A Report of Three Cases from an Ongoing Prospective Clinical Study on a Novel Pink Biomimetic Implant System
Kevin G. Murphy, DDS, MS | Mariano A. Polack, DDS, MS | Joseph M. Arzadon, MD, DDS | Ronald D. Hickerson, DDS | E. Todd Scheyer, DDS, MS
Abstract: While, overall, dental implants are highly efficacious, consistently achieving predictable esthetic results can pose color-driven challenges at the implant/soft-tissue interface, particularly in the esthetic zone. An ongoing, 5-year, multicenter (eight sites) prospective study is following a total of 168 implants placed in 120 patients using a novel biomimetic (osteoconductive) implant system with pink collars and abutments that afford better gingival color matching, enhanced design variability, and streamlined fabrication and customization of the abutment/implant-tissue interface. The full results of this study will be published at the 3-year and 5-year timepoints; however, clinical analyses of the 18-month interim survival rates, marginal bone and soft-tissue level changes, and esthetics have been completed, showing an overall success rate among all of the implanted sites of 95.8%. For the three representative cases described in this article (two anterior, one posterior), all of the implant site probing depths were ≤3 mm at the final crown placement as well as at 6, 12, and 18 months after implant placement. Facial soft-tissue heights were stable or increased by ≥0.86 mm; lingual soft-tissue heights were reduced by ≤1.11 mm. Facial and lingual attached-gingiva widths were reduced by ≤1.20 mm and ≤0.63 mm, respectively. Interproximal marginal alveolar bone levels were stable (gain/loss range: +0.40 to -1.1 mm) from the start of the study through 18 months. Gingival inflammation, bleeding on probing, and plaque were infrequently observed, and esthetic results were uniformly excellent at the 18-month follow-up visit.
BACKGROUND
Predictable treatment outcomes, especially in dental implant esthetics, can be achieved when bone and soft-tissue volume at the implant site and the adjacent teeth are not compromised. Soft- or hard-tissue deficiencies of the edentulous space are the most common obstacles to achieving gingival esthetics around the final restoration. Previously published work on thin peri-implant mucosa around dental implants has demonstrated a distinguishable gray show-through effect of the underlying titanium implant and abutment.
Attempts have been made by various manufacturers to compensate for this effect using white ceramic abutments (zirconia) or pink1 or yellow2 coloring of the neck/abutment through anodizing processes.3,4 The use of dental implants in the treatment of totally or partially edentulous patients has demonstrated a high degree of success, with documented survival rates of 90% to 97%.5-9
For the past three decades, dental implants have been a routine and clinically predictable treatment method for the functional replacement of missing teeth. Esthetic challenges frequently arise for the prosthodontist, restorative dentist, and periodontist regarding the visible (or, ideally, indistinguishable) facial implant/soft-tissue interface, of particular concern in the esthetic zone.
The implant system used in the following three cases (Genesis®, Keystone Dental, Inc, www.keystonedental.com) has been in global use since November 2010. In addition to its osteoconductive implant–bone interface, produced by application of anodic spark deposition or discharge (ASD) to the titanium–implant surface (BioSpark™, Keystone Dental),10-13 the main distinguishing feature of this implant system is the pink external coloring of its restorative components, including the implant collar and matching prefabricated customizable titanium abutments. Since the initiation of this study, additional custom abutments with a pink subgingival portion and a white zirconia core have become available for this system.
Published studies evaluating the chemistry and roughness of this implant system’s surface include three animal studies on bone-to-implant contact,10,14,15 two in-vitro studies on cell behavior,11,12 and three studies on the effects of pink on the gingival esthetics.1,2,16
An additional treatment to the threaded portion of the implant surface involves ASD,17 also known as microarc oxidation or glow discharge deposition. This process produces an osteoconductive implant surface rich in calcium and phosphorus ions as a bone interface.11,13
The pink color of this implant system is produced on the implant surface by a proprietary electrochemical anodization process (AnaTite™, Keystone Dental) that produces a layer of titanium oxide on the implant surface. The resulting pink coloration reduces the potential of gray soft tissue, thus offering the clinician an alternative to zirconia for creating, enhancing, and refining gingival esthetics.
