Taking Action
State legislation to elevate the profession and crack away at the gray market gains momentum
Jason Mazda
When Washington Gov. Jay Inslee officially signed HB 1177 on April 19, 2019,1 Matthew Chapman, CDT, and his colleagues in the Washington State Dental Laboratory Association (WSDLA) were finally able to celebrate. The bill, officially described as "creating the dental laboratory registry within the Department of Health and establishing minimum standards for dental laboratories serving dentists in Washington state,"1 was the culmination of several years of hard work by the WSDLA membership. Washington became one of just a handful of states to have implemented the American Dental Association's (ADA's) policy on dental laboratory registration and disclosure.2 While HB 1177 is a patient advocacy bill at its heart, it also was a significant step toward elevating the dental laboratory profession as well as creating a more level playing field nationwide for US laboratories competing against the gray and black markets.
"Now, we are riding the momentum that Washington created," says Eric Thorn, In-House Counsel for the National Association of Dental Laboratories (NADL).
HB 1177 established four basic requirements for all dental laboratories serving dentists in the state:1
• Register with the Department of Health beginning in 2020
• Disclose to the dentist what materials are in each device
• Disclose to the dentist the city, state, and country where the device was made
• Have the owner or at least one employee complete 12 hours of CE annually or be a CDT beginning in 2021 (by 2025, one CDT per laboratory will be the requirement, with a grandfather clause exempting those who have owned their laboratories for at least 20 consecutive years)
"The registration of dental laboratories and state oversight regulatory authority have the potential to enhance patient health and safety," Inslee says, "and better ensure that laboratories are operating under high standards consistent with other clinical practices."
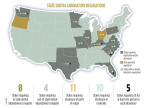
The concept seems so obvious that a 2009 ADA member survey indicated nearly 65% of dentists believed dental technicians and laboratories already were regulated in their state.2 In 2011, Pankey Institute President and CEO Ricki Braswell, CAE, spoke of "a growing national trend of dental societies working to establish basic minimum standards for dental laboratories."3 However, Washington became just the eighth state to require laboratories to register, and the fourth to require out-of-state laboratories to register.2 It is the 11th state to require point-of-origin disclosure, the 10th to require material disclosure, and the fifth to require CE for at least one person in each laboratory.2 State registration fees range from $25 (Pennsylvania) to $300 (Oklahoma) per year.2
Several other states have explored similar measures, some with more success than others. Oregon seems poised to be next, following in its neighbor's footsteps. Ohio is in the early stages of expanding on regulations that already include disclosures.
The process of getting dental laboratory legislation passed is arduous, and not everyone experiences the same level of success that those in Washington did. However, understanding their experiences could help others in the future.
Why Change?
The ADA policy on dental laboratory registration and disclosure states in part that, "in order to enhance dental patient health and safety, the ADA urges all state dental boards to register US dental laboratories;" "the laboratory/technician should inform the dentist of the materials present in the case;" and "the laboratory/technician should inform the dentist of any subcontracting laboratory/technician employed for preparation of the case."4
With the ADA's position clear, the next question is whether dental laboratories do, in fact, desire regulation. Support for this concept is not universal—a 2012 thread on the Dental Lab Network message boards shows several people who were unhappy with regulations being established in Virginia5—but it is strong. A survey of laboratories commissioned by the NADL last year and provided to IDT for this article found that 75% agree that laboratories should be required to register with a state agency, and 83.5% agree that dental laboratories should be required to employ at least one CDT. Only 12% and 2%, respectively, disagreed with those notions (the rest were neutral).
"State regulation helps to level the playing field for all laboratories," says Travis Zick, VP and COO of Apex Dental Laboratory Group and the 2019 NADL President. "Material and point of origin disclosure requirements are absolutely imperative, and they should be enforced in every state right now regardless of registration. Dentists should know what they are putting in a patient's mouth, who made it, and where it came from. This would eliminate or severely restrict the opportunity for laboratories to ‘secretly' send their work to other countries and represent that they are doing it themselves. Registration legislation should include these requirements.
