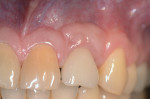
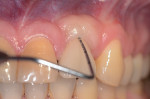
Peri-Implant Disease: A Primer
Etiology, prevention, and treatment
Paul S. Rosen, DMD, MS | Stuart J. Froum, DDS
Peri-implant diseases, which are defined as inflammatory processes in the tissues surrounding an implant, include peri-implant mucositis (ie, a soft-tissue inflammatory condition affecting implant sites with no signs of bone loss) and peri-implantitis (ie, an inflammatory condition affecting implant sites that is also characterized by loss of peri-implant bone).1 Although reported rates vary, studies have shown that peri-implant diseases are prevalent among patients who have received dental implant treatments. In a retrospective study that assessed implant recipients at 9- to 14-years postoperatively, peri-implant mucositis was present in 48% of the implants and peri-implantitis was identified in 6.6% of the implants.2 A separate study found that peri-implant mucositis occurs in approximately 80% of implant recipients and peri-implantitis occurs in about 28% to 56% of recipients.3 In addition, a systematic literature review concluded that, during the 5 to 10 years following implantation, the prevalence of peri-implantitis is 10% among implant sites and 20% among patients.4
These numbers imply a worrisome trend. An independent survey conducted in 2013 predicted that in the US dental implant market alone, more than 2.5 million dental implants would be placed in 2016.5 If 10% of these develop peri-implantitis, that would equal approximately 257,000 affected implants. Over a 5-year time period, that translates to 1.2 million diseased dental implants requiring treatment. To help reverse this trend, clinicians must be familiar with the etiology and risk factors for peri-implant mucositis and peri-implantitis as well as how to properly diagnose and treat these two clinical entities in their earliest stages.
Etiology of Peri-Implant Diseases
Peri-implant mucositis results from bacterial plaque buildup.6 Although reversible at the biomarker level, gingivitis and peri-implant mucositis require more than 3 weeks of resumed plaque control and healing to reestablish peri-implant mucosal health.7
Fortunately, much like teeth with gingivitis that might not advance in their disease severity to periodontitis, not all sites with peri-implant mucositis will progress to peri-implantitis.8,9 However, when peri-implant lesions advance, the compensatory mechanisms around implants are poor at limiting the inflammatory insult from spreading apically, which could lead to the loss of the implant in rather short order.10,11
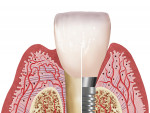
The entry of inflammatory cell exudate into the connective tissue and bone (Figure 1 and Figure 2) differentiates peri-implant mucositis from peri-implantitis.1 In a natural tooth, supracrestal transgingival and gingival fibers act as a defense mechanism to this inflammatory insult; however, none of these fibers are present around an implant (Figure 3). This allows the inflammatory cell exudate to progress unchecked toward the supporting bone, resulting in the loss of both the surrounding hard and soft tissues.1
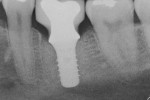
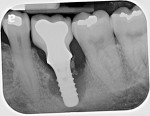
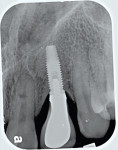
The rate of bone loss associated with peri-implantitis varies. In most individuals, this can occurs quite rapidly (Figure 4 through Figure 6).12 Because the loss of bone support can be characterized as being nonlinear in its progression, the necessity of early detection and treatment is all the more important.12
Risk Factors forPeri-Implant Diseases
Seven risk factors have been suggested to be associated with peri-implant diseases: oral hygiene, history of periodontitis, cigarette smoking, diabetes with poor metabolic control, alcohol consumption, genetic traits, and implant surface condition.3 The ability to clean around a dental implant is significant to the development of peri-implantitis. One study demonstrated that in situations where accessibility to cleaning was obviated, peri-implantitis developed in 48% of implants and that in situations where plaque control was possible, it developed in only 4% of implants.13 In a study by Serino and colleagues, among patients with unaddressed peri-implant mucositis, 44% of those implants went on to develop peri-implantitis within a 5-year period; among patients without peri-implant mucositis, 18% of the implants developed peri-implantitis.14 A history of periodontitis and cigarette smoking have also demonstrated an association with peri-implantitis that is unequivocal.4 In addition, although data is limited, research suggests that regardless of whether the surface is machined or roughened, no implant is immune to peri-implantitis.15
