Subjective Assessment of Enamelon® Preventive Treatment Gel in a Self-Reported Dry-Mouth Population
Jeffery L. Milleman, DDS, MPA; Kimberly R. Milleman, RDH, BSEd, MS; Sylvia L. Santos, RDH, MS, CCRP; Howard M. Proskin, PhD; Kirstin K. Battershell, RN, MBA; and James C. DiMarino, DMD, MSEd
Abstract
Background: The efficacy of stannous fluoride toothpastes is well established for reducing caries, plaque, and gingivitis and relieving the discomfort of dentin hypersensitivity. Management of dry mouth may include relief remedies in addition to usual oral hygiene methods to maintain oral health and improve quality of life. This 6-week, single-blind, randomized, two-period crossover clinical study was designed to evaluate the oral tolerance and potential of Enamelon® Preventive Treatment Gel (EPTG), with 0.4% stannous fluoride, to relieve subject-perceived dry-mouth symptoms in a self-reported dry-mouth population, after 14 days of use, compared to a marketed over-the-counter (OTC) artificial saliva gel product (Biotene® Oral Balance Gel [BOBG]).
Methods: Following a 7-day washout period, 52 qualified subjects with self-reported dry-mouth symptoms received EPTG or BOBG for once-daily use for 14 days. All subjects brushed each morning and evening with a standard fluoride toothpaste (Sensodyne® ProNamel). Each evening and following brushing with the Sensodyne ProNamel, subjects used their assigned gel product (EPTG or BOBG) as directed. On Days 1, 8, and 15, subjects received an oral examination and assessed relief of dry-mouth symptoms using a product-performance questionnaire (PPQ). Procedures were repeated with the alternate product for another 14-day period, following a 7-day washout. Data for each efficacy endpoint were analyzed using crossover ANOVA model.
Results: No treatment-related adverse events were reported in this study, and both products were well tolerated by the subjects. Compared to pre-study ratings of usual dry-mouth remedies, both BOBG and EPTG significantly reduced dry-mouth symptoms following 14 days of use. BOBG was statistically significantly better than EPTG in relieving many of the principal dry-mouth symptoms such as providing immediate relief, having an immediate moisturizing and immediate lubricating effect, feeling comfortable in the mouth, soothing the mouth, effectively lubricating the mouth, and protecting the mouth from drying out (P < .05). Compared with subjects’ ratings of their usual dry-mouth remedies, EPTG also provided relief of several dry-mouth symptoms at both Days 8 and 15 (P < .05).
Conclusion: Based on subjective measures of dry mouth and compared with pre-study ratings of usual remedies, subjects perceived that EPTG helped to manage symptoms such as relieving the discomfort of dry mouth, immediately moisturizing and lubricating, effectively lubricating, protecting from drying out, and providing long-lasting moisturization and long-lasting lubrication, and was not irritating to dry-mouth tissues. BOBG, the positive control, was significantly better than EPTG in relieving dry-mouth symptoms over a 2-week period and was not irritating.
Practical Implications: Patients with dry-mouth symptoms may benefit from daily use of a non-irritating, OTC fluoride preventive treatment gel product to relieve symptoms while also reducing the risks of developing dental caries, demineralization, dentin hypersensitivity, and gingivitis.
The purpose of this two-period crossover study was to evaluate the safety and efficacy of Enamelon® Preventive Treatment Gel (EPTG) (Premier Dental Products Co., www.premusa.com) in relieving subject-perceived dry-mouth symptoms in a dry-mouth population after 14 days of use, compared to Biotene® Oral Balance, Dry Mouth Moisturizing Gel (BOBG) (GlaxoSmithKline Consumer Healthcare, www.gsk.com).