"Furthermore, increased regulation adds legitimacy to our industry."
The Oregon Association of Dental Laboratories (OADL) is in the process of surveying its membership this month, but OADL Executive Director Carrie Peil-Warner, CDT, says that a town hall meeting and an open panel discussion at last year's annual meeting indicated strong support.
"I have heard the statements, ‘This is just another fee,' and, ‘This will not help me,'" Peil-Warner says. "However, that opens the topic for discussion, and once we talk about what we are really trying to achieve, those positions tend to change."
Peil-Warner characterizes regulations as a win-win for both dentists and laboratories. She says her own business, Birch Dental Laboratory, would benefit.
"We want our dentists to feel confident they are working with reputable laboratories that are educated, that disclose what the product is, that disclose point of origin, and that are playing by a particular rule set they feel confident supports the kind of dentistry they want to practice," Peil-Warner says. "We benefit from it because it elevates our professionalism and how we feel about what we do in our everyday grind. It is another formal way to recognize a laboratory as being truly professional. As a laboratory owner, I want my clients and their patients to know that I am required to give them a great product."
Gary Iocco, Chairman of Apex Dental Laboratory Group and a champion of legislation in Minnesota, notes that if laboratories want to be respected by dentists, then this is a necessity.
"You cannot say, ‘I want to be treated like an ultimate professional,' but not register or take CE," Iocco says.
Indeed, the current US Bureau of Labor Statistics job classification system assigns higher-ranking "job zones" to occupations that require either licensure or mandatory certification.6 Thorn says that while that is not the primary motivation for the NADL's support of state legislation, widespread regulation across the country could spur a change in job zone classification.
"Technicians hoped to be categorized in the same job zone as dental hygienists, which is one zone higher than where they are currently," Thorn says. "The different job zone classifications are based on required amount of training, education, etc."
Beyond that, Iocco notes that registration databases would help in the event of product recalls. Ohio Dental Laboratory Association (ODLA) President Lonni Thompson, CDT, adds that such databases would offer significant benefits in implant dentistry as well.
"We are already experiencing cases of patients having implants placed 20 years ago, and we are scrambling to find parts and to restore them," Thompson says. "Accurate documentation needs to be established to make sure we can restore implant cases properly in the future. Laboratories are dwindling rapidly. Currently, there is not an accurate way of finding many laboratories. Registration will help documentation for when patients need problems fixed for what we are creating for them today."
Regulations also theoretically should hinder the growth of the gray and black markets—though that will depend on how stringently the laws are enforced. In some states that have regulations on the books, enforcement is challenging. For example, in Virginia, the Board of Dentistry insisted that dentists, not laboratories, be responsible for ensuring the point of origin on prescriptions. Legislation in Washington and perhaps Oregon could prove more effective, but Peil-Warner notes that only time will tell.
Washington's Blueprint
Washington's pursuit of legislation was ongoing for approximately 7 years, Chapman says. The process began when a piece of legislation was proposed by the Washington State Dental Association (WSDA) that would have required laboratories to preauthorize work before it arrived to them. According to Chapman, the president of the Washington State Dental Laboratory Association (WSDLA) at the time was asked for input and said the paperwork required would have a significantly negative effect on laboratories.
"The light went on as we realized that we did not have any voice at the table and were fairly vulnerable as a result," Chapman says.
In 2014, then-WSDLA President Mike Dominguez, CDT, met with the WSDA and found a resounding desire for some form of regulation of laboratories. The WSDLA's membership subsequently voted to pursue the issue.
The first step was researching options, including regulations in other states. Chapman says a decision was made not to pursue licensure because the state had tried that in 1989 and failed. Registration, point of origin disclosure, and materials disclosure were identified as key issues.