It is the opinion of the authors of this article that the list of risk factors for peri-implant diseases should be expanded to possibly include poor prosthetic fit and micromotion, poor implant positioning, occlusal disharmonies, retained cement, hypertension, inadequate gingiva, growth and development, reuse of healing abutments, and foreign particles (eg, cement and titanium).16-26
Poorly positioned dental implants and ill-fitting restorations can contribute to peri-implant diseases by interfering with daily hygiene efforts, allowing plaque to accumulate due to overhanging or open margins, and permitting exposed cement to be left behind that encourages pathogenic bacteria-all of which can result in increased inflammation and crestal bone loss,16,19-21 which can then further lead to exposure of the rough implant surface and greater plaque accumulation. Light marginal ridge contacts allow for food impaction and the development of traumatic inflammation in the peri-implant soft tissue. In these circumstances, the dynamics governing the relationships between teeth and implants may have been ignored.16,19
Poor positioning of a dental implant can lead to the development of disease and loss of the implant. When implants are improperly placed (eg, too far buccally), the results may include buccal bone resorption, subsequent recession, and exposure of rough implant surfaces-all of which encourage biofilm development and peri-implantitis.16,17 In addition, disregarding the potential need for ongoing occlusal adjustments to ensure favorable functional dynamics can further contribute to peri-implant bone loss by creating circumstances conducive to occlusal overload, implant loosening, crestal bone loss, and biofilm development leading to co-destruction.16,19 Unlike natural teeth, implants are incapable of physiologic movement to adapt to developmental and occlusal forces, thereby necessitating occlusal adjustments-when indicated-at each patient recall appointment.
Retained cement is a growing area of concern that is increasingly cited in the literature as a risk factor for peri-implant diseases.18,20,21 A retrospective in which an endoscopic examination was performed on dental implants with peri-implant disease revealed that 81% of the cases were associated with cement that required removal via a surgical flap approach.20 Following cement removal, 74% of those implants achieved disease resolution, leaving 26% with unresolved problems.20 In patients with a history of periodontitis, the presence of residual cement greatly increases the likelihood of developing peri-implantitis.21 In fact, one study found that among patients with residual cement and a history of periodontitis, 100% of these individuals developed peri-implant disease; by comparison, among residual cement cases without a history of periodontitis, significantly fewer developed peri-implant disease.21
The presence of adequate gingiva surrounding endosseous dental implants is essential in providing resistance to any inflammatory insults that may occur.24,25 Insufficient gingiva around dental implants appears to be associated with increased inflammation and diminished oral hygiene efficacy, resulting in increased plaque accumulation, recession, and attachment loss.24,25
Growth and development of the craniofacial region, which occurs throughout an individual's lifetime, is a consideration and possible risk for developing peri-implant diseases.22,23For example, vertical and horizontal changes can continue to occur beyond puberty,27 and over time, such growth and development can cause alterations in the relationships of the teeth and dental implants to one another. This may lead to the dental implants appearing labially angled or "intruded" relative to the adjacent teeth.