Xerostomia is the chronic and subjective sensation of dry mouth and, in most cases, is the result of low salivary flow or salivary gland hypofunction.1,2 The major causes of xerostomia are xerogenic medications, such as, but not limited to: attention-deficit/hyperactivity disorder medications, antibiotics, anticoagulants, antidiarrheal/gastric medications, some anti-ulcer medications and gastroesophageal reflux disease medications, antidepressants, antihistamines, antihypertensives, antipsychotic drugs, antiretrovirals, bronchodilators, diuretics, hypoglycemics, levothyroxine, multivitamins and supplements, non-steroidal anti-inflammatory drugs, pain medications (some narcotics and opiates), urinary incontinence drugs, and steroid inhalers, as well as radiation therapy to the head and neck region and systemic conditions such as Sjögrens syndrome and type 2 diabetes.1,3,4
Chronic xerostomia significantly increases the risk for experiencing dental caries, demineralization, tooth sensitivity, gingival inflammation/gingivitis, dry-mouth discomfort, ulceration of mucosal tissues, candidiasis, and other oral diseases that may affect quality of life negatively.5 Individuals with xerostomia may be at high risk for dental caries because of their compromised salivary buffering and remineralization capabilities. Dry-mouth sufferers may also take measures to relieve their dry-mouth symptoms such as frequently using sugared mints and candies, which further increases their risk for caries.6 Chronic dry mouth impacts oral-health–related quality of life through the effects of difficulties with swallowing, chewing, and sometimes even speaking.2,6,7 Other complaints of dry-mouth sufferers include taste disturbance, halitosis, stomatodynia, and intolerance of acidic or spicy foods.8,9
Management of dry-mouth symptoms can be multifaceted and may include use of symptom-relief remedies paired with a preventive oral-hygiene program to help reduce the risk for developing dental caries, demineralization, dentin hypersensitivity, and gingivitis.5,10,11 Easy remedies may include proper hydration, avoidance of irritating dentifrices and crunchy/hard foods, and the use of sugar-free chewing gums/candy to help stimulate saliva production. Other therapies for those with little or no salivary gland function include mucosal lubricants, saliva substitutes (artificial saliva), and saliva stimulants.4 BOBG, a saliva substitute, has been shown to improve dry-mouth symptoms and improve dry-mouth–related quality of life in patients with self-reported dry mouth and in patients with post-radiotherapy xerostomia.12-14
The prevalence of xerostomia in the general population has been estimated to be as high as 46%, afflicting both young and elderly individuals.4,6,7,15 This has generated a market for various over-the-counter (OTC) products for dry mouth, including rinses, lozenges, toothpastes, sprays, gels, and sugarless chewing gums. A rational approach to help manage the symptoms of dry mouth while also preventing the oral complications of the condition would be to develop a single OTC product targeting these benefits.
EPTG contains 0.4% stannous fluoride and delivers amorphous calcium phosphate (ACP). When used once daily, it helps prevent dental caries. When used twice daily, EPTG also helps reduce/prevent gingivitis and dentin hypersensitivity.16,17 In vitro data show that EPTG provides a greater reduction in enamel solubility, thus preventing demineralization of the tooth surfaces when exposed to acid challenges,18 while also providing greater fluoride uptake than other fluoride-containing products on the market, including a prescription-strength fluoride dentifrice with five times the amount of fluoride (5,000-ppm fluoride).19 The gel is formulated without sodium lauryl sulfate (SLS), a common irritant to dry oral mucosa.20 The nonabrasive gel contains patented Ultramulsion® technology and includes a combination of slowly dissolving muco-adhesive copolymers. The ingredients in EPTG help to coat the oral mucosa and are retained in the mouth to provide a long-lasting moisturizing and lubricating effect. Other commercially available products with stannous fluoride are not known to produce dry-mouth benefits.
This study was designed to evaluate the oral tolerance and potential of EPTG to relieve subject-perceived dry-mouth symptoms in a self-reported dry-mouth population after 14 days of use, compared to a marketed OTC artificial saliva gel product.