Once that basis had been established, WSDLA board members requested a meeting with the WSDA's executive board. With Thorn's help, they drafted a proposal.
"They were not only intrigued, but they actually helped us with some of the verbiage, including a grandfather clause to make sure well-established laboratories that did not have CDTs would not be put out of business," Chapman says. "That not only improved the proposal but also gave them some ownership of it."
The WSDLA initially proposed passing it by rule, but the Department of Health declined. Thorn then helped them formulate the verbiage into a bill, and the WSDLA hired a lobbyist to help move it into the state legislative process.
Chapman and a few others joined members of the WSDA for Dental Action Day, an annual effort when Washington dentists lobby for dental causes in the capitol.
"We were very fortunate to have the level of teamwork that we did from the WSDA," Chapman says.
While the bill received wide support from both sides of the aisle and passed in the House with a 96-1 vote, it did not reach the Senate floor the first year. Some legislators were slow to act. Lobbyists representing corporate dentistry and denturists raised some issues with the bill, resulting in some changes to the verbiage.
"We were pretty naïve to think we could get it through the first year," Chapman says.
WSDLA members remained in contact with the WSDA and again participated in Dental Action Day the following year. They emphasized that their bill was about patient safety and transparency.
"We spent a lot of time educating lawmakers about what we were trying to accomplish," Chapman says. "Even the Department of Health had nothing on the books defining a dental laboratory. We had to educate people on the industry and the fact that we had no mandatory credential in our state. Many of them had either assumed otherwise or never thought about it—so much so that we needed to be careful in our approach because there was some talk of requiring every laboratory employee to register with the state, and we definitely did not want that."
Republican lawmakers, in particular, needed to be persuaded of the necessity for new regulations when there was no major incident that had prompted change. Chapman says one legislator in particular was adamant about not wanting to expand government unnecessarily.
"He took us to task," Chapman says, "and we explained that we did not have a smoking gun because issues get swept under the carpet and fixed. We explained the gray and black markets to him, but still he viewed the registration fee as a tax that would burden small businesses. I told him I was sensitive to that as an owner of a five-person laboratory, but the law requires a regulation like this to fund itself, so the fee was based on the estimated administrative costs. Eventually this gentleman was convinced that the necessity for regulation outweighed the impact of the fee. He even spoke in favor of our bill on the floor, which was powerful because it showed that he understood the importance of what our industry does for our clients and ultimately for our patients."
Thorn says it often is a matter of dispelling misperceptions that the regulations will be too cumbersome or that any legislation could create a slippery slope toward more invasive regulations in the future or that it could increase costs.
"Laboratories would not be asking for this if it were going to be expensive to comply or expensive for their customers to use their services," Thorn says.
The WSDLA's bill again passed in the House in that second year, but even with widespread support it did not reach the Senate floor.
"It would have passed, but some higher-profile issues in our state overshadowed our bill," Chapman says. "A three-page bill can be overlooked easily."
After 2 years, it becomes necessary to get a new bill number and sponsor. Fortunately, Rep. Monica Jurado Stonier reaffirmed her commitment to championing the WSDLA's bill.
"It was a complete do-over," Chapman says, "but we had laid the groundwork for success."
On January 16, 2019, the bill was read in the House and referred to the House Committee on Health Care & Wellness.1 Immediately prior to a February 6 vote, Rep. Stonier told fellow committee members simply, "It's a good bill. It's a patient protection bill." The committee voted unanimously on a "do pass" recommendation, and the bill was referred to the House Committee on Appropriations on February 11 and then referred to the Rules Committee.1 On March 7, it passed with 97 yeas and 0 nays.1 Two days later, it was read in the Senate, and on March 18, Rep. Stonier told the Senate, "This is a bill that very simply provides some patient protections and safety by making sure that dental laboratories know what is in the product and where they are coming from." After moving through two Senate committees, the bill passed on April 10 with 46 yeas and 0 nays.1 Inslee ultimately signed it on April 19, effective July 28.1
"I "I was very proud that it passed with 0 nays," Chapman says. "We worked hard to educate people about our cause, and it paid off."