Preventing Peri-ImplantDiseases
Conscientious oral hygiene, proper implant spacing/placement, removal of residual cement, and ongoing professional maintenance are essential measures for preventing the occurrence of peri-implant diseases.28 In addition to screening implant candidates for other risk factors, ensuring that patients have plaque control and are capable of maintaining proper oral hygiene is essential.28 Adequate plaque control should be established prior to implant placement. In addition, the use of presurgical antibiotics, although still debated, has been shown to increase implant survival rates.28,29
To ensure proper implant spacing/placement, consideration should be given to the implant's ultimate 3-dimensional position and the use of a surgical template.17 Orofacially, at least 2 mm of bone on the buccal aspect of the dental implant is desired at the time of placement, and the implant's screw access should merge toward the cingulum of the future restoration. Mesiodistally, at least 3 mm of bone between implants and 2 mm between an implant and an adjacent tooth is recommended.17 From an apicoincisal perspective, if there is no gingival recession, placing an implant that is 2 to 3 mm apical to the cementoenamel junction of the adjacent tooth is advised.17 In the presence of recession, the implant should be placed 3 mm apical to the anticipated gingival margin, provided that this does not compromise esthetics that are acceptable to the patient.17
These exacting positions can be ascertained by working "from the top down." Collaborating with the restorative dentist to fabricate a stent of where the future teeth/restorations will be positioned enables accurate placement of the implant(s).17
Such an approach has implications for facilitating the removal of-if not altogether preventing-residual cement. The deeper the implant-supported restorations are, the more likely it is that residual cement will remain.19,20 Compounding this problem is that not all cements can be seen radiographically (eg, when deep and on the buccal aspect).27 Therefore, cementing crowns extraorally to extrude the cement prior to their final intraoral placement is advised.19,20,27
Diagnosing Peri-ImplantDiseases
Early and accurate diagnosis of peri-implant diseases is predicated on specific, reliable diagnostic indicators (eg, bleeding on probing, suppuration).30,31 Although debate continues regarding the necessity of probing dental implants, the absence of bleeding on probing around implants indicates healthy peri-implant tissue.30,32 When probing implants, a force of 25 Ncm is recommended. The soft tissue seal inhibits probing depths both in healthy and slightly inflamed peri-implant tissue, but not in tissues affected by peri-implantitis. Therefore, if bleeding on probing occurs and there are significant probing depths (ie, 5 mm to 8 mm), the site should be treated as soon as possible.31,32
Because an implant can lose as much as 70% to 90% of supporting bone yet still remain immobile, mobility is not a good diagnostic indicator of peri-implantitis.33 In addition, radiographs may produce false negatives and possess a low degree of sensitivity for detecting the earliest changes in bone level; thus, they are not an absolute diagnostic indicator for peri-implantitis.33
It is advisable to view radiographs over time to better observe any changes in bone level and determine not only if/when treatment should be initiated, but also whether the provided care has been successful in arresting the disease.31,33
Treatments for Peri-Implant Diseases
Successfully treating peri-implant diseases is dependent upon identification and classification of disease severity (ie, early, moderate, advanced). For example, an 8-mm long implant with 4 mm of bone loss is likely to be a more severe case than a 13-mm implant with the same amount of bone loss. Other indicators of disease severity include bleeding on probing, suppuration, and deep pocket depths.34-36
Peri-implant mucositis has typically been treated by eliminating biofilm, removing any residual cement, and administering adjunctive plaque control therapies.37,38 This may require administering local anesthesia, performing flap reflection, remaking the prosthesis with adequate embrasures, and/or repreparing the abutments to provide sufficient space for better at-home oral hygiene. However, studies have suggested that professionally administered plaque control efforts and adjunctive agents (eg, antimicrobials, systemic antibiotics) do not improve the efficacy of peri-implant mucositis treatment and that a low percentage of cases treated with adjunctive therapies have realized disease resolution.38,39
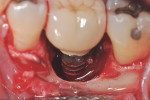
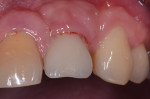
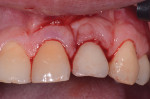
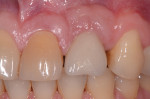
Fortunately, newer techniques such as laser-assisted peri-implant mucositis treatment may enhance the success of peri-implant disease treatments. Using an Nd:YAG laser and a laser-assisted peri-implant mucositis treatment procedure (LAPIMP™) has yielded promising results (Figure 7 through Figure 12).40-42
Treatment for peri-implantitis, on the other hand, can involve approaches ranging from mechanical debridement to explantation based upon the degree of pocket depth and bone loss.3,29 In particular, recommended treatments include mechanical debridement when pockets are 3- to 4-mm deep, additional use of an antiseptic when pockets are 4- to 6-mm deep, antibiotic therapy when pockets are > 6-mm deep, resective regenerative therapy when prior treatments are ineffective, and explantation when pockets are > 8-mm deep with bleeding and pain.3,29