Methods
Prior to the initiation of this study, the study protocol, informed consent document, and subject instructions received ethical review and approval from the US Investigational Review Board (Miami, FL) (U.S.IRB2015SRI/14). The study was conducted in accordance with the International Conference on Harmonization Good Clinical Practice guidelines (ICH GCP), and all subjects signed an informed consent prior to entering the study. At the screening visits, subjects were qualified for participation based on self-reported dry-mouth symptoms according to at least one positive response on the dry-mouth inventory (DMI) and if they were currently taking at least one drug associated with causing salivary hypofunction or xerostomia (eg, anxiolytics, anorexiants, anti-asthmatics, anticholinergics, antidepressants, antiemetics, antihistamines, antihypertensives, antiparkinsonians, antipsychotics, decongestants, diuretics, and sedatives) and planned to continue taking the prescribed medication(s) for at least 1 week prior to the study and for the duration of the trial. Subjects were excluded from participation if they had known allergy to any ingredient in the study products, were pregnant or breastfeeding, had history of oral cancer or head and neck radiotherapy, or history of primary or secondary Sjögrens syndrome.
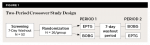
The 6-week, randomized, two-period crossover clinical study included a screening visit followed by a 7-day washout period, a 14-day treatment period (Period 1), a 7-day washout period, and a second 14-day treatment period (Period 2). Subjects assigned to EPTG in Period 1 used the positive control, a commercially available dry-mouth gel product, BOBG, in Period 2; subjects assigned to BOBG in Period 1 received EPTG in Period 2 (Figure 1). During the 7-day washout phase between Periods 1 and 2, subjects brushed only with Sensodyne® ProNamel (SPN) Daily Fluoride toothpaste (GlaxoSmithKline Consumer Healthcare) because it is free of SLS. The purpose of this second washout period was to allow the effects of the Period 1 treatment product to dissipate before starting the Period 2 treatment assignments and included the SLS-free toothpaste to minimize a potential source of soft-tissue irritation.
When assigned to EPTG, subjects brushed each morning in their usual manners with a soft toothbrush and with SLS-free fluoride toothpaste (SPN) and rinsed thoroughly with water. Each evening subjects brushed with the SLS-free fluoride toothpaste (SPN) in their usual manners, rinsed thoroughly with water, then placed approximately a full ribbon of EPTG on their toothbrushes and brushed thoroughly, allowing the gel to remain on their teeth for 1 minute, and then expectorated. Subjects were instructed to not swallow EPTG and to not rinse, eat, or drink for 30 minutes after brushing. When subjects were assigned to the positive control group, they brushed each morning in their usual manners with the SLS-free fluoride toothpaste (SPN) and rinsed thoroughly with water. For the evening regimen, subjects brushed with the SLS-free fluoride toothpaste (SPN) in their usual manners, rinsed thoroughly with water, then placed approximately a half-inch length of BOBG directly on the tongue and spread thoroughly. Subjects were instructed to not spit out the gel. All subjects were prohibited from using any other oral-hygiene products, except for dental floss or an interdental aid if it was part of their normal routine. Throughout each test period, subjects maintained daily diaries to document their compliance with use of their assigned home care products.
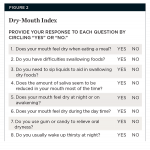
Subjects presented to the research clinic for seven visits: screening; Period 1 - Days 1, 8, and 15; and Period 2 - Days 1, 8, and 15. At the screening visit, subjects completed the following activities: signed an informed consent document; provided demographics and medical/dental history; received an oral examination; listed their current remedies for managing their dry-mouth symptoms; completed a DMI1,21 confirming their subject complaints of dry-mouth condition; and completed a 16-question product performance questionnaire (PPQ) assessing their usual dry-mouth remedies in managing their dry-mouth symptoms. The DMI, which has been used routinely in clinical studies evaluating dry-mouth symptoms, assessed subject impressions of oral dryness and oral functions (Figure 2).1,12,21-24
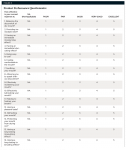
The PPQ consisted of a series of questions asking the subjects to rate the performance, benefits, and attributes of their usual remedies and the test materials following 7 and 14 days of using their assigned treatment. Post-product use questionnaires, such as the PPQ, have been used to measure the efficacy of dry-mouth symptom relief with various treatment remedies such as rinses, gels, and sprays. Currently, there is no known objective standard for the evaluation of effective treatments for dry mouth, particularly because there is a large variation of salivary flow rates in individuals for xerostomia to even occur. Hence, measures of treatment efficacy have relied on subjective assessments using post-product use questionnaires.12,22,23,25-27 For this study, the subjects rated their perceptions using a five-point scale of “poor” to “excellent” (Figure 3).