Positive Momentum
Soon after the bill passed, Chapman and 2019 WSDLA President Megan Nakanishi traveled to visit with leaders of the OADL, which hopes to use Washington's bill to gain credibility with the Oregon legislature.
"We told them, ‘Here is the playbook that worked for us,'" Chapman says.
Peil-Warner says she and other members of the OADL had long believed dental laboratory regulation would be a positive development in any state, but Washington's success prompted them to take action.
"They really inspired us," she says. "What they did was really clean, simple, and achievable, even though it took some time."
Assuming the OADL's membership survey confirms a desire to move forward, the plan is to request a formal letter of support from the Oregon Dental Association (ODA), and perhaps even further efforts on their part. Peil-Warner says the OADL and ODA have a strong relationship and she expects a positive response.
Once the ODA is on board, the OADL plans to submit the bill to the Oregon Legislative Assembly for its 2021 session. The assembly meets for 160-day sessions in odd-number years, as opposed to 35-day sessions in even-number years.7
"Aiming for the 2021 session allows us to feel that we have done our due diligence and not rushed anything," Peil-Warner says. "We want to ensure that our bill says exactly what we want it to say and that we do everything correctly."
The bill likely will be very similar to Washington's, if not identical, Peil-Warner says. If it passes, then the OADL hopes to help other states in the same way Washington has helped Oregon.
"The WSDLA's willingness to give us their entire toolkit—what they did, how they did it, and the whole process—was key to Oregon's fledgling idea that we might want to do this," Peil-Warner says. "We saw that it can be done."
The Ohio association hopes to be next. The state already requires material disclosure and point of origin disclosure,2 but ODLA leaders have been working diligently on a draft bill that is more consistent with Washington's HB 1177, Thompson says. The stumbling block currently is coming to terms with the Ohio Dental Association on how this change can benefit all parties without becoming burdensome, Thompson says, but the ODLA plans to continue its efforts this year.
"Our membership is united because we see the results of what is taking place in our profession," Thompson says. "It is an exciting time to be in dentistry with the shift from analog to digital, but it is expensive and there is a learning curve. Possibilities are endless for what the future holds, but dentists and laboratories are a team, and we need to depend on each other; we should help one another be accountable to our patients."
Peil-Warner hopes that if Oregon can achieve success similar to Washington's, then their examples could help other states such as Ohio.
"If more states can do it, then the process could become streamlined to the point where it might move more quickly and smoothly," she says. "It would be really cool if it snowballs and gains momentum."
Potential Pitfalls
The two most significant obstacles to streamlining the process are legislative complications and opposition from the clinical side. The WSDLA faced both challenges but overcame both. Others have faced more damaging resistance.
Minnesota passed legislation in 2012 to establish requirements for laboratories to register, for dentists to use registered laboratories (only for laboratory work done in the state), and for disclosures of materials and point of origin.8 Iocco, the driving force behind that legislation, was somewhat disappointed with the process.
First, Iocco needed to convince the Minnesota Dental Association (MDA) of the bill's value. The first question the MDA's lobbyists asked was, "How much will this cost our members?" They needed to be assured that laboratory fees for crowns, dentures, and other restorations would not be impacted before finally supporting the bill.
Like Chapman, Iocco also needed to convince lawmakers that the regulations were necessary.
"The biggest challenge is that one side of the aisle does not want any more regulations," Iocco says, "and the other believes there are not enough."
Iocco made his point by noting to legislators that his handkerchief and the pens they were using said where they were made, but a medical device going into someone's body is not required to.
"It was a long process," he says. "Senators and representatives have so many bills in front of them that their legislative assistants read them and give reports. I met with those legislative assistants, and once I explained the situation, the typical reaction was, ‘Wait a minute—you don't register or have a license now?'"