Removal of the implant-along with rebuilding the site and placing a new implant-is commonly recommended when bone loss exceeds half of the implant length.32 Unfortunately, there is little validation in the literature to substantiate this recommendation.43 In fact, the available evidence does not support any specific recommendations for peri-implantitis therapy.43 Even the application of grafting materials and barrier membranes, which have resulted in pocket depth reduction and radiographic bone fill, lack high-quality comparison studies to support their efficacy, according to the systematic reviews.44
One retrospective study looked at the results of some of the more commonly used treatments for peri-implantitis.45 The overall success rate at the patient level was 69%.45 Interestingly, the effectiveness of the therapy was found to be impaired by severe periodontitis and compliance with maintenance, indicating that treating periodontitis prior to placing implants and emphasizing follow-up with maintenance on a tightly controlled interval are both essential.45
Access surgery combined with surface decontamination (eg, chemical agents, air abrasives, lasers) and antibiotics is also being used to treat peri-implantitis; however, the rate of successful resolution using this method is only about 60%.3,29 Consistent with other research regarding the treatment of peri-implantitis, no single method for surface decontamination has been found to be superior to others.3,46 At this time, all techniques and agents have been shown to be equally effective or ineffective for detoxifying contaminated implant surfaces.46
A Combination Approach
Recently, the literature has cited an approach for successfully managing peri-implantitis that combines mechanical and chemical methods for decontamination.36,40,41 The steps in the approach include critically evaluated case selection, flap access, defect debridement, surface decontamination, placement of a biologic on the implant surface, filling the defect with mineralized freeze-dried bone and/or using anorganic bovine bone coverage with an absorbable membrane or subepithelial connective tissue graft, and coronal positioning of the flap. A subsequent requisite step is professional maintenance.40,41 The successful outcomes achieved with this approach are largely credited to diligence in both case selection and in following the treatment protocol.36 For example, radiographic examination may reveal that the defect on the implant extends to the apex or that surface decontamination would need to go around the apex of the implant. In this case, surface decontamination alone could result in paresthesia; therefore, this represents a scenario in which implant removal may be prudent.40,41
Contributing to the success of the combined approach is the manner in which surface detoxification is completed.36 The authors' algorithm includes using air powder abrasion with glycine powder for 1 to 2 minutes, followed by thorough rinsing with a sterile saline or water for 1 to 2 minutes to wash away the powder. Although some concern exists regarding air embolisms resulting from the use of air abrasives, blocking off the flap with non-woven gauze during this procedure prevents the powder from reaching underneath the flap.40,41 After rinsing, cotton pellets are used to burnish the affected implant surface with minocycline, slurry, or citric acid (ie, 50% solution) for approximately 30 seconds to 1 minute. Following this step, the site is rinsed again with sterile water for 1 minute. This protocol recently received validation in vitro by demonstrating that it enabled improved attachment and proliferation of osteoprogenitor cells on a previously diseased implant surface when compared with implants that were untreated or those that only received mechanical debridement followed by sterile water application.47 At 3- to 7.5-year follow-up appointments, this combined approach to per-implantitis treatment demonstrated an increase of 3.7 mm of bone fill radiographically, more than 3 mm of new bone on sounding, a more than 5 mm average pocket depth reduction, and a more than 1 mm coronal apposition of the soft tissue.36
Conclusion
Despite the increasing prevalence of peri-implant diseases, the literature indicates that when problems such as bleeding on probing and bone loss are identified and treated in their early stages, the progression of disease can be arrested or ameliorated. Therefore, routine implant monitoring and maintenance are essential for preventing peri-implant mucositis and its progression to peri-implantitis. Moreover, before implants are placed, it behooves dentists to understand the risk factors for peri-implant diseases to avoid patient- and implant placement-related characteristics that could contribute to negative outcomes.
About the Author
Paul S. Rosen, DMD, MS
Clinical Associate Professor of Periodontics
University of Maryland Dental School
Baltimore, Maryland
Private Practice
Yardley, Pennsylvania
Private Practice
New York, New York
Stuart J. Froum, DDS
Clinical Professor and Director of Clinical Research
Department of Periodontology and Implant Dentistry
New York University College of Dentistry
New York, New York
Private Practice
New York, New York
References
1. Albrektsson TO, Johansson CB, Sennerby L. Biological aspects of implant dentistry: osseointegration. Periodontol 2000. 1994;4:58-73.
2. Roos-Jansåker AM, Renvert H, Lindahl C, Renvert S. Nine- to fourteen-year follow-up of implant treatment. Part II: presence of peri-implant lesions. J Clin Periodontol. 2006;33(4):290-295.
3. Lindhe J, Meyle J, Group D of European Workshop on Periodontology. Peri-implant diseases: Consensus Report of the Sixth European Workshop on Periodontology. J Clin Periodontol. 2008;35(8 suppl):282-285.
