Dental Implant Uncovering Techniques with Emphasis on Increasing Keratinized Mucosa
Abstract:
The focus in implantology today is on the need to achieve results that are not only functional but also long-lasting and highly esthetic. Increased width and thickness of keratinized mucosa (KM) around dental implants, which is now widely recognized to be associated with better long-term success than implants without KM, addresses these goals. This is especially important in the esthetic zone, where marginal recession can be of concern. The author describes the following methods of implant uncovering that emphasize enhancement of the keratinized mucosa: the punch technique; buccally advanced flap; roll flap; tissue preservation technique; free connective tissue graft; and pedicle flap.
Over the past two decades, dental implant therapy has advanced dramatically. The need to achieve not only functional results but also long-lasting, highly esthetic outcomes has been elevated to the forefront. The criteria for success have shifted from a focus on implant “survival” to those that signal implant “success.”1
In 1989, Smith and Zarb published an updated set of criteria for success that included an “esthetic” category, writing, “the implant design does not preclude placement of a crown or prosthesis with an appearance that is satisfactory to the patient and dentist.”2 Over the next decade and a half, much attention was given to quantifying what constituted a “satisfactory appearance.” In 2005, Furhauser et al3 published a succinct yet thorough set of criteria to objectively evaluate the appearance of an implant-supported restoration in the esthetic zone. Their “pink esthetic score” serves not only as a means of evaluating outcomes, but of predicting in the planning stages how successful an esthetically sensitive treatment may turn out to be and the modalities that may be employed to enhance that treatment. If an implant-supported restoration is to be “in symmetry with the reference teeth” as Furhauser et al suggest, attention must be given to pre-surgical levels of the hard and soft tissue that will support and surround the implant restoration, with an eye towards executing any augmentations or other strategies—eg, using pink restorative material—that may enhance the outcome.
As the term “pink esthetics” implies, the soft tissue surrounding an implant-supported restoration plays a critical role in achieving an esthetic result. While the need for keratinized mucosa (KM) around a “functional” implant is still a contentious subject, it has been shown that the development of KM around an implant/restorative interface will enhance the long-term health of the bone and soft tissue surrounding an implant.4-7 Yeung5 wrote: “…for many clinicians there is a case for routinely providing a keratinized gingival band around implant restorations to facilitate plaque control and hence reduce the incidence of plaque-related peri-implant disease. In a recent publication in the International Journal of Oral and Maxillofacial Implants, in a Current Issues Forum, the editor posted the question, ‘To minimize complications, is it essential that implant abutments be surrounded by keratinized tissue?’ The consensus of opinions expressed by the authors participating in this debate was, in general, supportive of the clinical practice of providing a keratinized gingival band around implant restorations where possible and practical.” In a comparative study on the significance of KM around dental implants, Boynueğri et al6 wrote that the results of their study “showed that an adequate band of KM was related with less plaque accumulation and mucosal inflammation as well as pro-inflammatuar mediators, suggesting that it may be critical, especially for plaque control and plaque-associated mucosal lesions around dental implants.”
Zigdon and Machtei8 have demonstrated a significant difference in both clinical and immunological parameters around implants that have thick versus thin KM. They found that the average gingival thickness was 1.1 mm and that KM thicker than 1 mm resisted recession twice as much as KM thinner than 1 mm. They also found that the width of the KM affected marginal recession. A thick band of KM, greater than or equal to 1 mm, had three times less recession than bands less than 1 mm. In a study to evaluate gingival biotype assessment, Kan et al9 performed direct measurement of gingival thickness immediately after atraumatic extraction. They found that “mean gingival thickness obtained from direct measurements was 1.06 mm ± 0.27 mm, with an equal distribution (50%) of sites with gingival thicknesses of ≤ 1 mm and > 1 mm.”
In a study to evaluate the effectiveness of connective tissue grafting on facial tissue stability, Kan et al10 demonstrated that thick KM, which they quantified as 1 mm or greater, is significantly more resistant to recession.
Where there is inadequate KM around an implant—ie, less than 1 mm thick and 1 mm wide—enhancement should be considered. Soft-tissue enhancement may be performed at various stages of implant therapy, prior to, during, or after the implant has been placed. While tissue augmentation may be performed at these various stages, the most advantageous time to consider soft-tissue enhancement is at implant uncovering in a two-stage, submerged approach. It is worthwhile to note that where a single-stage approach may be feasible—ie, able to achieve adequate primary stability—the need for soft-tissue enhancement may, in some cases, make treatment better suited to a two-stage approach.
When uncovering an implant at second-stage surgery, several surgical approaches are available. Determining the most appropriate one depends on the width and thickness of KM available. The preservation of any existing keratinized gingiva demands careful surgical manipulation. Although a thickness and width of 1 mm seems to be adequate, to be practical, surgical manipulation and post-surgical soft-tissue contraction warrant developing a 2-mm to 3-mm width and a 2-mm thickness of KM.
The discussion below presents several techniques for implant uncovering, with a special focus on increasing the KM surrounding the implant.
