Hot Alone Will Not Do the Trick!
An analysis of the various elements contributing to the quality of a sintering furnace
By Manfred Herweg
Users of CAD/CAM systems for the production of full-contour zirconia restorations employ a sintering furnace in addition to a CAD scanner and a CAM milling machine. The requirements for processing such materials continue to change, along with the growing diversity of zirconia round blanks and coloring liquids. In addition to improvements in material characteristics to ensure the long-term service life of the finished product, the visual esthetics of the finished crowns, bridges, abutments, and frameworks play an equally essential role.
The temperature ranges for sintering are increasing and sintering times are decreasing. Temperatures for sintering zirconia ceramics may in the future range from below 1400°C (2552°F) to above 1600°C (2912°F). Today, the duration of the sintering cycle ranges from less than 15 minutes for individual crowns to overnight firings for complete restorations.
Sintering furnaces are exposed to extreme temperature ranges during their years of use. In addition, transport from the factory puts the mechanical parts under considerable strain in addition to stress on the electronics system, heating elements, and insulation of these high-temperature furnaces.
Heating Elements
Approximately 5,000 to 7,000 zirconia sintering furnaces are produced every year, and the numbers are rising. Two heating element types are installed in furnaces manufactured for the dental market. Most are built with molybdenum disilicide (MoSi2) heating elements. In some cases, silicon carbide (SiC) heating elements are used.
SiC heating elements usually are used up to 1530°C (2786°F), as recommended by the furnace manufacturer, and firing cycles take several hours. MoSi2 heating elements can be used up to 1750°C (3182°F), depending on the quality and furnace type. Additionally, their surface load-the power (W) that can be emitted per square centimeter (cm²)-is more than twice that of SiC, at above 25 W/cm2. This permits much faster heating-up rates and much higher application temperatures. Heating to 1550°C (2822°F) in less than 15 minutes is possible.
MolyCom®-Hyper 1800 (MoSi2) heating elements have been used on the international dental market since 2005 and have lasted much longer than SiC elements in continuous tests, according to the experience of various manufacturers. SiC elements usually need to be replaced after just a few hundred cycles due to the strong change of resistance. MolyCom-Hyper 1800 elements, by contrast, have been operated for more than 2500 cycles without any failure or need for exchange. In the event of a problem, exchange of the defective element is usually sufficient in MoSi2 heating elements. In contrast, all parts of the SiC elements must be exchanged due to the strongly impaired resistance.
Zirconia Discoloration
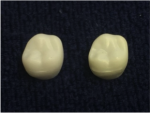
It is often said that zirconia structures suffer discoloration (Figure 1) from molybdenum (VI) oxide (MoO3), which can be released by MoSi2 heating elements. This is not accurate.
MoO3 is released via oxidation of MoSi2 at temperatures of 400°C (752°F) to 600°C (1112°F). Usually, the MoSi2 heating elements form a protective SiO2 layer on the surface that prevents oxidation of the basic material. MoO3 can be released when the SiO2 layer grows too thick and spalling results. Formation of a too-thick SiO2 layer is mostly prevented by additives in MolyCom-Hyper 1800 elements. If spalling does occur, regeneration firing at temperatures of 1450 °C (2642 °F) for several hours can be the solution. No other structures should be in the furnace for this firing procedure.
Testing has shown that MoO3 does not discolor zirconia. Many different oxides were dissolved in a liquid and dripped onto zirconia samples (Figure 2). The samples that were treated with MoO3 showed no discoloration after sintering (Figure 3). Samples treated with Al2O3, SiO2, Cr2O3, MgO, and in particular Fe2O3 did show discoloration. This demonstrated that the release of these oxides can contribute considerably to discoloration of zirconia. The visual impression was confirmed by a colorimeter.*
These discolorations occur when heating elements of standard MoSi2 are used in zirconia furnaces. MolyCom-Hyper 1800 heating elements are highly pure and have a much lower degree of contamination than standard elements, making it possible to nearly exclude contamination. MolyCom-Hyper 1800 Super Clean elements, meanwhile, have an even lower degree of contamination. Figure 4 shows samples after sintering with different MoSi2 heating element types. To increase contamination, the samples were sintered at temperatures of 1630°C (2966°F) and 1680°C (3056°F), which is far above the usual range. In a chemical analysis*, the standard heating elements showed a much higher degree of contamination, in particular by iron oxide. No contamination could be measured in the MolyCom-Hyper 1800 elements up to a temperature of 1630°C (2966°F). No contamination was measured for MolyCom-Hyper 1800 Super Clean elements up to 1680°C (3056°F).
