Vertical Bone Augmentation Using Customized Titanium Mesh
Reduced operative times and greater stability contribute to better outcomes
Craig M. Misch, DDS, MDS | Maggie A. Misch-Haring, DMD, MS
The use of titanium mesh for the augmentation of atrophic jaws was first introduced by Philip J. Boyne, DMD.1 To reconstruct the jaws, the mesh was filled with cancellous bone from the iliac crest. Titanium mesh acts as a rigid protective matrix to maintain space and facilitate bone fill. The mesh lattice is porous and permits the passage of nutrients, growth factors, and cells as well as vascular ingrowth. This process is different from guided bone regeneration because mesh is not a cell-occlusive barrier.
In the past, a sheet of titanium mesh was trimmed and formed on a model or modified freehand at the time of surgery. But today, customized titanium mesh can be fabricated using additive manufacturing, which offers several advantages over the conventional method of freehand cutting and shaping. It reduces operative time and produces a form-stable mesh with a better fit, cleaner edges, and no sharp margins.2,3 These patient-specific meshes can be especially useful in managing longer spans and large bone defects.
Custom Fabrication
The workflow for the development of a customized titanium mesh starts with a cone-beam computed tomography (CBCT) scan of the patient's mandible and/or maxilla, depending on the location of the defect. The digital imaging and communications in medicine (DICOM) files are then uploaded to the manufacturer's platform, where digital technicians create virtual 3D models of the jaw. Next, implant planning software is used to virtually position the desired implant sites. With the implant positions planned, CAD software is then used to digitally reconstruct the ridge to possess ideal dimensions for implant support. The customized mesh is designed over the virtually augmented ridge in the software, which allows the clinician to choose the structure, thickness, grid, and sites for screw fixation. Because the improved fit of customized mesh makes it more stable, it may be possible to minimize the border extension and use fewer fixation screws.3 The design is then shared with the clinician for final approval.
Once the design is approved, the mesh is fabricated using direct metal laser sintering (DMLS) technology. In this process, a focused high-energy laser beam, is used to directly fuse a thin layer of metal powder into a 3D extended structure. The completed mesh is decontaminated in an ultrasonic machine, packaged in a clean room, and sent to the surgeon for final sterilization and clinical use. Although the cost of fabricating customized mesh is higher than implementing commercially available mesh, the advantages of reduced surgical time, better mesh stability, and reduced risk of exposure may justify the added expense.2,4
Surgical Placement and Retrieval
To restore an atrophic ridge with customized titanium mesh, a midcrestal incision is made over the ridge with vertical releasing incisions extending remotely from the graft site. The atrophic ridge is exposed, and the customized mesh is tried in to confirm the fit. Particulate autogenous bone is then harvested using bone scrapers or bone harvesting burs. In the maxilla, bone may be harvested from the zygomatic buttress or the tuberosity. In the mandible, the body and ramus are preferred donor sites when compared with the symphysis because they are associated with less morbidity. The addition of bone substitutes to particulate autogenous bone can help to expand graft volume and reduce graft resorption during healing. According to the literature, the ratio of autograft to bone substitute used is generally between 50:50 and 70:30.5,6 Anorganic bovine bone mineral is often selected for this graft mixture because it provides an osteoconductive matrix with low resorption qualities. To facilitate placement, the particulate graft may be mixed with platelet-rich fibrin (PRF) to create "sticky bone" and then condensed into the mesh. At least two buccal fixation screws are placed to secure the mesh, and an additional screw may be placed on the palatal or lingual aspect if needed for added stability.
Covering titanium mesh with a collagen membrane prior to flap closure can provide a layer of cushion, promote mucosal healing, and confer additional cell-occlusive properties.7,8 Placing a PRF membrane over the mesh has also been shown to reduce exposure rates.9-11 To obtain flap advancement over the mesh, techniques such as periosteal releasing incisions and submucosal dissection are performed, and then the flaps are closed using 4-0 or 5-0 vicryl or PTFE horizontal mattress sutures. Achieving tension-free primary closure is an important prerequisite to reduce the risk of exposure of the titanium mesh during healing.
Titanium mesh grafts are typically allowed to heal for approximately 6 months.6 As a general rule, vertical bone augmentation requires a longer healing period than horizontal bone augmentation. A CBCT scan can be used to assess the volume gained and plan for implant placement. Following healing, a mucoperiosteal flap is reflected to expose the mesh for removal. The recovery of titanium mesh can be time consuming because the soft tissues tend to grow into its open lattice. Customized mesh can be designed to be cut into two segments (buccal and palatal/lingual) to permit easier retrieval. Once the mesh has been removed, the underlying pseudoperiosteum is incised and reflected to expose the regenerated bone for implant placement.
Case Report
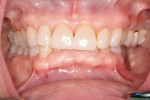
A 61-year-old female patient was referred to our office for a second opinion regarding re-treatment with dental implants. Approximately 10 years prior, she had two dental implants placed in the left mandible at the sites of teeth Nos. 19 and 20. The implants developed peri-implantitis, and progressive bone loss eventually necessitated their removal.
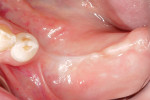
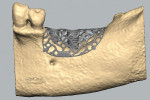
A clinical exam was performed, and a CBCT scan was acquired, which revealed a vertical bone defect in the left posterior mandible (Figure 1 and Figure 2). After the patient accepted a treatment plan involving ridge augmentation and implant placement, the DICOM files from the CBCT scan were sent for the design of a customized titanium mesh (Yxoss CBR®, ReOss) (Figure 3).