4. Mombelli A, Müller N, Cionca N. The epidemiology of peri-implantitis. Clin Oral Implants Res. 2012;23 (suppl 6):67-76.
5. iData Research, Inc. 2013.
6. Pontoriero R, Tonelli MP, Carnevale G, et al. Experimentally induced peri-implant mucositis. A clinical study in humans. Clin Oral Implants Res. 1994;5
w(4):254-259.
7. Salvi GE, Aglietta M, Eick S, et al. Reversibility of experimental peri-implant mucositis compared with experimental gingivitis in humans. Clin Oral Implants Res. 2012;23(2):182-190.
8. Marinello CP, Berglundh T, Ericsson I, et al. Resolution of ligature-induced peri-implantitis lesions in the dog. J Clin Periodontol. 1995;22(6):475-479.
9. Ericsson I, Persson LG, Berglundh T, et al. The effect of antimicrobial therapy on periimplantitis lesions. An experimental study in the dog. Clin Oral Implants Res. 1996;7(4): 320-328.
10. Lindhe J, Berglundh T, Ericsson I, et al. Experimental breakdown of peri-implant and periodontal tissues. A study in the beagle dog. Clin Oral Implants Res. 1992;3(1):9-16.
11. Gotfredsen K, Berglundh T, Lindhe J. Bone reactions at implants subjected to experimental periimplantitis and static load. A study in the dog. J Clin Periodontol. 2002;29(2):144-151.
12. Fransson C, Tomasi C, Pikner SS, et al. Severity and pattern of peri-implantitis-associated bone loss. J Clin Periodontol. 2010;37(5):442-448.
13. Serino G, Ström C. Peri-implantitis in partially edentulous patients: association with inadequate plaque control. Clin Oral Implants Res. 2009;20(2):169-174.
14. Costa FO, Takenaka-Martinez S, Cota LO, et al. Peri-implant disease in subjects with and without preventive maintenance: a 5-year follow-up. J Clin Periodontol. 2012;39(2):173-181.
15. Renvert S, Polyzois I, Claffey N. How do implant surface characteristics influence peri-implant disease? J Clin Periodontol. 2011;38(suppl 11):214-222.
16. Froum SJ, Rosen PS. A proposed classification for periimplantitis. Int J Periodontics Restorative Dent. 2012;32 (5):533-540.
17. Salama H, Salama MA, Li TF, et al. Treatment planning 2000: an esthetically oriented revision of the original implant protocol. J Esthet Dent. 1997;9 (2):55-67.
18. Linkevicius T, Vindasiute E, Puisys A, et al. The influence of the cementation margin position on the amount of undetected cement. A prospective clinical study. Clin Oral Implants Res. 2013;24(1):71-76.
19. Merin RL. Repair of peri-implant bone loss after occlusal adjustment: a case report. J Am Dent Assoc. 2014;1 45(10):1058-1062.
20. Wilson TG Jr. The positive relationship between excess cement and peri-implant disease: a prospective clinical endoscopic study. J Periodontol. 2009;80 (9):1388-1392.
21. Linkevicius T, Puisys A, Vindasiute E, et al. Does residual cement around implant-supported restorations cause periimplant disease? A retrospective case analysis. Clin Oral Implants Res. 2013;24(11):1179-1184.
22. Albert AM, Ricanek K Jr., Patterson E. A review of the literature on the aging adult skull and face: Implications for forensic science research and applications. Forensic Science International. 2007;172(1):1-9.
23. Daftary F, Mahallati R, Bahat O, Sullivan RM. Lifelong craniofacial growth and the implications for osseointegrated implants. Int J Oral Maxillofac Implants. 2013;28(1):163-169.
24. Gobbato L, Avila-Ortiz G, Sohrabi K, et al. The effect of keratinized mucosa width on peri-implant health: a systematic review. Int J Oral Maxillofac Implants. 2013; 28(6):1536-1545.
25. Lin GH, Chan HL, Wang HL. The significance of keratinized mucosa on implant health: a systematic review. J Periodontol. 2013;84(12):1755-1767.
26. Wilson TG Jr, Valderrama P, Burbano M, et al. Foreign bodies associated with peri-implantitis human biopsies. J Periodontol. 2015;86(1):9-15.