Ishikawa et al18,19 quantitatively characterized color variabilities in healthy and inflamed marginal gingiva; these analyses helped establish a basis for critical assessment of the colorimetric influence of various implant, abutment, and restoration materials relative to such a soft-tissue color parameter range.
Spectrophotometric analyses published by Park et al confirmed that there is a measurable difference between the colors of natural maxillary labial gingiva and the surfaces of conventional titanium implants.16 More specifically, colorimetric data from Ishikawa-Nagai et al suggest that, in comparison to other colors, light pink coloration of the implant neck produces an optimal color that is clinically indistinguishable from that of natural-tooth gingiva.1 Patient-specific shading of the implant collar using a similar approach has also been described in a three-case series published by Sumi et al, who reported such specificity to provide stable gingival esthetics at a 1.5-year follow-up, especially in patients with a thin gingival biotype.4
Yellow/gold and pink can both produce harmonious esthetics when juxtaposed to natural gingiva. However, pink usually offers more of an advantage, ie, the closer natural shades are mimicked, the better the esthetic result.
Another case report by Polack20 specifically evaluated the pink nano-rough implant system presented in the current case series (Genesis). An excellent result was achieved in an esthetically demanding case requiring multiple extractions and site development for the replacement of two maxillary lateral incisors (using narrow-diameter, 3.8-mm x 13-mm fixtures) in a severely resorbed ridge.20
Similarly to other implant systems from a functional standpoint, the aggressive thread pitch of this implant fixture, in these authors’ experience, also facilitates its use in immediate placement and loading scenarios. Of note, a substantial proportion of implants in this study population have reached or surpassed 3 years of survival and function.
Platform Switching
Platform switching (PS) has become a standard feature in the design of conventional implant systems, and has expanded the clinician’s control over crestal bone preservation. Numerous studies21-26 and systematic reviews27-29 have reported reduced bone resorption for platform-switched implants compared with platform-matching implants. The implant system used in this study incorporates a platform-switch ranging between 0.50 mm to 1.38 mm, depending on implant fixture diameter (IFD):
• IFD = Ø3.8 mm: 0.50 mm PS
• IFD = Ø4.5 mm: 0.57 mm PS
• IFD = Ø5.5 mm: 0.70 mm PS
• IFD = Ø6.5 mm: 1.38 mm PS
Cappiello et al reported a significant bone-protective effect of platform-switching equal to 0.72 mm in a controlled clinical trial of 131 implants (all placed at the crest) in 45 patients.24 Clinical studies by Prosper et al30 and Canullo et al25 have also demonstrated advantages of platform-switched implants over regular implants with respect to crestal bone stability, with a minimum of 24 months’ follow-up. Recent systematic reviews consistently confirm that implants with platform-switched abutments are associated with better crestal bone preservation than implants with platform-matched abutments.28,29,31
Five-Year Prospective Clinical Study
An ongoing 5-year study continues to evaluate the use of this implant system (168 implants placed in 120 partially edentulous patients). Its objectives include assessment of the 5-year survival rate of this implant system, implant success, incidence of excessive bone loss, peri-implant infection and other complications, incidence of adverse device effects, change in marginal bone level, soft-tissue esthetic outcomes, and the number and nature of prosthetic revisions.
Alignment, orientation, and magnification of the periapical radiographic images of all subjects’ implants and alveolar bone levels were standardized by rotating and translating each image such that all were uniformly aligned, oriented, and scaled, using a semi-automated program (MATLAB®, MathWorks, Inc; https://www.mathworks.com/products/matlab/).
For angles, imaging differences in both elevation (above or below correct plane) and azimuth (mesial–distal) between images in the same series were computed. All of the images in this data set have a percentage error of less than 3.5%.
Consistent with other implant designs, most osseointegration failures occurred during the healing period following placement or shortly after prosthetic loading. However, unlike other designs, the location (mandible versus maxilla) and length of the implant had no effect on the survival rate. After loading, this implant system has a survival rate of over 99%, based on the 18-month interim data collected from all subjects in the multicenter study.
