Opioid, NSAID, and OTC Analgesic Medications for Dental Procedures: PEARL Network Findings
Ying Jo Wong, MS, DMD; James Keenan, DDS, MS; Keith Hudson, DDS; Henry Bryan, DDS; Frederick Naftolin, MD, PhD; Van P. Thompson, DDS, PhD; Ronald G. Craig, DMD, PhD; Don Vena; Damon Collie, MSHS; Hongyu Wu, MPH; Abigail G. Matthews, PhD; Ashley C. Grill, MPH, RDH; and Frederick A. Curro, DMD, PhD
Abstract:
Objective: Dental treatment is often categorized as a moderately or severely painful experience; however, no clinical data reported by the patient and dentist currently exists to support this degree of pain. This has contributed possibly to the overprescribing of analgesics, in particular the opioid class of medications. The primary objective of the study was to document the dentists’ postprocedural prescriptions and recommendations for analgesic medications and their effectiveness for a 5-day period. Medications prescribed or recommended in the patient-reported outcomes included: opioid, non-steroidal anti-inflammatory drugs (NSAIDs), and over-the-counter (OTC) analgesics. Methods: This study used both dentist and patient responses to evaluate the use of opioid, NSAID, and OTC recommended or prescribed analgesics following one of seven classes of dental procedures encompassing over 22 specific coded procedures thought to elicit pain. The patient-centered study included a 5-day postprocedural patient follow-up assessment of the medication’s effectiveness in relieving pain. Results: Baseline questionnaires were completed by 2765 (99.9%) of 2767 eligible patients, and 2381 (86%) patients responded to the Day 5 follow-up questionnaires. Conclusion: The data suggest NSAIDs, both OTC and prescribed dosages, may be a sufficient analgesic to treat most postoperative dental pain. Clinical judgment as to the use of an opioid should include the physiological principles related to the pharmacology of pain and inflammation and may include a central effect.
This study originated as an outcome of a joint meeting with the National Institute on Drug Abuse and participants in dental practice-based research networks.1 The meeting raised questions that were used to frame the current study and identified the need for analgesic data from dental procedures to assess the pain levels experienced by patients and what dentists anticipated. The literature describes surveys of dentists’ prescribing patterns of analgesics; however, no clinical studies were found that accurately relate the prescribing of analgesic medications to the patient’s pain response from a dental procedure.2-4 Although pain can bring the most reluctant person to seek relief from the dentist, dental treatment itself has been considered traditionally by dentists and patients to be painful.
In 2013, the Office of Inspector General drafted a report released in 2015 on opioid prescribing by healthcare providers, which included dentists.5 However, the degree of the pain involved with dental procedures, to date, with input from both the patient and dentist has not been determined. Recently, the use of opioid analgesics has come under scrutiny due to overprescribing by medical and dental practitioners. This overprescribing has resulted unintentionally in increased misuse and abuse, leading to addiction and deaths, and the rescheduling of certain opioids, eg, hydrocodone, from Schedule III to Schedule II.6 Of all Medicare Part D participants, dentists ranked third in the number of healthcare professionals writing prescriptions for Schedule III drugs, raising questions regarding how painful dental procedures truly are and whether opioids are being prescribed appropriately.5 States such as Pennsylvania7 are now responding to this crisis by instituting dental guidelines for the use of opioids.
Objective
The primary objective of the study was to document the dentists’ patterns regarding postprocedural prescriptions and recommendations for analgesic medication and the effectiveness of these medications and the side effects associated with their use. Patient-reported outcomes for specific dental procedures would be used for the analysis. Medications were: opioids, non-steroidal anti-inflammatory drugs (NSAIDs), and over-the-counter (OTC) analgesics. The study included a 5-day postprocedural patient follow-up to determine how accurately the dentist had assessed and treated pain.
Methods
The study was conducted by the Practitioners Engaged in Applied Research and Learning (PEARL) Network, a practice-based research network (PBRN), adhering to the principles of good clinical practice (GCP).8 GCP refers to the standard for design, conduct, recording, auditing, and reporting of studies. Since its initiation with a National Institutes of Health/National Institute of Dental and Craniofacial Research (NIH/NIDCR) grant, PEARL has conducted more than 20 clinical studies involving more than 300 dental and medical practitioners. This study utilized general dentists from the PEARL network to recruit patients into the study. All participating dentists were screened for licensure limitations and credentials, and they had been trained in the principles of GCP. Abstracts9-11 for this study have been presented previously.