Following the screening visit, a 7-day washout period occurred, during which all subjects brushed with a regular soft toothbrush and regular fluoride toothpaste (Crest® Regular, The Procter & Gamble Co., www.pg.com) to permit the effects of the previously used home care products to dissipate before starting the first treatment period. Subjects returned to the clinic site for the Period 1-Day 1 visit following the 7-day washout period. Prior to each study visit, subjects abstained from oral hygiene, smoking, eating, or drinking (except water) within 1 hour of the visit. With the exception of Days 1, 8, and 15 of each study period, subjects used their assigned test materials unsupervised at home, once daily in the evening. On Days 1, 8, and 15 of each test period, subjects received an oral examination and supervised use of their assigned product. Fifteen minutes after supervised product use, subjects rated immediate relief of dry-mouth symptoms using an abbreviated PPQ represented by the first seven questions of the 16-question PPQ. At the Day 8 and 15 visits of each period and prior to the oral examination and supervised product use, subjects were asked to consider the oral care products used in the past week and to rate the product performance in managing their dry-mouth symptoms using the full 16-question PPQ.
Safety was assessed based on query of subjects for adverse events and clinical oral examination findings. Efficacy was evaluated based on subjective responses to: pretreatment PPQ (16 questions) administered at the screening visit and Days 8 and 15 of each study period; abbreviated PPQ (seven questions) administered on Days 1, 8, and 15 of each study period, within 15 minutes after regimen use.
Statistical Methods
An estimated sample size of 48 completed subjects per treatment group provided 80% power to detect between-treatment difference of 0.35 for mean PPQ scores, with a standard deviation of 0.6, using a two-sided test at the 0.05 significance level. (Note: In a clinical trial, a sufficient number of subjects should be enrolled to assure study completion with the sample size estimated to detect treatment difference.) With an estimated dropout rate of 10%, 52 qualified subjects were randomized in the expectation to complete with 48. Data for each efficacy endpoint were analyzed by an ANOVA model suitable for the analysis of data from a crossover study. Longitudinal (ie, within-treatment) comparisons were performed for each endpoint using a one-sample t-test on the changes (ie, delta values) between the scores at pairs of visits. All tests of hypotheses were two-sided and employed a level of significance of alpha = 0.05.
Results
Five adverse events were reported during the trial but none were considered to be treatment-related adverse events. All events were noted for the oral cavity. While using the EPTG product, two subjects experienced soft-tissue irritation in the anterior palatal region due to food burns. Two additional subjects presented with traumatic ulcers while using BOBG: a traumatic ulcer on the lower lip mucosa and an aphthous ulcer adjacent to the maxillary cuspid. A fifth subject presented with a broken tooth restoration while using the BOBG product. Both products were well-tolerated by the subjects.
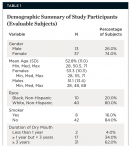
Fifty-three subjects were screened, and 52 were randomized. Of the 52 subjects randomized, 51 completed Period 1, and 50 completed Period 2. Data were available for the 50 evaluable subjects who completed all study periods. Two subjects withdrew due to schedule conflicts. Subjects’ ages ranged from 28 to 71 years with a mean age of 52.8; 74% of the subjects were female, and 80% were white, non-Hispanic. Most subjects were nonsmokers (84%), and 96% of subjects suffered from dry-mouth symptoms for at least 1 year (Table 1).
