root Repair Material in the presence of human blood and minimal essential media. J Endod. 2013;39(8):1071-1072.
72. About I, Laurent P, Tecles O. Bioactivity of Biodentine™: a Ca3SiO5-based dentin substitute. J Dent Res. 2010;89:165-169.
73. Nowicka A, Lipski M, Parafiniuk M, et al. Response of human dental pulp capped with Biodentine and mineral trioxide aggregate. J Endod. 2013;39(6):743-747.
74. Strassler HE, Levin R. Biodentine tricalcium-silicate cement. Inside Dentistry. 2011;7(10):98-100.
75. Gandolfi MG, Siboni F, Prati C. Chemical-physical properties of TheraCal, a novel light-curable MTA-like material for pulp capping. Int Endod J. 2012;45(6):571-579.
76. Chaudhari WA, Jain RJ, Jadhav SK, et al. Calcium ion release from four different light-cured calcium hydroxide cements. Endodontology. 2016;28:114-118.
77. Camilleri, J. Hydration characteristics of Biodentine and Theracal used as pulp capping materials. Dent Mater. 2014;30(7):709-715.
78. Camilleri J, Laurent P, About I. Hydration of Biodentine, Theracal LC, and a prototype tricalcium silicate–based dentin replacement material after pulp capping in entire tooth cultures. J Endod. 2014;40(11):1846-1854.
79. Gandolfi MG, Siboni F, Taddei P, et al. Apatite forming ability of TheraCal pulp capping material. J Dent Res. 2011;90(spec iss A). Abstract 2521.
80. Lee BN, Lee BG, Chang HS, et al. Effects of a novel light-curable material on odontoblastic differentiation of human dental pulp cells. Int Endod J. 2017;50(5):464-471.
81. Cannon M, Gerodias N, Viera A, et al. Primate pulpal healing after exposure and TheraCal application. J Clin Pediatr Dent. 2014;38(4):333-337.
82. Menon NP, Varma BR, Janardhanan S, et al. Clinical and radiographic comparison of indirect pulp treatment using light-cured calcium silicate and mineral trioxide aggregate in primary molars: A randomized clinical trial. Contemp Clin Dent. 2016;7(4):475-480.
83. Lee H, Shin Y, Kim SO, et al. Comparative study of pulpal responses to pulpotomy with ProRoot MTA, RetroMTA, and TheraCal in dogs’ teeth. J Endod. 2015;41(8):1317-1324.
84. Nicola S, Petrolo F, Allegra C, et al. 2-years clinical outcome of direct pulp capping with resin-based materials. J Dent Res. 2015;94(spec iss A). Abstract 3709.
85. Priyadarshini BM, Selvan ST, Narayanan K, Fawzy AS. Characterization of chlorhexidine-loaded calcium-hydroxide microparticles as a potential dental pulp-capping material. Bioengineering (Basel). 2017;4(3). pii:E59.