This article presents the 18-month interim results of three representative cases from that study, descriptively evaluating esthetic and hard- and soft-tissue study endpoints in two esthetic-zone and one posterior application of this pink implant system.
CASE REPORTS
Demographics, medical histories, and implant indications for these three patients are presented in Table 1. All of the subjects signed an informed consent document prior to enrollment in the study; an institutional review board at each study center approved the informed consent document and study protocol. The study is being conducted in accordance with the United States 21 CFR Parts 11, 50, 56; the Health Insurance Portability & Accountability Act of 1996 (HIPAA); and the Declaration of Helsinki and its amendments per the most recent assembly of the World Medical Association.
Case 1
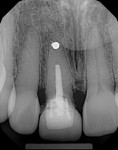
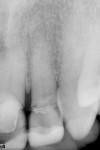
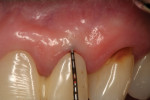
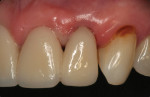
In November 2011 a healthy nonsmoking 52-year-old perimenopausal African-American woman presented to Dr. Scheyer’s periodontal practice on referral from her restorative dentist with a symptomatic endodontically treated (with history of amalgam retrofill) maxillary right central incisor (tooth No. 8); in addition to endodontic pathology, this tooth had a failing porcelain jacket crown (recurrent caries) over a pin-retained resin buildup (Figure 1). Significant probing depth on the mid-facial and bleeding on probing (BOP) were observed.
She was taking enalapril for hypertension and progesterone USP for perimenopausal symptoms. She had no known drug allergies, and an otherwise unremarkable medical history other than routine surgeries (Table 1). Azithromycin was administered beginning the day before surgery and continued for 5 days postoperatively.
With the patient under conscious sedation, a surgical extraction of tooth No. 8 was performed without complication. Bleeding on probing and purulence were noted at the implant placement visit and the site was aggressively debrided.
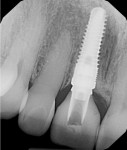
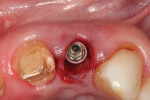
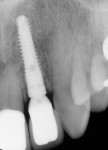
There was a narrow bony defect in the facial cortical plate measuring approximately 8 mm from the interproximal crestal bone heights. The implant osteotomy was prepared along the lingual cortical plate using a surgical guide for the placement of a 4.5-mm x 16-mm Genesis implant (Figure 2). The implant was placed with favorable stability (above 45 Ncm of insertion torque). A temporary cylinder abutment was used to fabricate a screw-retained provisional restoration (Figure 3). The retention screw for the provisional was torqued to 20 Ncm.
The facial ridge deficiency was grafted with mineralized freeze-dried bone allograft and covered with a resorbable collagen membrane using minimally invasive facial tunneling to allow for placement of the graft and membrane. The occlusion was adjusted, and there were no centric or excursive contacts. An Essix retainer was fabricated to prevent any parafunctional stresses on the implant-supported provisional restoration. A postoperative periapical radiograph confirmed full seating of the provisional abutment. The patient responded well to therapy and showed normal healing at the 10-day follow-up visit.
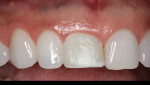
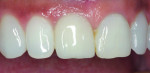
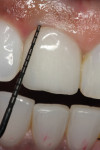
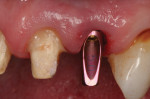
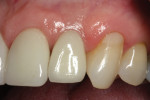
Restorative treatment began in February 2012. The implant-supported provisional was removed and the open-tray technique was used to fabricate a customized coping for the final impression, to maintain gingival contours that were sculpted with the provisional (Figure 4). The objective was to mimic this emergence profile in the final crown. The patient visited the laboratory to obtain a custom shade match. A custom-prepared UCLA abutment was fabricated and torqued to 30 Ncm, and a custom-layered all-ceramic crown (IPS e.max® Ceram, Ivoclar Vivadent, www.ivoclarvivadent.us/) was cemented with RelyX™ Ultimate adhesive cement (3M www.3m.com/3M/en_US/Dental/) in July 2012. The patient was very pleased with the restorative outcome, and all gingival tissues were healthy with no probing depth ≥3 mm at the 18-month follow-up visit (Figure 5 through Figure 7).