Punch Technique
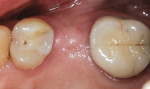
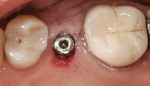
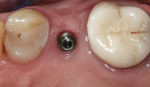
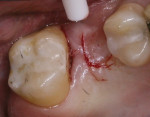
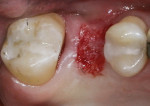
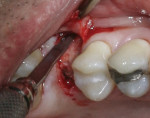
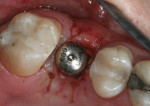
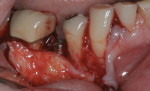
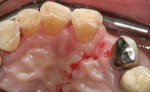
When an adequate band—ie, at least 1 mm—of KM surrounds the implant (Figure 1) and there is no need to build up the buccal root profile, a circular incision of the same diameter as the implant may be made to remove the tissue covering the implant (Figure 2 and Figure 3). This can be performed with a scalpel, rotary tissue punch, or a single-use dermatological punch.
Buccally Advanced Flap
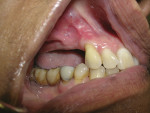
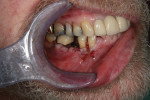
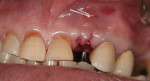
Where there is a need to increase the width of KM on the buccal of a maxillary implant (Figure 4), a buccally advanced flap can be used.11 Starting in the buccal mucosa, two parallel vertical incisions are carried out palatally 3 mm into the KM. A horizontal incision is then made on the palate to connect both vertical incisions (Figure 5). The flap is reflected and moved to the buccal and secured with single interrupted sutures and sling sutures around the healing abutment (Figure 6). The tissue should be allowed to mature for 12 to 24 weeks prior to finalizing restorative treatment (Figure 7). This technique can only be used infrequently in the mandible.
Roll Flap
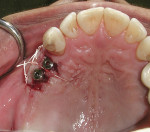
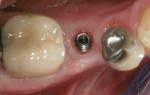
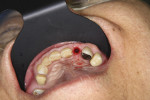
If an adequate width of KM exists on the buccal but there is a need to enhance its thickness (Figure 8), a roll flap uncovering12 can be used. A u-shaped, full-thickness incision is made around the proximal and palatal/lingual margins of the implant (Figure 9). The demarcated area is then de-epithelialized (Figure 10). This is reflected from the lingual/palatal side. A pouch is created with a micro-scalpel on the buccal aspect (Figure 12). The de-epithelialized pedicle is rolled into the pouch and is secured with an internal sling suture. Simple sutures are placed around the healing abutment to secure the buccal tissue (Figure 12).
Tissue Preservation Technique
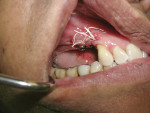
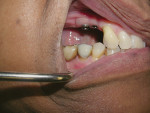
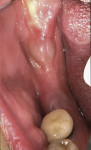
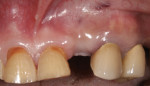
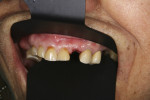
In the mandible, an inadequate band of KM—ie, less than 4 mm—is often present in an edentulous site (Figure 13). This narrow band of KM should be incised precisely in half. Next, a buccal and lingual reflection should be performed, and the flap margins repositioned bucally and lingually to a point 2 mm to 3 mm from the margins of the implant (Figure 14). Granulation and keratinization of the intervening connective tissue will result in an increased band of KM (Figure 15).
Free Connective Tissue Graft
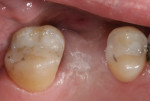
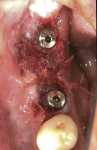
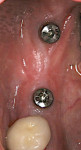
Where a large amount of tissue augmentation is needed (Figure 16), a free connective tissue (CT) graft can be harvested from the palate or maxillary tuberosity.13,14 This is placed under a partial or full-thickness buccal flap (Figure 17) or pouch as described by Kan et al.15 The grafted tissue must be immobilized with internal periosteal sutures or cross-mattress sutures prior to flap closure (Figure 18). Figure 19 shows the peri-implant tissue development at 4 months. (It should be noted that this procedure may be utilized at any stage of treatment and is not limited to a “two-stage” protocol.)
Pedicle Flap
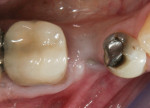
In the maxilla, where a large amount of soft tissue must be augmented (Figure 20), it is possible to use a large connective-tissue pedicle rather than a free CT graft.16,17 The use of pedicle flaps preserves vascularization, which is critical to graft survival and helps in reducing graft shrinkage.18 The pedicle flap is started similarly to the roll flap but is extended further and wider onto the palate (Figure 21), and a subepithelial connective tissue dissection is done without separating the buccal aspect. It may be possible to use a “single-incision” approach in performing the CT dissection.19 Once reflected, the connective tissue pedicle is rolled into a buccal pouch and secured with an internal sling suture (Figure 22), and the palatal epithelium is closed with simple interrupted sutures (Figure 23). At 3 months, there is a healthy band of thick KM (Figure 24). The buccal view in Figure 25 demonstrates the “virtual” root form that has been created.