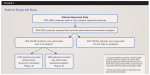
One of the important components of GCP is establishing an audit trail to support the generalizability of the data. Data were monitored using a risk-based approach, ensuring that reporting accuracy met the Emmes Data Coordinating Center (The Emmes Corporation, emmes.com) and PEARL network’s joint data collection requirements (21 CFR Part 11 compliant). The study design was multisite, prospective, observational, and patient centered. PEARL Network Practitioner-Investigators (P-Is) documented postprocedural analgesic prescribing patterns for both over-the-counter (OTC) and prescription (Rx) analgesics associated with 1 of 7 classes of dental procedures encompassing more than 22 specific coded procedures12 deemed by PEARL Network P-Is to require the use of local anesthetic for the procedure and thought to elicit postoperative pain. The 7 classes of dental procedures (Appendix 1 at dentalaegis.com/go/cced1013) included in the study were: a crown preparation in which enamel was removed from the tooth in preparation for a crown, extraction, endodontic therapy, periodontal surgery, pulp capping (direct–the exposed pulp was covered with a sedative dressing to promote healing), pulp capping (indirect–the nearly exposed pulp was covered with a protective dressing to promote healing), onlay restoration (a type of dental restoration that fits primarily over the cusps of the tooth), and abscess treatment (included incision and draining). Procedures were selected on the basis of pain emanating from the pulpal and/or supporting tissues. Eligibility criteria included presence of erupted second molars, having fully erupted permanent dentition, and the patient’s capacity to judge pain level. A parent or legal guardian provided consent for patients younger than age 18 years, and those older than 18 assented by informed consent. A flowchart of the study treatment and follow-up is depicted (Figure 1). Participants meeting the eligibility criteria provided consent, and, prior to the start of procedures, completed a patient baseline questionnaire with a 5-point verbal rating scale (VRS-5) (1 = no pain, 2 = mild pain, 3 = moderate pain, 4 = severe pain, and 5 = very severe pain), and an oral health impact profile (OHIP-14), which is an oral health–related quality-of-life survey.13
The P-Is performed the scheduled treatment according to the respective practice’s standard of care, and then each P-I completed a questionnaire rating his or her expectation of the patient’s postoperative level of pain while the patient was anesthetized and in the absence of analgesic medication. In addition, the P-I recorded whether a recommendation or discharge prescription was given as a “yes” for an analgesic (OTC or Rx) (Figure 1); recorded the medication, patient instructions (including dose and frequency), and the patient’s current medication use; and indicated whether the treatment was performed. If the P-I chose not to prescribe or recommend an analgesic following the procedure, this was noted as “no,” (Figure 1), but the participants continued in the study and recorded any postoperative pain and whether they subsequently took any analgesics for relief. Before leaving the dental offices, the participants were reminded to contact the dentists if they experienced any undue pain or required further evaluation. At postoperative Day 5, the participants completed a questionnaire inquiring about analgesic use, pain relief, and side effects, and took the OHIP-14.
All side effects were reported and reviewed by the Pearl Network Medication Safety Committee and were addressed by familiarizing study P-Is and their practice research coordinators with MedWatch.14 All adverse events in the study were known; therefore, no adverse events were reported to MedWatch. The study was powered to detect a difference in side-effect rate, used as a boundary limit, of 1% for OTC medications and 3% for Rx medications for all procedures. The study protocol was reviewed and approved by the protocol review committee of the NIH/NIDCR and the New York University School of Medicine Institutional Review Board.
Results
A total of 2773 patients was enrolled; 2767 (99.8%) met the coded dental procedure class requirements (Appendix 1) and were eligible (6 patients did not meet the criteria and were excluded from the analysis). Among the 2767 eligible patients, 2765 patients completed baseline questionnaires. The study-design diagram (Figure 1) described the dentist and patient study flow. Dentists reported recommending or prescribing an analgesic for 2504 patients; 1516 (60.5%) were recommended OTC, and 988 (39.5%) were prescribed an analgesic. Dentists wrote 698 prescriptions for opioids and recommended 1254 NSAIDs and 544 other OTC medications (Figure 1).
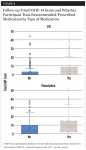
Of the 2381 (86.0%) patients who responded to the Day 5 follow-up questionnaires, 1935 (81.2%) self-reported an analgesic being recommended (Figure 2), and 1192 (50.1%) reported actually taking the recommended pain medication, with pain-medication utilization of 64.5% when there was agreement between dentist and patients. Among patients taking the recommended pain medication, pain-relief and side-effects data were available for 1040 (87.2%) and 1065 (89.3%) patients, respectively.
