Treating Anterior Lingual Gingival Recession Defects
SCTG with an envelope flap design provides solution in an area with limited access
Douglas H. Mahn, DDS
Mandibular anterior teeth are prone to high rates of plaque growth and supragingival calculus accumulation.1,2 Given the difficulty of gaining access for oral hygiene measures, the lingual surfaces of the mandibular incisors are a particular concern. A gingival recession defect (GRD) can act as a nidus for the aggregation of plaque and calculus. GRDs with an absence of attached keratinized gingiva are vulnerable to inflammation.3 In addition, exposed roots have the potential to develop caries.4 It has been proposed that a width of at least 2 mm of keratinized tissue, of which 1 mm is attached, is adequate for the average patient to maintain gingival health.3
Comparatively few reports focus on the treatment of GRDs affecting the lingual aspect of mandibular incisors. This may be due, in part, to the technical challenges involved in treating GRDs in this area. Nevertheless, GRDs with inadequate attached keratinized tissue require treatment because they can deteriorate if left untreated.5
The goal of soft-tissue grafting is to stabilize the soft-tissue foundation and, when possible, achieve root coverage. Although new and improved surgical techniques exist, complete root coverage is not always possible.6 However, an esthetic result is not necessary when treating the lingual aspect of anterior teeth. Several strategies have been implemented to address GRDs that affect the lingual aspect of mandibular incisors, and all of them possess both strengths and weaknesses. Considering the delicate nature of the soft tissues in this area, suitable technique selection is critical. The palatal subepithelial connective tissue graft (SCTG) is considered by some to be the gold standard.7,8 The following case report demonstrates the use of an SCTG to treat a GRD affecting the lingual aspect of a mandibular incisor.7,8
Case Report
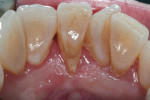
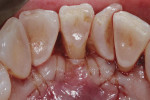
A 54-year-old female patient presented for the chief concern of a progressive GRD that was affecting the lingual aspect of tooth No. 24 (Figure 1). She did not report root surface sensitivity. The clinical examination revealed that tooth No. 24 had a lingual GRD of approximately 3 mm. The straight lingual probing depth was 3 mm, and there was no attached keratinized tissue; however, teeth Nos. 23 through 26 did not exhibit mobility. Plaque and calculus accumulations were found to be more substantial at the lingual aspect of tooth No. 24 than in adjacent areas. After discussing the findings, treatment plan options, and risks, the patient agreed to have the lingual GRD on tooth No. 24 treated with an SCTG.
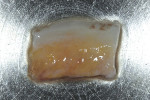
To begin, profound local anesthesia of the palatal donor site and treatment site was achieved using lidocaine HCL 2% with epinephrine 1:100,000. Incisions were made palatal to teeth Nos. 3 through 6 using a No. 15 scalpel blade (Bard-Parker® Stainless Steel Blades, Aspen Surgical Products), and an SCTG was harvested from beneath a palatal flap and trimmed to shape. The SCTG, which retained its epithelial surface, was approximately 2 mm × 6 mm × 12 mm (Figure 2). After the SCTG was acquired, a continuous 4-0 plain gut suture was used to secure the palatal flap of the donor site.
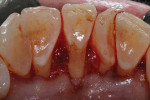
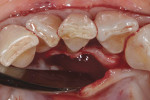
Using hand curettes, teeth Nos. 21 through 27 were cleaned, and their root surfaces were smoothed and flattened. Next, the No. 15 scalpel blade was used to make lingual intrasulcular incisions from teeth Nos. 22 through 26, and a 1/2 Orban periodontal knife was used to elevate a full thickness envelope flap (Figure 3). Once the Orban knife had released the flap and achieved flap mobility, the lingual root prominence and thin lingual plate of bone could be visualized (Figure 4). The SCTG was secured over the root of tooth No. 24 and the gingival flap was secured over the SCTG using a continuous suturing technique with a 4-0 chromic gut suture (Figure 5). Approximately 1.5 mm of the epithelial border of the SCTG was purposely left exposed.
The patient was prescribed amoxicillin (875 mg), which was to be taken twice a day for 1 week, and ibuprofen (600 mg) was prescribed for discomfort. The patient was instructed not to brush near the surgical site for 1 week. Instead, she would use a chlorhexidine gluconate 0.12% rinse twice per day.