At the screening visit, subjects reported a variety of current remedies for managing dry-mouth symptoms that included sipping water/liquids, chewing gum, and sucking on candy/mints. Many of the subjects, 40.4%, sipped liquids only, while 25% sipped liquids and chewed gum; 19.2% sipped liquids, chewed gum, and sucked on candy/mints; 8% sipped liquids and sucked on candy/mints. The remaining subjects either chewed gum only or sucked on mints/candy only. None reported using any specialized dry-mouth products.
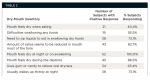
To qualify for entry into the study, subjects reported taking at least one OTC or prescription drug associated with causing salivary hypofunction or xerostomia and provided at least one positive response on the DMI. With the exception of one subject, all subjects reported experiencing three or more dry-mouth symptoms, and 82.7% of subjects complained that the amount of saliva seemed reduced most of the time (Table 2). As noted above, a majority of subjects (>70%) reported a positive response to five of the eight symptoms on the DMI and also reported having to take a medication known to present xerostomia as a side effect. Dry-mouth symptoms vary depending on the cause of dry mouth, and the authors are confident that subjects qualified for enrollment on the basis of the entry criteria.
The test products’ performances in relieving dry-mouth symptoms at Days 8 and 15 were evaluated using the 16-item PPQ. Compared to the subjects’ usual dry-mouth remedies, BOBG was effective in relieving 14 of the 16 dry-mouth symptoms at Day 8 and by Day 15 had relieved all of the 16 dry-mouth symptoms (Table 3 and Table 4). Following 7 and 14 days of use, EPTG was effective in relieving seven and eight of the dry-mouth symptoms, respectively, highlighted in Table 3 and Table 4, such as relieving discomfort of dry mouth, providing immediate relief, immediately moisturizing and lubricating, effectively lubricating, protecting from drying out, and providing long-lasting moisturization and long-lasting lubrication.
Following 7 and 14 days of use, the positive control (BOBG) was statistically significantly better than EPTG in relieving a majority of the principal dry-mouth symptoms (P < .05) (Table 3 and Table 4). By Day 8, the positive control was significantly better than EPTG for all 16 dry-mouth symptoms, which included key symptoms such as providing immediate relief, having an immediate moisturizing and lubricating effect, ease of use, comfort in mouth, soothing the mouth, allowing to speak without discomfort, effectively moistening and lubricating the mouth, having a pleasant taste, and refreshing the mouth (P < .05) (Table 3). Compared to EPTG at Day 15, subject ratings of dry-mouth symptoms were statistically significantly better for BOBG with respect to 12 of the 16 attributes (Table 4).
Table 5 provides a summary of percentage of subjects’ performance ratings in improving dry-mouth symptoms, compared to the ratings for their usual dry-mouth remedies. On the five-point rating scale of “poor” to “excellent,” 70% to 72% of subjects rated EPTG as “good” or “excellent” at relieving the discomfort of dry mouth and providing immediate relief, while 82% to 94% of subjects rated BOBG as “good” or “excellent” for the relief of the same symptoms. When asked about effectiveness at immediately moisturizing and lubricating, 68% to 70% of respondents rated EPTG as “good” or “excellent” compared to the ratings attributed to their usual remedies, while 86% to 94% of subjects rated BOBG as “good” or “excellent” for the same dry-mouth attributes at Days 8 and 15, respectively. Between 52% and 64% of subjects rated both products as “good” or “excellent” at providing “long-lasting moisturizing and lubricating effects” at Days 8 and 15.
Table 6 presents a summary of the responses to the immediate post-treatment abbreviated PPQ (seven questions) administered on Days 1, 8, and 15 of each study period. Subjects rated the effect of the products to provide immediate relief—within 15 minutes—of their dry-mouth symptoms. As indicated by the responses at Days 8 and 15, the mean ratings for immediate post-treatment were consistently higher for BOBG than EPTG.