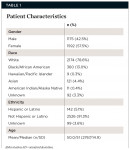
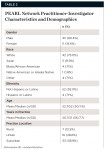
The demographics of the patient distribution (Table 1) was 1175 (42.5%) males and 1592 (57.5%) females. Recruitment of patients represented a broad cross-section of the population and the diversity of the P-Is (Table 2). Of the 56 dentists who enrolled patients in the study, there were 45 (80.4%) who were male and 11 (19.6%) who were female (Table 2). Dentists were representative of the population of dentists in the United States, and the average age of dentists was representative of the PEARL Network as described in previous studies.8 Dentists were in rural, urban, and suburban areas.
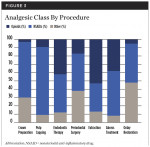
Figure 3 describes the analgesic medication usage for each procedure class. Categories included opioids, NSAIDs, and other (the most common “other” medication was acetaminophen). Opioids were mostly prescribed following extraction (53%), endodontic therapy (42%), and abscess treatment (38%).
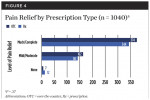
Figure 4 describes the level of pain relief by analgesic type (OTC or Rx) among patients taking the medications. OTC medications consisted of ibuprofen, naproxen, acetaminophen, and aspirin. Rx agents referred to opioids or NSAIDs (where the dosage is higher than the OTC analgesic recommendation). Table 3 describes prescribed medications: OTC, NSAIDs, and opioids. Almost all (98%) of the 1040 respondents experienced some pain relief with OTC and/or Rx analgesic medications (Figure 4). Patients reported no significant difference between Rx and OTC pain relief (P = .37), although we should note that opioids were more often prescribed for procedures for which more intense pain was anticipated.
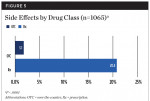
Side effects associated with Rx and OTC medications are described (Figure 5). There were significantly greater number (P < .0001) of side effects reported for the Rx analgesics (20.4%), compared with the number in people (3.1%) who took OTC medications, a 7-fold increase in side effects such as dizziness, nausea, and vomiting (Table 4). Within the Rx group, side effects were reported among 27.5% of participants receiving opioids and 7.5% receiving NSAIDs.
Table 5 describes whether a prescribed/recommended regimen was specified or if patients were instructed to take medication as needed (PRN). Most patients (85%) were advised to take medication PRN. For specified daily dose, the four most common doses were: 200 mg ibuprofen (n = 75, 48.1%), 600 mg ibuprofen (n = 33, 21.2%), 800 mg ibuprofen (n = 25, 16.0%), and 400 mg ibuprofen (n = 10, 6.4%). For PRN single dose, the four most common doses were: 200 mg ibuprofen (n = 651, 70.5%), 800 mg ibuprofen (n = 124, 13.4%), 600 mg ibuprofen (n = 51, 5.5%), and 400 mg ibuprofen (n = 43, 4.7%).
For both Rx and OTC analgesics, prescribed opioids accounted for 691 (27.5%) instances of all analgesic use. There were recommendations for NSAIDs for 1254 (50.2%) patients and 544 (21.8%) patients for “other” (the majority of “other” was acetaminophen, Table 3). Dentists and patients agreed a pain medication was recommended or prescribed in 84% of cases. Miscommunication regarding dentists’ recommendations for a pain medication occurred 10 times more often for OTC than Rx medications and increased with patient age.
Figure 6 describes patients’ OHIP-14 postprocedural quality-of-life scores. A higher OHIP-14 score is associated with greater pain levels. Patients who took a medication reported higher OHIP-14 scores than those who opted not to take an OTC or Rx drug. OTC medication use had lower OHIP-14 values than patients taking Rx drugs. There was no significant difference among the groups.
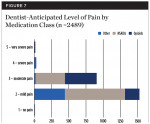
Figure 7 describes opioid analgesic use based on dentist-anticipated levels of pain. Within the dentist-anticipated pain levels, the numbers represent those patients within each pain level who were prescribed opioids: no pain (n = 2/15, 13.3%), mild pain (n = 194/1506, 12.9%), moderate pain (n = 446/916, 48.7%), severe pain (n = 45/47, 95.7%), and very severe pain (n = 4/5, 80%).