Even once the bill had widespread support, however, it stalled in a committee in the Minnesota House of Representatives in 2011, with no reason provided, according to Iocco. By the time the bill was pulled from that committee and attached to another bill to send through a different committee,8 it got stuck behind one other bill that was filibustered on the final day of the legislative session, Iocco says; when it was reintroduced the following year, it was met with new resistance.
"The legislative committee was hesitant about the implications for out-of-state laboratories," Iocco says, "so they eliminated the CE/CDT requirement before ultimately passing it. They also changed the enforcement end of it, removing the felony charges for dentists who use unregistered laboratories."
Around the same time as Iocco was championing that legislation, Dag Zapatero, DDS, MAGD, was working toward something similar in Virginia. Motivated by statistics showing a declining dental technician workforce and a simultaneous increase in the number of Chinese laboratories, as well as by national pride, Zapatero worked with Thorn and the Virginia Dental Association Task Force to steward the Virginia Dental Laboratory Safety Act through the Virginia Dental Association House of Delegates in 2011.9 The legislation would have required similar measures to those in Washington's bill. However, it encountered resistance from the Virginia Board of Dentistry.
"I shared with the Board that work is going overseas to companies that say they use FDA-cleared products, but nobody is checking or ensuring that those materials are ever used," Zapatero says.
The Board of Dentistry created a work group to discuss the matter, and the minutes from those meetings were provided to Inside Dental Technology. Department of Health Professions (DHP) Policy Analyst Elaine J. Yeatts "noted that the Board only has authority in Virginia and would not be able to travel out-of-state or out of [the] country to investigate complaints," to which Zapatero "responded that the Board could do paper investigations and have material assayed." Board of Dentistry Executive Director Sandra K. Reen cited timeline considerations. Board of Dentistry President Robert B. Hall Jr., DDS, suggested a study to help the Board "understand the problem being experienced," but Zapatero noted that at the VDA's annual meeting, attended by hundreds of Virginia dentists, members had voted "virtually unanimously," so there was "no need for a study to be done." Hall countered that he had "talked to five or six dentists in his area who are members of [the] VDA and who have stated that they do not understand the need for the bill."
According to subsequent meeting minutes, the Board ultimately decided to revise its existing Approved Template for Dental Laboratory Work Order Form and said that as a result no legislation was needed. Consequently, the burden of specifying the materials to be used and point of origin was placed on the dentist via special prescription requirements, but laboratories still are not required to provide any disclosure of whether those materials were actually used or where the restoration was actually fabricated.
"Virginia prescription rules require that the dentist authorize the use of laboratory subcontracting work," Zapatero says, "but that is hard to implement and determine in the digital age. According to the regulations, the dentist is responsible for ascertaining the point of origin and materials disclosures before putting a restoration inside a patient's mouth. How can that be enforced? Unfortunately, people who attempt to follow the rules end up frustrated because most laboratories and manufacturers are not aware of the disclosure requirements in Virginia, still leaving patients vulnerable."
Even Chapman cautioned about the changes that legislators can make to a bill during the process. The WSDLA was cognizant of that possibility when crafting its bill.
"Once you drop the bill, it is no longer your bill," Chapman says. "You just need to guide it the best you can down the hallway and pursue the core of what you envision. That is really, really important. We were very fortunate because our dental association's lobbyists spoke at every single committee hearing for us. They understood that there were no smoke and mirrors here, and we were trying to provide them with information that would only help them if something were to go awry. We all understood that it was a patient advocacy bill, not necessarily anything other than that."
Of course, even the most widely supported bill is not guaranteed to pass, as both Chapman and Iocco found when they ran out of time in legislative sessions.