27. Wadhwani C, Rapoport D, La Rosa S, et al. Radiographic detection and characteristic patterns of residual excess cement associated with cementretained implant restorations: a clinical report. J Prosthet Dent. 2012;107(3):151-157.
28. Wilson TG Jr, Valderrama P, Rodrigues DB. The case for routine maintenance of dental implants. J Periodontol. 2014;85(5):657-660.
29. Lang NP, Lindhe J. Clinical Periodontology and Implant Dentistry. Vol 2. 5th ed. Oxford, UK: Wiley Blackwell: 2008. Chapter 60.
30. Mombelli A, Lang NP. Clinical parameters for the evaluation of dental implants. Periodontol 2000. 1994;
4:81-86.
31. Decker AM, Sheridan R, Lin GH, et al. A prognosis system for periimplant diseases. Implant Dent. 2015;
24(4):416-421.
32. Parma-Benfenati S, Roncati M, Tinti C. Treatment of peri-implantitis: surgical therapeutic approaches based on peri-implantitis defects. Int J Periodontics Restorative Dent. 2013;33(5):627-633.
33. Brägger U, Hugel-Pisoni C, Bürgin W, et al. Correl-ations between radiographic, clinical and mobility parameters after loading of oral implants with fixed partial dentures. A 2-year longitudinal study. Clin Oral Implants Res. 1996;7(3):230-239.
34. Okayasu K, Wang HL. Decision tree for the management of periimplant diseases. Implant Dent. 2011; 20(4):256-261.
35. Padial-Molina M, Suarez F, Rios HF, et al. Guidelines for the diagnosis and treatment of peri-implant diseases. Int J Periodontics Restorative Dent. 2014;34
(6):e102-e111.
36. Froum SJ, Froum SH, Rosen PS. Successful management of peri-implantitis with a regenerative approach: a consecutive series of 51 treated implants with 3- to 7.5-year follow-up. Int J Periodontics Restorative Dent. 2012;32(1):11-20.
37. Schwarz F, Becker K, Sager M. Efficacy of professionally administered plaque removal with or without adjunctive measures for the treatment of peri-implant mucositis. A systematic review and meta-analysis. J Clin Periodontol. 2015;42(suppl 16):
S202-S213.
38. Heitz-Mayfield LJ, Needleman I, Salvi GE, Pjetursson BE. Consensus statements and clinical recommendations for prevention and management of biologic and technical implant complications. Int J Oral Maxillofac Implants. 2014;29(suppl):346-350.
39. Hallström H, Persson GR, Lindgren S, et al. Systemic antibiotics and debridement of periimplant mucositis. A randomized clinical trial. J Clin Periodontol. 2012;39(6):574-581.
40. Froum SJ, Froum SH, Rosen PS. A regenerative approach to the successful treatment of peri-implantitis: A consecutive series of 170 implants in 100 patients with 2- to 10-year followup. Int J Periodontics Restorative Dent. 2015;35(6):857-863.
41. Froum SJ, Rosen PS. Reentry evaluation following treatment of peri-implantitis with a regenerative approach. Int J Periodontics Restorative Dent. 2014;34(1):47-59.
42. Nevins M, Nevins ML, Yamamoto A, et al. Use of Er:YAG laser to decontaminate infected dental implant surface in preparation for reestablishment of bone-to-implant contact. Int J Periodontics Restorative Dent. 2014;34(4):461-466.
43. Heitz-Mayfield LJ, Mombelli A. The therapy of peri-implantitis: a systematic review. Int J Oral Maxillofac Implants. 2014;29(suppl):325-345.
44. Chan HL, Lin GH, Suarez F, et al. Surgical management of peri-implantitis: a systematic review and meta-analysis of treatment outcomes. J Periodontol. 2014;85(8):1027-1041.
45. Lagervall M, Jansson LE. Treatment outcome in patients with peri-implantitis in a periodontal clinic: a retrospective study. J Periodontol. 2013;84(10): 1365-1373.
46. Suarez F, Monje A, Galindo-Moreno P, Wang HL. Implant surface detoxification: a comprehensive review. Implant Dent. 2013;22(5):465-473.
47. Rosen PS, Qari M, Froum SJ, Dibart S, Chou, LL. A pilot study on the efficacy of a treatment algorithm to detoxify dental implant surfaces affected by peri-implantitis. Int J Periodontics Restorative Dent. 2018;38
(2)261-267.