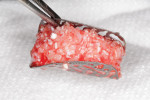
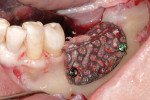
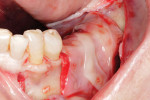
Upon approval of the design, the customized mesh was fabricated and delivered to the office for sterilization, and the patient was recalled. An incision was made along the crest of the ridge in the left posterior mandible. Vertical releasing incisions were then made lateral to the retromolar pad and mesial to the cuspid, and a mucoperiosteal flap was reflected to expose the ridge. After the customized mesh was tried in to confirm the fit, particulate autogenous bone was harvested from the mandibular body and ramus using a bone scraper (SafeScraper TWIST, Geistlich Pharma North America). The particulate autograft was mixed with a cancellous bovine bone mineral substitute (Bio-Oss®, Geistlich Pharma North America) and PRF to create a graft mixture, which was then loaded into the customized mesh (Figure 4). Once the mesh was filled with graft material, it was placed over the ridge and secured with two buccal fixation screws (Figure 5). The mesh was then covered with a resorbable collagen membrane (Bio-Gide®, Geistlich Pharma North America) as well as PRF membranes (Figure 6), and the flap was advanced to achieve tension-free primary closure using a horizontal mattress suture technique.
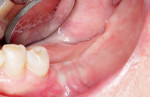
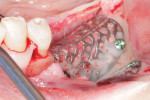
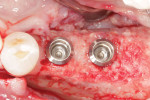
The patient healed uneventfully for 6 months and then returned to the office for an evaluation of the bone formation (Figure 7), which included a CBCT scan. A similar incision and flap design were used to expose and remove the mesh (Figure 8). With the vertical bone defect successfully reconstructed, two dental implants were placed (Straumann® Tissue Level Implant [4.1 mm × 8.0 mm], Straumann) (Figure 9).
Conclusion
The use of customized titanium mesh has improved the management of more complex bone defects of the jaw. Although customized mesh reduces surgical time and provides an improved fit with greater stability, vertical bone augmentation is a challenging surgical procedure that should only be performed by well-trained and experienced surgeons. Complications such as the opening of incision lines and mesh exposure are detrimental to graft success and may occur more commonly among procedures performed by novice clinicians.
About the Author
Craig M. Misch, DDS, MDS
Diplomate
American Board of Oral and Maxillofacial Surgery
Adjunct Clinical Associate Professor
Department of Periodontics
University of Michigan School of Dentistry
Ann Arbor, Michigan
Misch Implant Dentistry
Sarasota, Florida
Maggie A. Misch-Haring, DMD, MS
Specialist in Periodontics
Misch Implant Dentistry
Sarasota, Florida
References
1. Boyne PJ, Cole MD, Stringer D, Shafqat JP. A technique for osseous restoration of deficient edentulous maxillary ridges. J Oral Maxillofac Surg. 1985;43(2):87-91.
2. Sumida T, Otawa N, Kamata YU, et al. Custom-made titanium devices as membranes for bone augmentation in implant treatment: Clinical application and the comparison with conventional titanium mesh. J Craniomaxillofac Surg. 2015;43(10):2183-2188.
3. Sagheb K, Schiegnitz E, Moergel M, et al. Clinical outcome of alveolar ridge augmentation with individualized CAD-CAM-produced titanium mesh. Int J Implant Dent. 2017;3(1):36.
4. Zhou L, Su Y, Wang J, et al. Effect of exposure rates with customized versus conventional titanium mesh on guided bone regeneration: a systematic review and meta-analysis. J Oral Implantol. 2021;5. doi: 10.1563/aaid-joi-D-20-00200.
5. Cucchi A, Giavatto MA, Giannatiempo J, et al. Custom-made titanium mesh for maxillary bone augmentation with immediate implants and delayed loading. J Oral Implantol. 2019;45(1):59-64.
6. Chiapasco M, Casentini P, Tommasato G, et al. Customized CAD/CAM titanium meshes for the guided bone regeneration of severe alveolar ridge defects: Preliminary results of a retrospective clinical study in humans. Clin Oral Implants Res. 2021;32(4):498-510.
7. Degidi M, Scarano A, Piattelli A. Regeneration of the alveolar crest using titanium micromesh with autologous bone and a resorbable membrane. J Oral Implantol. 2003;29(2):86-90.
8. Cucchi A, Sartori M, Parrilli A, et al. Histological and histomorphometric analysis of bone tissue after guided bone regeneration with non-resorbable membranes vs resorbable membranes and titanium mesh. Clin Implant Dent Relat Res. 2019;21(4):693-701.
9. Torres J, Tamimi F, Alkhraisat MH, et al. Platelet-rich plasma may prevent titanium-mesh exposure in alveolar ridge augmentation with anorganic bovine bone. J Clin Periodontol. 2010;37(10):943-951.
10. Ghanaati S, Al-Maawi S, Conrad T, et al. Biomaterial-based bone regeneration and soft tissue management of the individualized 3D-titanium mesh: An alternative concept to autologous transplantation and flap mobilization. J Craniomaxillofac Surg. 2019;47(10):1633-1644.
11. Hartmann A, Seiler M. Minimizing risk of customized titanium mesh exposures - a retrospective analysis. BMC Oral Health. 2020;20:36.