"Occasionally, legislation dies for reasons unrelated to the merits of the proposal," Thorn says. "In Florida, we had one session when the person in line to be Senate President was accused of impropriety and ended up resigning, and then the Parkland school shooting happened, so during the time when bills typically would be going through the process, they were not, and a lot of legislation died that year."
Taking Action
Regardless of how smooth or challenging the process is in any given state, it will require a significant amount of work and initiative. In Virginia, Zapatero says, many laboratories voiced their support for his cause but did not back it up by attending legislative sessions.
"Laboratories need to speak up when they are not being treated fairly and when laws are not protecting them," Zapatero says. "I was telling people, ‘If you are a US company, this will be great. If you are sending work offshore, this will require disclosure. But neither side showed up at the table."
The NADL is ready and willing to help any laboratories that want to take action.
"We go where we are invited, and we respect each state's dental laboratories' rights to decide what is best for them, so we do not just tell them what they should do," Thorn says. "The first thing we do is put some information together and try to meet with that state's dental society. In Washington, it went swimmingly because it made sense to them. We make sure anything they are concerned about is carved out or addressed."
Thorn then can help craft the bill and provide guidance throughout the process as needed.
In states with dental laboratory associations, the leadership of those organizations is best equipped to start the process of pursuing new legislation. However, Thorn says the NADL would be eager to help laboratories in other states as well. According to NADL Executive Director Bennett Napier, 30 states are not currently represented by associations.
"If a nucleus of laboratories in a particular state asks us to meet to discuss implementing policies, we will provide time, subject matter, and financial support," Thorn says. "The initiative needs to be homegrown, but we can provide support in drafting something to fit into state law, helping them meet with the state's dental society, and more."
What the Future Holds
The impetus to take action could increase as technology and business models continue to evolve.
"Having laboratories registered is essential on many levels," Thompson says. "Laboratories are producing body parts, not widgets, helping patients with a necessary part of health. Dentists rely on laboratories to help educate them with this rapidly changing environment; then add the price war, and bad actors can appear without the capability of accountability. You can order any material off Amazon today, and it does not need to be FDA-approved or -cleared. Registration provides the path of making sure everyone is providing what the prescription form says because there is a measure of liability."
Zapatero already is pushing a second bill this year in Virginia, to regulate tele-dentistry. He opines that the gray and black markets in Asia and other places will no longer pose as much of a threat as new technology lowers costs and strengthens the US laboratory market.
"If I scan a patient's dentition and my prescriptions says, ‘Please print and mount models,' is that equivalent to submitting a prescription to pour and mount models from alginate impressions? I do not know," he says. "Most states do not have regulations governing scanning or defining if a scan is equivalent to a final impression if used in the fabrication of an appliance. Virginia is attempting to answer some of these questions. Do I need a prescription to have a laboratory print a model from a digital file? Can I give a scan file to my patient to print a model at home, if not used for diagnosis and treatment planning? We regulate the procedures used in the fabrication of an analog crown, but because we used a scanner to create an image file and a printed or digital model in its manufacturing, is the digitally produced crown then not restricted?
"SmileDirect can now print 50,000 unique models, used in the fabrication of its dental aligners, per day. Is SmileDirect a dental laboratory, and do they need to be regulated like one? What defines a dental laboratory, or even a laboratory technician, in the digital era? I believe these entities are dental laboratories. However, none of these processes exist in our regulations, and it creates loopholes in our profession."
The dental and dental laboratory professions working together will continue to drive future legislation in the industry that is advantageous for both sides.
"The key to future progress," Thorn says, "is for dentists to vocally urge their state dental society leaders and staff to meet with dental laboratory leaders in their state to develop a collaborative approach to ensure that laboratories are accountable for providing dentists with the information they need by establishing laboratory registration and disclosure requirements."
Indeed, Chapman notes that dentists and technicians are part of the same dental team.
"The most important thing for dentists to remember is that we are not their adversaries," Chapman says. "We rely on them for our income. We must view ourselves as specialists in the industry and act accordingly."