Discussion
This study examined the subjective assessment (sensory performance) of once-daily use of Enamelon Preventive Treatment Gel and Biotene Oral Balance Gel in relieving the symptoms of self-reported dry mouth over 14 days of use. The study subjects were symptomatic for xerostomia and were not currently using specialized dry-mouth products or treatments. In this study, the authors evaluated once-daily use of EPTG, a 0.4% stannous fluoride gel, which is indicated for use once a day to help prevent tooth decay, in accordance with the US OTC Monograph for anticaries drug products.28 Additional study is warranted to determine if more frequent use of EPTG could provide further relief of dry-mouth symptoms. As expected, the positive control (BOBG), a class II medical device designed as an artificial saliva product, provided relief of dry-mouth symptoms consistent with published clinical data.12-14,22,23,29 Furthermore, subjects perceived that both EPTG and BOBG provided relief from dry-mouth symptoms, and the products were rated as “good” or “excellent” in relieving many of such symptoms. Throughout this study, once-daily use of BOBG was consistently superior to once-daily use of EPTG in relieving dry-mouth symptoms.
The effectiveness of the EPTG formulation to provide sensory relief of dry-mouth symptoms compared to subjects’ usual remedies may be attributed to the gel’s formulation, which includes Ultramulsion® technology (slowly dissolving muco-adhesive copolymers) and Gantrez® (a polyvinylmethyl ether/maleic acid copolymer). The copolymer ingredients are retained in the mouth to help coat the oral mucosa and may enhance substantivity, thus providing a long-lasting moisturizing and lubricating effect.
The lack of oral adverse events related to the test products might be explained by the absence of SLS in EPTG’s formulation; SLS is a known mucosal irritant that can increase the potential for soft-tissue discomfort. To help control for potential irritation and drying of oral mucosa that can be attributed to the known mucosal irritant, all products used during Periods 1 and 2, as well as during the washout period between Periods 1 and 2, did not contain SLS. The fact that subjects were not exposed to SLS may have contributed to the perceived relief of dry mouth with only once-daily use of EPTG. Further study is needed to determine the effectiveness of EPTG relative to a placebo or negative control product.
A comprehensive home-care regimen for dry-mouth sufferers should include products that will help to manage the risks of developing root and coronal caries, demineralization, dentin hypersensitivity, and gingivitis/gingival inflammation, which are conditions that are thought to be related to the loss of the protective properties of saliva.5,10 Most commercially available anticavity and antimicrobial dentifrices and mouthrinses contain SLS, or similar surfactant, and have not been reported to relieve dry-mouth symptoms. Use of SLS-containing products may prompt more frequent use of dry-mouth relief products such as BOBG, which may, in turn, increase the complexity of dry-mouth sufferers’ home care regimen, as well as cost.
As previously stated, patients with dry mouth often need protection/relief not only from dry-mouth discomfort but also caries, gingivitis, and dentin sensitivity. While BOBG clearly provides relief from dry-mouth discomfort, it is not indicated for managing the aforementioned oral complications associated with chronic dry mouth. Dry-mouth sufferers need to incorporate additional treatment products into their oral care regimen to address anticavity, antigingivitis, and antihypersensitivity benefits. There is no evidence in the literature suggesting that other OTC anticavity, antigingivitis, and sensitivity relief products can also help to relieve dry-mouth symptoms. The data from this study show that subjects perceive EPTG to be effective in relieving some dry-mouth symptoms. In addition, the documented clinical benefits of its active ingredient, 0.4% stannous fluoride, in reducing caries, preventing plaque and gingivitis, and relieving dentin hypersensitivity point to an important preventive benefit for dry-mouth suffers. EPTG with 0.4% stannous fluoride, delivering ACP, is indicated for providing protection against caries and gingivitis and for relieving dentin sensitivity.16,17,28 Furthermore, in this study EPTG provided sensory relief to many symptoms of dry mouth compared to the prestudy ratings of usual remedies for dry mouth.