Table 2 through Table 4 show the soft-tissue height, attached-gingiva width, and marginal-bone change measurements at final crown placement, as well as 6, 12, and 18 months after the start of study treatment (see subject LDJ-01). BOP was noted at the 18-month follow-up visit.
Case 2
In December 2011, a healthy 54-year-old Caucasian woman who is a heavy smoker presented to Dr. Murphy’s periodontal-prosthodontics practice. She was taking over-the-counter omeprazole for gastroesophageal reflux disease with an otherwise unremarkable medical history, and had no known drug allergies. Gingivitis and BOP were observed (probably smoking-related), with no probing depth ≥3 mm.
The maxillary left lateral incisor (tooth No. 10) was partially supporting a 4-unit provisional splint spanning teeth Nos. 7 through 10, and had fractured at its gingival margin. Moderate gingival inflammation was present with a midfacial probing depth of approximately 2 mm at Visit 1 (Figure 8 and Figure 9). Because of an envisioned difficult endodontic treatment course/prognosis, the patient opted for extraction and implant placement.
A CAD/CAM-created surgical guide was fabricated using 3D diagnostic software (Sirona SICAT, https://www.sicat.com/) and used to manage the osteotomy preparation of the socket, followed by immediate placement of a 3.8-mm × 16-mm Genesis implant (Figure 10) at an insertion torque of 45 Ncm.
The “one abutment-one time” technique (as described by Mandelaris and Vlk32) was used to eliminate the traditional final-impression phase (and, importantly, the need to pack retraction cord in an inflamed area), and complete the first phase of fabrication of the definitive abutment, thus saving chair and laboratory time as well as expense. Compared with standard impression techniques, this approach proved to be a faster, easier, and more effective method of choosing and customizing the fit of the final pink abutment for this case.
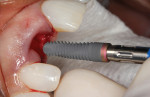
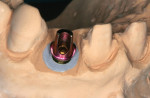
A stock Esthetic-contour Ti abutment (Genesis) was tried in at the time of implant placement (Figure 11), and a pickup transfer impression was used to create a laboratory model by attaching the implant analog to the abutment, seating it, and pouring stone around the transfer (Figure 12).
After adjusting the abutment for appropriate marginal and interproximal contouring and confirming a good fit, the abutment was repositioned on the laboratory model and scanned using an extraoral scanner to provide the laboratory with a working digital file. The model was then used to fabricate the 4-unit provisional splint (Figure 13).
This same abutment was subsequently torqued into the implant at 35 Ncm on the day of surgery. The immediate provisional restoration was inserted and cemented with temporary cement.
During the integration period, a NobelProcera zirconia coping (Nobel Biocare, www.nobelbiocare.com) was fabricated. Three months after implant placement, the coping was tried in to verify the fit and picked up in an impression using a polyether impression material. From this impression, the final working model was fabricated.
The final all-ceramic restoration (IPS e.max Ceram) was layered onto a NobelProcera zirconia coping in May 2012 and cemented with RelyX Unicem Self-Adhesive Universal Resin Cement (3M). The patient was very pleased with the restorative outcome, and all gingival tissues were healthy with no probing depth ≥3 mm at the 18-month follow-up visit (see subject SJH-02, Table 2 through Table 4; Figure 14 and Figure 15).
Case 3
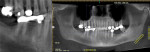
In November 2011, a healthy, nonsmoking 59-year-old Hispanic man (who has been a patient of record in Dr. Polack’s prosthodontic practice since 2007) presented with a partially edentulous mandibular right quadrant (Figure 16) after the recent extraction (in September 2011) of non-restorable teeth Nos. 28 and 30. His medical history was unremarkable except for a detached retina in 2005; he was taking no medications and had no known drug allergies. These two teeth had supported a bridge (pontic was position No. 29) that had been recemented twice as of June 2011, after which time he decided to have implants placed. Figure 17 shows a pre-extraction periapical view. Tooth No. 31 had been previously deemed nonrestorable, and was extracted by the consulting oral surgeon Dr. Arzadon, who also performed the surgical implant phase.