Discussion
The PEARL Network originated in 2005 with funding from the NIH/NIDCR.15 It was formed at New York University and has developed into a free-standing, interdisciplinary clinical translational network. Its current mission is to provide an infrastructure for data-driven healthcare with the goal of disseminating information between healthcare providers, patients, payers, and industry.16
This study provides insights into the prescribing patterns of general dentists and how patients respond to routine dental procedures rendered with a local anesthetic with the anticipation of pain. The demographics of the study included urban, suburban, and rural dental settings. The research demonstrated 1021 (98.2%) patients experienced mild or complete pain relief with the use of OTC and/or Rx analgesic medications and 19 (1.8%) experienced no pain relief (Figure 4). Dentists anticipated the level of pain to be mainly mild to moderate, with only 52 cases (2%) in which they anticipated severe or very severe pain (Figure 7). Dentists tended to expect more pain than patients actually experienced; however, the choice of medication prescribed or recommended did not reflect the dentists’ expectations or the patients’ outcomes (Figure 7). The study results did not support the prescribing of opioid medications for the reported procedures in which an anti-inflammatory medication would be more appropriate. The study reported almost 1 in 4 (24.9%) patients enrolled in the study was given prescriptions for opioid medications.
PEARL Network practitioners are screened, vetted, and highly motivated to conduct office-based clinical research. They represent a dedication to quality assurance by allowing a clinical research associate to monitor the progress and create an audit trail of the study. Results from the PEARL Network are most likely a best–case scenario for prescribing practices among general dentists. One explanation for the use of prescription opioids may be due to dentists prescribing analgesics while the patient is still anesthetized and the dentist’s primary concern being postoperative patient comfort. Pain is multidimensional, and opioids are distinctively prescribed when a central component for analgesia is needed to provide pain dissociation, which causes drowsiness. Opioid analgesic treatment of dental pain, as in a third–molar extraction, should be assessed following treatment, and should not exceed 3 days.17 Prescription NSAIDs have a dose range for analgesia (as per the package insert) with higher doses directed to treat inflammation. The choice of medication and its daily dose is dependent on the amount of inflammation incurred by the patient. Prescribers of prolonged opioid therapy should always consider the potential for abuse and the possible consequence of diversion.18 The study supports the recent findings of Maughan et al.19
The results from the OHIP-14 suggest no significant difference in scores between patients recommended OTC and prescribed analgesic medications. Provider and patient communication and adherence were additional findings of the study and miscommunication tended to increase with patient age. The study suggests patients’ adherence may be improved with written directions. This communication gap (16%) between the patient and dentist is an important component in the management of the multidimensionality of pain and in maximizing the placebo response, which historically has demonstrated the benefits of the patient–provider relationship and communication. The patient-centered data reported only 65% of patients took an analgesic medication. Possible explanations as to why patients did not take medications are that the procedures were not that painful (27% of patients reported no postprocedural pain and low OHIP-14 scores), the importance of the placebo effect, good dentist–patient rapport, the patient’s concern related to the risk for medication side effects, and the patient’s ability to implement past learned pain-coping strategies.
Prior to the availability of NSAIDs, prescribing opioids (acetaminophen with codeine) was the only treatment for moderate and severe pain, and provided a central effect of disassociating the patient from the pain, which was viewed as an advantage. This broad approach is no longer within the pharmacologic strategy for the treatment of pain, which practitioners consider targeted therapy. Some situations indicate using opioids as the preferred treatment, and recent US Food and Drug Administration initiatives20 to engage the patient through a patient–provider agreement is gaining acceptance.21 The study distinguishes between what the dentist is estimating the pain level will be and what the patient actually experiences; this provides further insight into conducting analgesic studies by getting responses from both the provider and patient.22 The findings suggest considering the severity of pain, due to routine dental procedures primarily induced by inflammation as an important component to optimize analgesic use. The results propose that pain from dental procedures can be managed by the use of NSAIDs, especially by optimizing their efficacy by applying pharmacokinetic principles on how a drug is delivered to its site of action, metabolized, and eliminated. In addition, patient outcomes can be furthered when prescribers are aware of pharmacologic nuances that NSAIDs within the same chemical class can produce different patient responses.23
Most dental pain is primarily induced by inflammation occurring in a confined localized area such as the periodontal ligament and/or pulpal tissue. Using NSAIDs requires a level of knowledge for proper use to account for blood levels, protein binding, consideration of a loading dose, and a dose that elicits the proper clinical outcome to treat either pain or inflammation.23 The data provided by the process of therapeutic–drug monitoring supports the pharmacodynamics needed to optimize the dosing regimen of NSAIDs, especially in areas of poor vascularization such as bone. The knowledge base for the understanding of the use of analgesic medication goes beyond a therapeutics course, which, over time, has replaced the traditional core courses in pharmacology in many dental and medical schools. Furthermore, as curriculums move away from the science of pharmacology and into therapeutics, there is a loss in the physiological principles bridging the level of pain induced with what the patient actually experiences, most likely resulting in a more potent analgesic.