Conclusions
This study showed that BOBG, the positive control, performed as expected for an approved medical device indicated for dry-mouth relief: it consistently provided reduction in dry-mouth symptoms compared to subjects’ usual remedies and was superior to EPTG, an OTC stannous fluoride preventive treatment gel product. Compared to subjects’ usual dry-mouth remedies, EPTG was beneficial in the management of many subject-perceived dry-mouth symptoms. EPTG soothes and moistens dry oral soft tissue, helps relieve the discomfort of dry mouth, has a long-lasting moisturizing and lubricating effect, and is non-irritating to dry-mouth tissues. While BOBG is effective in relieving dry-mouth symptoms, it is not indicated to help prevent/treat caries, gingivitis, or relieve dentin hypersensitivity. For dry-mouth sufferers, EPTG with 0.4% stannous fluoride and delivering ACP is indicated to help prevent/treat oral complications that can occur with chronic dry mouth, such as caries, gingivitis, and dentin hypersensitivity, and was also shown to help manage many of the sensory symptoms of dry mouth.
Disclosures
James C. DiMarino, DMD, MSEd, is an employee of Premier Dental Products Co. Sylvia L. Santos, RDH, MS, CCRP, is a consultant for Premier Dental Products Co.
About the Authors
Jeffery L. Milleman, DDS, MPA
Director
Clinical Operations
Salus Research Inc.
Fort Wayne, Indiana
Kimberly R. Milleman, RDH, BSEd, MS
Director
Compliance Specialist, and Examiner
Salus Research Inc.
Fort Wayne, Indiana
Sylvia L. Santos, RDH, MS, CCRP
President
SLS Clinical Research Consulting, LLC
Pine Brook, NJ
Howard M. Proskin, PhD
President
Howard M. Proskin & Associates
Rochester, New York
Kirstin K. Battershell, RN, MBA
Senior Clinical Coordinator
Salus Research Inc.
Fort Wayne, Indiana
James C. DiMarino, DMD, MSEd
Director of Clinical Affairs
Premier Dental Products Co.
Plymouth Meeting, Pennsylvania
References
1. Fox PC, Busch KA, Baum BJ. Subjective reports of xerostomia and objective measures of salivary gland performance. J Am Dent Assoc. 1987;115(4):581-584.
2. Cassolato SF, Turnbull RS. Xerostomia: clinical aspects and treatment. Gerodontology. 2003;20(2):64-77.
3. Gómez-Moreno G, Guardia J, Aguilar-Salvatierra A, et al. Effectiveness of malic acid 1% in patients with xerostomia induced by antihypertensive drugs. Med Oral Patol Oral Cir Bucal. 2013;18(1):e49-e55.
4. Villa A, Connell CL, Abati S. Diagnosis and management of xerostomia and hyposalivation. Ther Clin Risk Manag. 2014;11:45-51.
5. Plemons JM, Al-Hashimi I, Marek CL; American Dental Association Council on Scientific Affairs. Managing xerostomia and salivary gland hypofunction: executive summary of a report from the American Dental Association Council on Scientific Affairs. J Am Dent Assoc. 2014;145(8):867-873.
6. Thomson WM. Epidemiology of oral health conditions in older people. Gerodontology. 2014;31(suppl 1):9-16.
7. Thomson WM, Lawrence HP, Broadbent JM, Poulton R. The impact of xerostomia on oral-health-related quality of life among younger adults. Health Qual Life Outcomes. 2006;4:86.
8. Spielman AI. Interaction of saliva and taste. J Dent Res. 1990;69(3):838-843.
9. Atkinson JC, Wu AJ. Salivary gland dysfunction: causes, symptoms, treatment. J Am Dent Assoc. 1994;125(4):409-416.
10. Guggenheimer J, Moore PA. Xerostomia: etiology, recognition and treatment. J Am Dent Assoc. 2003;134(1):61-69.
11. Hopcraft MS, Tan C. Xerostomia: an update for clinicians. Aust Dent J. 2010;55(3):238-244.
12. Jose A, Siddiqi M, Cronin M, et al. A randomized clinical trial in subjects with dry mouth evaluating subjective perceptions of an experimental oral gel, oral rinse and a mouth spray compared to water. Am J Dent. 2016;29(1):58-64.