In January 2011, alveolar regenerative therapy was performed for preservation of the recent extraction sockets because of the extensive loss of buccal alveolar bone. An organic bovine bone matrix was placed to augment these buccal defects (Class B bone quantity in position Nos. 29 and 30) during osteotomy (type-1 bone density throughout the sites) and placement of three pink Genesis implants (4.5 mm × 11.5 mm, 4.5 × 11.5 mm, and 5.5 × 11.5 mm in position Nos. 28, 29, and 30, respectively) in November 2011.
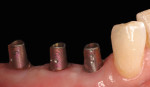
Prefabricated pink abutments were customized by preparing them just below the gingival margin and torqued to 30 Ncm (two 4.5-mm and one 5.5-mm prefabricated Genesis Ti Temporary Abutments, Figure 18). No provisionalization was used in this case. A final impression was obtained using the closed-tray technique.
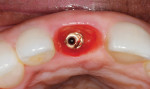
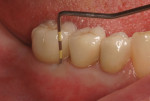
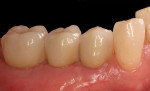
After placement of Teflon tape in the access holes, the final porcelain-fused-to-metal restorations were cemented in February 2012 with RelyX™ Unicem. The patient was very pleased with the restorative outcome, and all gingival tissues were healthy with no probing depth ≥3 mm at the 18-month follow-up visit. The final restorations, soft-tissue esthetics, probing measurements, and periapical radiograph are shown in Figure 19 through Figure 21; see Table 2 through Table 4 for soft-tissue and marginal-bone levels (subject AT-05).
Conclusion
This article presents three cases offering the first published data from this 5-year prospective study of a novel esthetic implant system. A representative case series such as this provides early and valuable exposure for prospective multicenter studies evaluating dental implant designs. Such previewing affords practicing clinicians access to data from new functional and esthetic implant technologies in advance of the publication of the final data from longer-term prospective studies. This can benefit patients earlier. McGuire et al recently published such a series presenting 12-month data from a comparative study of three different implant designs that is currently in its fifth year of follow-up.33
Overall, the three cases presented here (all of which are currently considerably beyond 18 months post-loading) provide further clinical evidence of favorable esthetic results, maintenance of marginal bone levels, and soft-tissue dimensions, and are in addition to those already observed in the first clinical study of this biomimetic pink-colored implant system.
All three cases have been followed well into 2015; all continue to maintain hard- and soft-tissue stability according to proscribed study endpoints, as well as good gingival health and excellent esthetics.
Importantly, our study’s endpoints do not include superiority or comparisons to any other implant system; these cases are a snapshot of esthetics and marginal bone stability after 18 months of follow-up in cases that did not pose major esthetic challenges. Future studies should address potential advantages of this collar type in esthetically and/or periodontally compromised cases.
Acknowledgments
The authors thank the patients who participated in the study and allowed their cases to be presented here; Keystone Dental, Inc. for conducting and supporting this clinical study; Biomedical Modeling Inc, Boston, Massachusetts, for standardization and measurement of radiographic bone levels; and Scott A. Saunders, DDS, ELS, CMPP, at Dental and Medical Writing and Editing, LLC, Lancaster, Pennsylvania, for professional dental and medical writing and editing services in preparation of the manuscript.
Disclosure
Drs. Murphy, Polack, and Scheyer, the principal investigators, received an educational grant from Keystone Dental, Inc. for this multicenter study. Drs. Arzadon and Hickerson have no financial relationships to disclose relative to this article.
References
1. Ishikawa-Nagai S, Da Silva JD, Weber HP, Park SE. Optical phenomenon of peri-implant soft tissue. Part II. Preferred implant neck color to improve soft tissue esthetics. Clin Oral Implants Res. 2007;18(5):575-580.