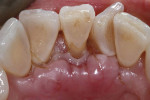
At the 1-week postoperative appointment, the treatment site was found to be healing well, and the chromic gut sutures were beginning to fall off (Figure 6). The remaining sutures were removed. For the next 5 weeks, the patient was permitted to brush the teeth associated with the surgical site but only gently and in an upward direction. After 6 full weeks of healing, she was allowed to resume normal tooth brushing and flossing.
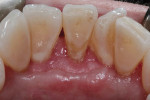
At the 12-week follow-up appointment, the treatment site was found to have healed well with complete root coverage and a broad zone of attached keratinized tissue (Figure 7). Overall, the mucogingival tissues appeared thicker than they did prior to treatment. The straight lingual probing depth was approximately 2 mm, shallow indentations demarcating the incision lines were visible, and the plaque accumulation at the site of tooth No. 24 appeared minimal and similar to adjacent areas. The patient was encouraged to maintain excellent oral hygiene.
Discussion
When selecting the manner in which to treat a mandibular lingual GRD, three key elements should be carefully considered. These elements are the accessibility of the site, the source of the graft material, and the type of suture material. Although these considerations may seem separate, each element must work in concert with the others.
Restricted mouth opening, narrow arch forms, tooth crowding, and retroclined teeth can limit access to the treatment site. Access to the treatment site will dictate the technique that is chosen and the instruments that are used. If the access is severely limited, great care must be taken when using a sharp instrument, such as a scalpel blade. An unfortunate slip with a scalpel blade into the fragile mucosal tissues can result in compromised flap perfusion, poor healing, and graft failure. In cases with limited access involving severely retroclined incisors, use of the Orban knife may be warranted.
Lingual GRDs have been treated with gingival augmentation using palatal autogenous grafts.9 With this technique, a split-thickness flap is used to expose a recipient bed around the GRD and prevent pulling of the mucosa. The autogenous graft is secured to the recipient site and left completely exposed. Close adaptation of the graft to the recipient bed and adequate immobilization of the graft are necessary for its success. Given the anatomy lingual to the mandibular incisors, this can prove to be difficult.
The treatment of lingual GRDs using a midline access incision, microsurgical blades, and specifically designed tunnel knives to create a subperiosteal tunnel has also been described.10 This SCTG technique requires adequate access to use the instruments involved. A double-crossed suture technique11 is also required. Despite success, this procedure has been described as high risk and accessible only to very experienced surgeons.10
Another approach, the connective tissue graft wall technique, was developed to treat deep lingual GRDs that affect mandibular incisors.12 The connective tissue graft wall technique involves placement of an SCTG below a trapezoidal-type coronally advanced flap. The SCTG acts as a lingual soft-tissue wall over the bone defect and root surface, and the lingual flap is coronally advanced and stabilized with lingual interrupted and sling coronal sutures. Adequate access and advanced suturing skills are required for this technique to be successful.
In the case demonstrated in this article, a lingual envelope flap design was used. At its coronal aspects, the flap was full thickness. At its more apical aspects, dissection into the mucosal tissues permitted its coronal advancement. Although the placement of intramarrow penetrations into the cortical bone to increase blood supply to the SCTG has been described,13 in this case, adequate graft perfusion from the overlying flap seemed sufficient for the success of the SCTG. In the treatment of lingual GRDs, Leaving the epithelial portion of the graft exposed has been shown to enhance the zone of keratinized tissue.14 As demonstrated by this article's case report, the SCTG survived, and the zone of keratinized tissue was increased.
Regarding the source of the graft material used, the use of acellular dermal matrices in the treatment of lingual GRDs has also been reported.15 Acellular dermal matrices must be covered because exposed portions can deteriorate. Because the SCTG develops a greater zone of keratinized tissue than the acellular dermal matrix,16 in cases where an increased zone of keratinized tissue is desired or concerns about graft exposure exist, the use of an SCTG would seem warranted.