Limitations
Practice-based clinical studies are primarily designed to fit office logistics and, when compared with controlled clinical studies, have both limitations and advantages.16 A limitation specific to this study was the lack of detail regarding the timing of pain medication relative to the patients’ pain assessment, masking the maximum level of pain the patient may have experienced. The dentist’s assessment of the anticipated pain may not have taken into account the patient’s worst pain level if the patient premedicated themselves prior to the procedure, known as preemptive analgesia. The authors based the design as an office-based clinical study. It did not allow for the dentists to know if the patients had premedicated with analgesics prior to the procedure. Appendix 1, which was included in the protocol, describes procedural codes to set boundary limits for the procedure class, but the dataset generally categorized by procedure class rather than procedure code.
Conclusion
The findings from this study suggest that the prescriptions being written by dentists are overcompensating for the level of pain experienced by the patient and may be contributing to the national problem of the misuse and abuse of opioids and may lead to the increase in deaths.24 This study further suggests that pain from dental treatment may have been historically overrated, by both dentists and patients, and that the image of dental treatment, as it relates to pain, should be reconsidered to improve oral health. Pain should no longer be the image of dentistry.
Acknowledgments
This study was funded by a grant U-01-DE016755 awarded to the New York University College of Dentistry by the National Institute of Dental and Craniofacial Research. ClinicalTrials.gov Identifier: NCT02929602
About the Authors
Ying Jo Wong, MS, DMD
Principal Clinical Practitioner-Investigator
PEARL Network
Currently Clinical Instructor
New York University
College of Dentistry
New York, New York
James Keenan, DDS, MS
Practitioner-Investigator
PEARL Network
Rockaway Beach, NY
Currently Clinical Assistant Professor
New York University
College of Dentistry
New York, New York
Keith Hudson, DDS
Practitioner-Investigator
PEARL Network
Beverly Hills, Michigan
Henry Bryan, DDS
Practitioner-Investigator
PEARL Network
Roseville, Michigan
Frederick Naftolin, MD, PhD
Medical Director
PEARL Network
Professor Department of Obstetrics and Gynecology
Professor Department of Environmental Medicine Obstetrics and Gynecology
Director Reproductive Biology Research New York University
New York, New York
Van P. Thompson, PhD, DDS
Executive Management Team
PEARL Network
Currently Professor
Tissue Engineering and Biophotonics
Kings College
London, United Kingdom
Ronald G. Craig, DMD, PhD
Executive Management Team
PEARL Network
Professor
New York University
College of Dentistry
New York, New York
Don Vena, BS
Principal Investigator
The Emmes Corporation
Rockville, Maryland
Damon Collie, MSHS
Project Manager
The Emmes Corporation
Rockville, Maryland
Hongyu Wu, MPH Biostatistician
The Emmes Corporation
Rockville, Maryland
Abigail G. Matthews, PhD
Biostatistician
The Emmes Corporation
Rockville, Maryland
Ashley C. Grill, MPH, RDH
Clinical Coordinator
PEARL Network
Adjunct Clinical Assistant Professor
Dental Hygiene Programs
New York University
College of Dentistry
New York, New York
Frederick A. Curro, DMD, PhD Director
PEARL Network
Adjunct Clinical Professor
Department of Oral and Maxillofacial Pathology, Radiology, and Medicine
New York University, College of Dentistry
New York, New York
References
1. Opioid Prescribing to Adolescents in Dental Settings. National Institute on Drug Abuse, National Institute of Dental and Craniofacial Research website. February 23, 2009. Rockville, MD. https://archives.drugabuse.gov/meetings/Dental/. Accessed July 18, 2016.
2. Moore RA, Wiffen PJ, Derry S, et al. Non-prescription (OTC) oral analgesics for acute pain - an overview of Cochrane reviews. Cochrane Database Syst Rev. 2015;4(11):CD010794.