2. Pecnik CM, Roos M, Muff D, et al. In vitro color evaluation of esthetic coatings for metallic dental implants and implant prosthetic appliances. Clin Oral Implants Res. 2015;26(5):563-571.
3. Kim YS, Ko Y, Kye SB, Yang SM. Human gingival fibroblast (HGF-1) attachment and proliferation on several abutment materials with various colors. Int J Oral Maxillofac Implants. 2014;29(4):969-975.
4. Sumi T, Takeshita K, Takeichi T, et al. Patient-specific gingiva-colored abutments: a case series. Int J Periodontics Restorative Dent. 2014;34(4):469-475.
5. Schwartz-Arad D, Laviv A, Levin L. Failure causes, timing, and cluster behavior: an 8-year study of dental implants. Implant Dent. 2008;17(2):200-207.
6. Paquette DW, Brodala N, Williams RC. Risk factors for endosseous dental implant failure. Dent Clin North Am. 2006;50(3):361-374, vi.
7. Rosenberg ES, Cho SC, Elian N, et al. A comparison of characteristics of implant failure and survival in periodontally compromised and periodontally healthy patients: a clinical report. Int J Oral Maxillofac Implants. 2004;19(6):873-879.
8. Perry J, Lenchewski E. Clinical performance and 5-year retrospective evaluation of Frialit-2 implants. Int J Oral Maxillofac Implants. 2004;19(6):887-891.
9. Weng D, Jacobson Z, Tarnow D, et al. A prospective multicenter clinical trial of 3i machined-surface implants: results after 6 years of follow-up. Int J Oral Maxillofac Implants. 2003;18(3):417-423.
10. Bertollo N, Sandrini E, Dalla Pria P, Walsh WR. Osseointegration of multiphase anodic spark deposition treated porous titanium implants in an ovine model. J Arthroplasty. 2015;30(3):484-488.
11. Sandrini E, Giordano C, Busini V, et al. Apatite formation and cellular response of a novel bioactive titanium. J Mater Sci Mater Med. 2007;18(6):1225-1237.
12. Giordano C, Chiesa R, Sandrini E, et al. Physical and biological characterizations of a novel multiphase anodic spark deposition coating to enhance implant osseointegration. J Mater Sci Mater Med. 2005;16(12):1221-1229.
13. Giordano C, Sandrini E, Del Curto B, et al. Titanium for osteointegration: Comparison between a novel biomimetic treatment and commercially exploited surfaces. J Appl Biomater Biomech. 2004;2(1):35-44.
14. Giavaresi G, Fini M, Chiesa R, et al. A novel multiphase anodic spark deposition coating for the improvement of orthopedic implant osseointegration: an experimental study in cortical bone of sheep. J Biomed Mater Res A. 2008;85(4):1022-1031.
15. Giavaresi G, Chiesa R, Fini M, Sandrini E. Effect of a multiphasic anodic spark deposition coating on the improvement of implant osseointegration in the osteopenic trabecular bone of sheep. Int J Oral Maxillofac Implants. 2008;23(4):659-668.
16. Park SE, Da Silva JD, Weber HP, Ishikawa-Nagai S. Optical phenomenon of peri-implant soft tissue. Part I. Spectrophotometric assessment of natural tooth gingiva and peri-implant mucosa. Clin Oral Implants Res. 2007;18(5):569-574.
17. Chiesa R, Sandrini E, Santin M, et al. Osteointegration of titanium and its alloys by anodic spark deposition and other electrochemical techniques: a review. J Appl Biomater Biomech. 2003;1(2):91-107.
18. Ishikawa S, Furukawa K, Ishibashi K. Colorimetric studies of the gingiva: color variation of the gingiva in the upper anterior region. The Journal of the Japan Prosthodontic Society. 1988;32(4):821-828.
19. Ishikawa S. Colorimetric study of marginal gingiva: inflammatory effects on color difference analyses from the standpoint of gingival transmission. The Journal of the Japan Prosthodontic Society. 1988;32(4):829-38.