Finally, the suture material must be considered. Suture materials can be divided into absorbable and nonabsorbable. Pulling of the mucosa at the lingual gingival margins of the mandibular incisors can be strong. Rapidly absorbable sutures, such as plain gut sutures, may not secure the graft site for an adequate period of time, but more slowly absorbing suture material, such as chromic gut sutures, can secure the graft site for a longer period of time to ensure proper healing. If any suture remnants prove difficult to remove entirely at the postoperative appointment, they will eventually fall out on their own. The speed at which chromic gut sutures are absorbed has been described as unpredictable.17 Synthetic nonabsorbable monofilament sutures, such as polypropylene, are durable and exhibit minimal tissue reactivity when compared with chromic gut sutures17; however, complete removal of nonabsorbable sutures can be difficult in areas with poor access. Choice of the ideal suture material may reasonably vary between cases and clinicians.
Conclusion
GRDs affecting the lingual aspect of mandibular incisors can be successfully treated with an SCTG. Clinicians have several surgical techniques available to them. Given the limited access to lingual GRDs, clinicians should carefully consider their options and select techniques and instruments based on the variables presented by each case.
About the Author
Douglas H. Mahn, DDS
Private Practice
Manassas, Virginia
References
1. Quirynen M, Dekeyser C, van Steenberghe D. The influence of gingival inflammation, tooth type, and timing on the rate of plaque formation. J Periodontol. 1991;62(3):219-222.
2. Dawes C. Why does supragingival calculus form preferentially on the lingual surface of the 6 lower anterior teeth? J Can Dent Assoc. 2006;72(10):923-926.
3. Lang NP, Löe H. The relationship between the width of keratinized gingiva and gingival health. J Periodontol. 1972;43(10):623-627.
4. Chi DL, Berg JH, Kim AS, et al. Correlates of root caries experience in middle-aged and older adults in the Northwest Practice-based REsearch Collaborative in Evidence-based DENTistry research network. J Am Dent Assoc. 2013;144(5):507-516.
5. Agudio G, Nieri M, Rotundo R, et al. Periodontal conditions of sites treated with gingival-augmentation surgery compared to untreated contralateral homologous sites: a 10- to 27-year long-term study. J Periodontol. 2009;80(9):
1399-1405.
6. Miller PD. Miller classification of marginal tissue recession revisited after 35 years. Compend Contin Educ Dent. 2018;39(8):514-520.
7. Langer B, Langer L. Subepithelial connective tissue graft technique for root coverage. J Periodontol. 1985;56(12):715-720.
8. Chambrone L, Chambrone D, Pustiglioni FE, et al. Can subepithelial connective tissue grafts be considered the gold standard procedure in the treatment of Miller Class I and II recession-type defects? J Dent. 2008;36(9):659-671.
9. Assis G, Nevins M, Kim DM. The use of autogenous gingival graft for treatment of lingual recession on mandibular anterior teeth. Int J Periodontics Restorative Dent. 2017;37(5):667-671.
10. Alves JP, Marques TM, Malta Santos NB, et al. Lingual incision subperiosteal tunnel access: proof of concept 18-month follow-up. Clin Adv Periodontics. 2019;9(4):196-202.
11. Zuhr O, Rebele SF, Thalmair T, et al. A modified suture technique for plastic periodontal and implant surgery--the double-crossed suture. Eur J Esthet Dent. 2009;4(4):338-347.
12. Zucchelli G, Bentivogli V, Ganz S, et al. The connective tissue graft wall technique to improve root coverage and clinical attachment levels in lingual gingival defects. Int J Esthet Dent. 2016;11(4):538-548.
13. Wilcko MT, Wilcko WM, Murphy KG, et al. Full-thickness flap/subepithelial connective tissue grafting with intramarrow penetrations: three case reports of lingual root coverage. Int J Periodontics Restorative Dent. 2005;25(6):561-569.
14. Kinaia BM, Kazerani S, Hsu YT, Neely AL. Partly deepithelialized free gingival graft for treatment of lingual recession. Clin Adv Periodontics. 2019;9(4):160-165.
15. Mahn DH. Correction of lingual gingival recession using an acellular dermal connective tissue allograft. Pract Proced Aesthet Dent. 2002;14(6):507-510.
16. Tal H, Moses O, Zohar R, Meir H, Nemcovsky C. Root coverage of advanced gingival recession: a comparative study between acellular dermal matrix allograft and subepithelial connective tissue grafts. J Periodontol. 2002;73(12):1405-1411.
17. Selvig KA, Biagiotti GR, Leknes KN, Wikesjö UM. Oral tissue reactions to suture materials. Int J Periodontics Restorative Dent. 1998;18(5):474-487.