3. Derry S, Wiffen PJ, Moore RA. Relative efficacy of oral analgesics after third molar extraction—a 2011 update. Br Dent J. 2011;211(9):419-420.
4. Paulozzi LJ, Strickler GK, Kreiner PW, Koris CM. Controlled substance prescribing patterns—prescription behavior surveillance system, eight states, 2013. MMWR Surveill Summ. 2015;64(9):1-14.
5. Prescribers With Questionable Patterns in Medicare Part D. Bethesda, MD: US Dept of Health and Human Services, Office of Inspector General; 2013.
6. Curro FA. FDA advisory panel assesses hydrocodone prescription writing: every dentist’s responsibility. J Am Dent Assoc. 2013;144(5):462-464.
7. Pennsylvania Guidelines on the Use of Opioids in Dental Practice. Pennsylvania Dental Association and Commonwealth of Pennsylvania. https://www.ddap.pa.gov/Document%20Library/Presecriber%20Guidelines%20on%20the%20Use%20of%20Opioids
%20in%20Dental%20Practice.pdf Naftolin F, et al. The PBRN initiative: transforming new technologies to improve patient care. J Dent Res. 2012;91(7 suppl):12S-20S.
9. Wong YJ, Craig RG, Thompson VP, et al. Analgesic use and patient-reported effectiveness: PEARL interim results. J Dent Res. 2009;88(A):0278.
10. Keenan J, Wong Y, Craig R, et al. Practice-based research assessment of analgesic effectiveness: PEARL Interim findings. J Dent Res. 2010;89(B):2085.
11. Hudson K, Bryan H, Thompson V, et al. Analgesics prescribed/recommended for coded dental procedures: PEARL practitioner profiles. J Dent Res. 2011;90(A):2375.
12. American Dental Association. Current Dental Terminology 2009-2010. Chicago, IL.
13. Slade GD, Spencer AJ. Development and evaluation of the Oral Health Impact Profile. Community Dent Health. 1994;11(1):3–11.
14. US Food and Drug Administration. MedWatch: The FDA Safety Information and Adverse Events Reporting Program website. https://www.fda.gov/Safety/MedWatch/. Accessed July 18, 2016.
15. Tabak L. NIDCR-sponsored practice-based research networks. Presented at PEARL Network Annual Meeting, National Institutes of Health campus, Washington, DC. 2010. Accessed April 10, 2012. https://web.emmes.com/study/pearl/newsletters/spring2010%202.pdf.
16. Curro FA, Robbins DA, Naftolin F, et al. Person-centric clinical trials: defining the N-of-1 clinical trial utilizing a practice-based translational network. Clin Investig (Lond). 2015;5(2):145-159.
17. Tarakji B, Saleh LA, Umair A, et al. Systemic review of dry socket: aetiology, treatment, and prevention. J Clin Diagn Res. 2015;9(4):ZE10-ZE13.
18. Volkow ND, McLellan TA. Curtailing diversion and abuse of opioid analgesics without jeopardizing pain treatment. JAMA. 2011;305(13):1346-1347.
19. Maughan BC, Hersh EV, Shofer FS, et al. Unused opioid analgesics and drug disposal following outpatient dental surgery: a randomized controlled trial [published online ahead of print September 16, 2016]. Drug Alcohol Depend. doi: 10.1016/j.drugalcdep.2016.08.016.
20. Safe use initiative. Food and Drug Administration website. https://www.fda.gov/Drugs/DrugSafety/SafeUseInitiative/. Updated August 20, 2015. Accessed October 12, 2016.
21. Kraus CN, Baldwin AT, Curro FA, McAllister RG Jr. Clinical implications of patient provider agreements in opioid prescribing. Curr Drug Saf. 2015;10(2):159-164.
22. Guidance for Industry Patient-Reported Outcome Measures: Use in Medical Product Development to Support Labeling Claims. Bethesda, MD: US Department of Health and Human Services. December 2009. https://www.fda.gov/downloads/Drugs/.../Guidances/UCM193282.pdf. Accessed October 13, 2016.
23. Brunton L, Chabner B. Goodman and Gilman’s The Pharmacological Basis of Therapeutics. 12th ed. New York, NY: McGraw-Hill. 2011.
24. Zosel A, Bartelson BB, Bailey E, et al. Characterization of adolescent prescription drug abuse and misuse using the Researched Abuse Diversion and Addiction-Related Surveillance (RADARS®) System. J Am Acad Child Adolesc Psychiatry. 2013;52(2):196-204.e2.