Additionally, a contaminated zirconia sample on the surface was analyzed with a secondary ion mass spectrometry (SIMS).* Increased contents of iron oxide could be documented in the sample up to a depth of 0.15 µm. While the degree of contamination is very low, it has a considerable visual effect.
The vapor pressures of many different oxides at the surface temperatures of the MoSi2 heating elements in the furnace were calculated with FactSage (factsage.com) thermochemical software.* It becomes evident that MoO3, tungsten (VI) oxide (WO3), and iron oxide have high vapor pressures at the application temperatures in the dental furnace. Again, it becomes evident that iron oxide is the essential factor that contributes to contamination of the zirconia.
Color Liquids
The liquids often used to color zirconia contain metal salts and other dissolved compounds. Acids were once the common base for these liquids, but today most are water-based. Liquids that form many different oxides during firing, such as Fe2O3, CuO, MnO, Co2O3, Bi2O3, ZrO2, V2O3, CrO2, and others, are used to color the material. The zirconia blank is either soaked or painted with them. Sufficient drying of the blank after application of these liquids is very important. If liquid remains in the blank, it will evaporate while firing and the dissolved compounds will spread in the furnace, contaminating it. This affects the heating elements and insulation, and may cause severe contamination of the furnace, which in turn will discolor zirconia structures.
Furthermore, the inside of the furnace and the MoSi2 heating elements will be stressed as the protective layer of SiO2 will be destroyed, which in turn will reduce service life. To avoid this, structures can be fired in a closed saggar box, such as the UltraSaggar - ZTA 1600 from SCHUPP Ceramics. Made of zirconia-toughened alumina (ZTA), the saggar can be used even at heating-up rates of up to 100 K/minute. Tests were performed with zirconia samples fired in a closed saggar and on top of it in a contaminated furnace. The color differences between the samples were considerable.
Insulation
Very high demands are put on the insulation of a sintering furnace. The material is subject to extreme temperature changes and must resist temperatures of up to 1600°C (2912°F). As a result, manufacturers must observe very precise production accuracy. UltraBoards from SCHUPP Ceramics, insulation boards made of polycrystalline alumina wool (PCW), feature outstanding thermal shock resistance and low heat capacity, permitting even the fastest firing cycles of less than 30 minutes. It is recommended to not use insulation boards with refractory ceramic fibers (RCF), as they pose a health concern.
Additionally, a newly developed recipe permitted development of a nearly dust-free UltraBoard/UltraVac 1750/400PS (RCF-free) that withstands transport from the factory without any significant abrasion. The insulation structure must be considered in this as well. Backup insulation with an extremely low heat transmission factor permits using much thinner insulation than when using conventional refractory ceramic fibers.
SCHUPP Ceramics also has been offering complete furnace sets adjusted to the specific requirements of the dental market in cooperation with customers for more than 10 years. SCHUPP Ceramics can implement complex geometries with insulation materials that can be used at up to 1800°C (3272°F) and supply the necessary heating elements.
*Charts and graphs can be seen at insidedentaltech.com/idt923.
Manfred Herweg is the Technical Director for SCHUPP Ceramics in Aachen, Germany.
Disclaimer: The statements and opinions contained in the preceding material are not of the editors, publisher, or the Editorial Board of Inside Dental Technology.
SCHUPP Ceramics