Conclusion
Today, it is widely recognized that increased width and thickness of keratinized mucosa around dental implants is associated with better long-term success than implants without KM. This is especially important in the esthetic zone, where marginal recession can be of concern. Several studies have demonstrated the long-term stability of the implant/tissue interface when KM is present.20,21 As well, tissue augmentation around implants has been shown to have a high degree of long-term predictability.22 This article has presented several methods of implant uncovering, with an emphasis on enhancement of the keratinized mucosa.
About the Author
David E. Azar, DDS
Private Practice
New York, New York
References
1. Sennerby L, Roos J. Surgical determinants of clinical success of osseointegrated oral implants: a review of the literature. Int J Prosthodont. 1997;11(5):408-420.
2. Smith DE, Zarb GA. Criteria for success of osseointegrated endosseous implants. J Prosthet Dent. 1989;62(5):567-572.
3. Fürhauser R, Florescu D, Benesch T, et al. Evaluation of soft tissue around single-tooth implant crowns: the pink esthetic score. Clin Oral Implants Res. 2005;16(6):639-644.
4. Block MS, Kent JN. Factors associated with soft- and hard-tissue compromise of endosseous implants. J Oral Maxillofac Surg. 1990;48(11):1153-1160.
5. Yeung, SC. Biological basis for soft tissue management in implant dentistry. Aust Dent J. 2008;53(suppl 1):S39-S42.
6. Boynueğri D, Nemli SK, Kasko YA. Significance of keratinized mucosa around dental implants: a prospective comparative study. Clin Oral Implants Res. 2013;24(8):928-933.
7. Warrer K, Buser D, Lang NP, Karring T. Plaque-induced peri-implantitis in the presence or absence of keratinized mucosa. An experimental study in monkeys. Clin Oral Implants Res. 1995;6(3):131-138.
8. Zigdon H, Machtei EE. The dimensions of keratinized mucosa around implants affect clinical and immunological parameters. Clin Oral Implants Res. 2008;19(4):387-392.
9. Kan JY, Morimoto T, Rungcharassaeng K, et al. Gingival biotype assessment in the esthetic zone: visual versus direct measurement. Int J Periodontics Restorative Dent. 2010;30(3):237-243.
10. Kan JY, Rungcharassaeng K, Morimoto T, Lozada J. Facial gingival tissue stability after connective tissue graft with single immediate tooth replacement in the esthetic zone: consecutive case report. J Oral Maxillofac Surg. 2009;67(11 suppl):40-48.
11. Tinti C, Parma-Benfenati S. Coronally positioned palatal sliding flap. Int J Periodontics Restorative Dent. 1995;15(3):298-310.
12. Barone R, Clauser C, Prato GP. Localized soft tissue ridge augmentation at phase 2 implant surgery: a case report. Int J Periodontics Restorative Dent. 1999;19(2):141-145.
13. ten Bruggenkate CM, Krekeler G, van der Kwast WA, Oosterbeek HS. Palatal mucosa grafts for oral implant devices. Oral Surg Oral Med Oral Pathol. 1991;72(2):154-158.
14. Silverstein LH, Lefkove MD. The use of the subepithelial connective tissue graft to enhance both the aesthetics and periodontal contours surrounding dental implants. J Oral Implantol. 1993;20(2):135-138.
15. Kan JY, Rungcharassaeng K, Lozada JL. Bilaminar subepithelial connective tissue grafts for implant placement and provisionalization in the esthetic zone. J Calif Dent Assoc. 2005;33(11):865-871.
16. Nemcovsky CE, Artzi Z. Split palatal flap. I. A surgical approach for primary soft tissue healing in ridge augmentation procedures: technique and clinical results. Int J Periodontics Restorative Dent. 1999;19(2):175-181.
17. Nemcovsky CE, Artzi Z. Split palatal flap. II. A surgical approach for maxillary implant uncovering in cases with reduced keratinized tissue: technique and clinical results. Int J Periodontics Restorative Dent. 1999;19(4):385-393.
18. Sclar A. Soft Tissue and Esthetic Considerations in Implant Therapy. Quintessence Publishing Co.; 2003:165.
19. Hürzeler MB, Weng D. A single-incision technique to harvest subepithelial connective tissue grafts from the palate. Int J Periodontics Restorative Dent. 1999;19(3):297-287.
20. Speroni S, Cicciu M, Maridati P, et al. Clinical investigation of mucosal thickness stability after soft tissue grafting around implants: A 3-year retrospective study. Indian J Dent Res. 2010;21(4)474-479.
21. Schrott AR, Jimenez M, Hwang JW, et al. Five-year evaluation of the influence of keratinized mucosa on peri-implant soft-tissue health and stability around implants supporting full-arch mandibular fixed prostheses. Clin Oral Implants Res. 2009;20(10):1170-1177.
22. Kan JY, Rungcharassaeng K, Lozada JL, Zimmerman G. Facial gingival tissue stability following immediate placement and provisionalization of maxillary anterior single implants: a 2-to 8-year follow-up. Int J Oral Maxillofac Implants. 2010;26(1):179-187.