20. Polack MA. Restoration of maxillary incisors with an innovative biomimetic implant system: a case report. J Implant Adv Clin Dent. 2012;4(5):39-49.
21. Sahabi M, Adibrad M, Mirhashemi FS, Habibzadeh S. Biomechanical effects of platform switching in two different implant systems: a three-dimensional finite element analysis. J Dent (Tehran). 2013;10(4):338-350.
22. Gultekin BA, Gultekin P, Leblebicioglu B, et al. Clinical evaluation of marginal bone loss and stability in two types of submerged dental implants. Int J Oral Maxillofac Implants. 2013;28(3):815-823.
23. Trammell K, Geurs NC, O’Neal SJ, et al. A prospective, randomized, controlled comparison of platform-switched and matched-abutment implants in short-span partial denture situations. Int J Periodontics Restorative Dent. 2009;29(6):599-605.
24. Cappiello M, Luongo R, Di Iorio D, et al. Evaluation of peri-implant bone loss around platform-switched implants. Int J Periodontics Restorative Dent. 2008;28(4):347-355.
25. Canullo L, Rasperini G. Preservation of peri-implant soft and hard tissues using platform switching of implants placed in immediate extraction sockets: a proof-of-concept study with 12- to 36-month follow-up. Int J Oral Maxillofac Implants. 2007;22(6):995-1000.
26. Lazzara RJ, Porter SS. Platform switching: a new concept in implant dentistry for controlling postrestorative crestal bone levels. Int J Periodontics Restorative Dent. 2006;26(1):9-17.
27. Kinaia BM, Shah M, Neely AL, Goodis HE. Crestal bone level changes around immediately placed implants: a systematic review and meta-analyses with at least 12 months’ follow-up after functional loading. J Periodontol. 2014;85(11):1537-1548.
28. Al-Nsour MM, Chan HL, Wang HL. Effect of the platform-switching technique on preservation of peri-implant marginal bone: a systematic review. Int J Oral Maxillofac Implants. 2012;27(1):138-145.
29. Atieh MA, Ibrahim HM, Atieh AH. Platform switching for marginal bone preservation around dental implants: a systematic review and meta-analysis. J Periodontol. 2010;81(10):1350-1366.
30. Prosper L, Redaelli S, Pasi M, et al. A randomized prospective multicenter trial evaluating the platform-switching technique for the prevention of postrestorative crestal bone loss. Int J Oral Maxillofac Implants. 2009;24(2):299-308.
31. Annibali S, Bignozzi I, Cristalli MP, et al. Peri-implant marginal bone level: a systematic review and meta-analysis of studies comparing platform switching versus conventionally restored implants. J Clin Periodontol. 2012;39(11):1097-1113.
32. Mandelaris GA, Vlk SD. Guided implant surgery with placement of a presurgical CAD/CAM patient-specific abutment and provisional in the esthetic zone. Compend Contin Educ Dent. 2014;35(7):494-504.
33. McGuire MK, Scheyer T, Ho DK, et al. Esthetic outcomes in relation to implant-abutment interface design following a standardized treatment protocol in a multicenter randomized controlled trial—a cohort of 12 cases at 1-year follow-up. Int J Periodontics Restorative Dent. 2015;35(2):149-159.
About the Authors
Kevin G. Murphy, DDS, MS
Department of Periodontics
University of Maryland
Baltimore College of Dental Surgery
Baltimore, Maryland
Kevin G. Murphy & Associates, P.A.
Baltimore, Maryland
Mariano A. Polack, DDS, MS
Dental Design Drs. Polack and Olano
Gainesville, Virginia
Joseph M. Arzadon, MD, DDS
Northern Virginia Surgical Arts
Arlington, Virginia
Ronald D. Hickerson, DDS
Hickerson & Associates DDS
Houston, Texas
E. Todd Scheyer, DDS, MS
University of Texas Dental Branch
Houston, Texas
University of Texas
Health Science Center at San Antonio
San Antonio, Texas