13. Shahdad SA, Taylor C, Barclay SC, et al. A double-blind, crossover study of Biotene Oralbalance and BioXtra systems as salivary substitutes in patients with post-radiotherapy xerostomia. Eur J Cancer Care (Engl). 2005;14(4):319-326.
14. Epstein JB, Emerton S, Le ND, Stevenson-Moore P. A double-blind crossover trial of Oral Balance gel and Biotene toothpaste versus placebo in patients with xerostomia following radiation therapy. Oral Oncol. 1999;35(2):132-137.
15. Oong EM, Griffin SO, Presson S, et al. Estimates of dry mouth in adult U.S. population, NHANES 1999-2002 [abstract]. J Dent Res. 2007;86(spec iss A). Abstract 961.
16. US Food and Drug Administration. Oral health care drug products for over-the-counter human use; antigingivitis/antiplaque drug products; establishment of a monograph; proposed rules. Dept of Health and Human Services; 2003; Fed Reg 68:32232-32287.
17. US Food and Drug Administration. Oral health care drug products for over-the-counter human use; amendment to tentative final monograph to include OTC relief of oral discomfort drug products; Notice of proposed rulemaking. Dept of Health and Human Services; 1991; Fed Reg 56:48302.
18. Schemehorn BR, DiMarino JC, Movahed N. Comparison of the enamel solubility reduction from various prescription and OTC fluoride toothpastes and gels. J Clin Dent. 2014;25(3):61-64.
19. Schemehorn BR, DiMarino JC, Movahed N. Comparison of the incipient lesion enamel fluoride uptake from various prescription and OTC fluoride toothpastes and gels. J Clin Dent. 2014;25(3):57-60.
20. Rantanen I, Tenovuo J, Pienihäkkinen K, Söderling E. Effects of a betaine-containing toothpaste on subjective symptoms of dry mouth: a randomized clinical trial. J Contemp Dent Pract. 2003;4(2):11-23.
21. Torres SR, Peixoto CB, Caldas DM, et al. Relationship between salivary flow rates and Candida counts in subjects with xerostomia. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2002;93:149-154.
22. Jose A, Targett D, Ta J, et al. Evaluation of the oral tolerance of an experimental oral gel [abstract]. J Dent Res. 2014;93(spec iss A). Abstract 600.
23. Lomax A, Jose A, Akwagyiram I, et al. Evaluation of the oral tolerance of two experimental oral rinses [abstract]. J Dent Res. 2014;93(spec iss A). Abstract 599.
24. Thomson WM, van der Putten GJ, de Baat C, et al. Shortening the xerostomia Inventory. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;112(3):322-327.
25. Shirodaria S, Kilbourn T, Butler A, Middleton S. Subjective assessment of a new moisturizing mouthwash for the symptomatic management of dry mouth. J Clin Dent. 2006;17(2):39-44.
26. Shirodaria S, Kilbourn T, Richardson M. Subjective assessment of a new moisturizing mouth spray for the symptomatic relief of dry mouth. J Clin Dent. 2006;17(2):45-51.
27. Tappuni AR, Moutasim KA, Shirodaria S, Challacombe SJ. Dry mouth relief: evaluation of two preparations in Sjogren’s syndrome [abstract]. J Dent Res. 2008;87(spec iss B). Abstract 1568.
28. US Food and Drug Administration. Anti-caries drug products for over-the-counter human use. Dept of Health and Human Services; 1995; 21 CFR 355.
29. Matear DW, Barbaro J. Effectiveness of saliva substitute products in the treatment of dry mouth in the elderly: a pilot study. J R Soc Promot Health. 2005;125(1):35-41.