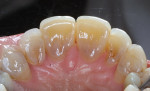
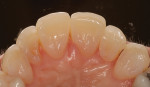
Bleaching Tetracycline-Stained Teeth
Considerations and recommendations for treatment
Van B. Haywood, DMD | Rhoda J. Sword, DMD
In order to understand how to successfully bleach tetracycline-stained teeth, one must first understand the basics of bleaching normal teeth. When considering bleaching, most dentists choose the most cost-effec-tive, safest, and efficacious technique for both the dental office and the patient, which is generally recognized to be overnight tray bleaching using a 10% carbamide peroxide bleaching agent—the most researched ADA approved bleaching material.1 In bleaching cases involving tetracycline-stained teeth, the extended time required for the teeth to change color makes choosing the appropriate bleaching material and technique even more important in terms of both cost and safety. When addressing patients, the word “bleaching” should be used instead of “whitening.” Although many over-the-counter products claim that they can produce “whitening” that rivals dentist treatments, “whitening” removes surface stains only, whereas “bleaching” changes the inherent color of the teeth. The following article compares and contrasts tray-bleaching techniques for normal teeth with those for tetracycline-stained teeth.
How Teeth Respond to Bleaching
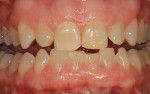
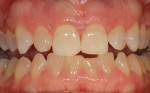
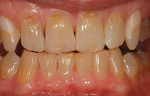
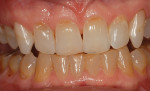
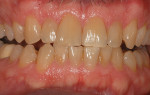
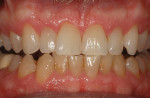
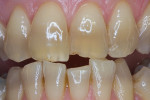
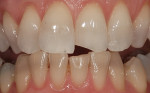
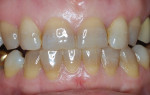
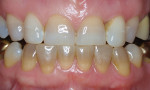
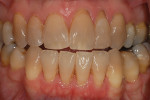
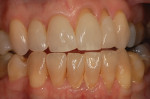
Every patient's teeth respond differently to bleaching, both in the level of bleaching that they obtain and how quickly that bleaching occurs. Depending on the final shade desired, overnight tray bleaching of normal discolored teeth takes approximately 3 days to 6 weeks with an average time of about 2 weeks.2 Comparatively, tetracycline-stained teeth can take 2 to 12 months or longer to bleach using the same method.3 Although they do not necessarily become “white,” the average time for tetracycline-stained teeth to lighten is 3 to 4 months (Figure 1 and Figure 2). The final outcome of bleaching tetracycline-stained teeth depends on their initial color and the consistency of the color. Tetracycline is available in several analogues, which induce different colors in the teeth. Gray discolorations are the hardest to bleach, whereas brown and yellow discolorations respond better. All teeth will lighten with bleaching; it is the extent to which they will lighten and the uniformity that is unpredictable (Figure 3 and Figure 4).
When bleaching treatment is initiated for normal teeth, there may be minimal color change for a day or two, then the teeth begin to significantly lighten. The teeth will continue to lighten until they reach an endpoint or plateau, which is determined by their composition, not the product or technique. This contributes to the unpredictable nature of bleaching. Once the teeth reach their maximum whiteness, any further treatment or use of different products/concentrations will fail to improve that shade. Teeth look the best when they match the color of the sclera of the eyes, which is the desired endpoint for bleaching.4 However, because the final shade is unpredictable, promising achievement of a super-light shade can result in patient dissatisfaction with the treatment.
When bleaching treatment is initiated for tetracycline-stained teeth, a slight color change may occur during the first few days, followed by no observable change for a month or so of treatment. After approximately 1 to 2 months, the teeth will begin to lighten in the same manner as normal teeth, progressing until they reach their maximum whiteness potential (Figure 5 and Figure 6). Although the average treatment requires 3 to 4 months of nightly bleaching with 10% carbamide peroxide, reaching the maximum whiteness could take as long as 15 months, depending on the initial color and location of the tetracycline staining. To ensure that the lightest shade possible is achieved, before terminating treatment, the patient should continue bleaching for an entire month despite no discernible color change.
The Importance of the Bleaching Examination
Prior to using any bleaching technique, a proper examination and analysis of the smile should be conducted.5 In addition to the smile itself, this evaluation should include the entire face and account for the sclera of the eyes. The examination should evaluate the extent of the teeth visible in the full smile because the gingival areas of teeth do not bleach as well as the incisal portion (Figure 7 and Figure 8). This difference in tooth response exists because the tooth is thicker at the gingiva—with less enamel and more dentin—and the composition of the dentin varies from the incisal to the root tip. With tetracycline-stained teeth, the gingival area is even more difficult to bleach. This is because the tooth color comes primarily from the dentin where the tetracycline molecule is more tightly bound (Figure 9 and Figure 10).
When planning to bleach normal teeth, identification of a “gummy smile” is important because whiter teeth will make it more noticeable. Periodontal therapy for altered passive eruption may be indicated prior to bleaching to expose more of the teeth and eliminate the excessive gingival display, which can reduce the need for bleaching. In order to appear normal, most teeth should be at least 10 mm in length. The presence of a gummy smile may be less of a problem when planning to bleach tetracycline-stained teeth. Because their gingival areas are often darker and do not respond as well, tetracycline-stained teeth will not create the same sharp contrast with the gingiva that is observed in a gummy smile with lighter teeth.
Existing restorations revealed in a full smile should be evaluated for visibility and criticality of color because they will not lighten with bleaching, regardless of the material. Some surface stain removal may occur on composites, but the basic color of the restorations will remain the same. Therefore, if a restoration matches the existing color of the tooth, it may become unesthetic after bleaching and need to be replaced. Patients must clearly understand the financial implications of replacing unesthetic, tooth-colored restorations for an additional fee, because that cost may present an obstacle to proceeding with bleaching.
Periodontal conditions and gingival recession should be evaluated during smile analysis because exposed roots do not bleach. Tetracycline-stained teeth are even darker at the gingival than normal teeth, so the expectations of these patients should be adjusted to a less-than-ideal outcome. In addition, the smile analysis for both types of teeth should include an evaluation for gingival disharmony because lightening teeth with bleaching will accentuate any less-than-ideal areas (Figure 11 and Figure 12).
As with normal teeth, tetracycline-stained teeth should be examined for existing decay. Because the decay process is halted during bleaching with 10% carbamide peroxide, small non-sensitive caries do not have to be restored prior to treatment.6 However, if a deep or sensitive lesion is detected, a protective restoration using a resin-modified glass ionomer (RMGI) can be placed prior to initiating bleaching. Because predicting the final bleaching color outcome is impossible, the shade selection for the RMGI must be estimated. After bleaching is complete, if the RMGI no longer matches or has the desired surface gloss, its surface can be removed, leaving the deeper portion as a base. Then a composite resin that matches the shade of the bleached teeth can be bonded to the RMGI and surrounding enamel to achieve better esthetics.
Sensitivity during Bleaching
All patients who are considering bleaching treatment should be evaluated for a history of sensitivity. Those with existing sensitivity issues are more likely to experience increased sensitivity during bleaching. Sensitivity during bleaching treatment results from the easy passage of the peroxide through the intact enamel and dentin and into the pulp, which occurs in 5 to 15 minutes.7 Bleaching sensitivity is a transient pulpitis resulting from this easy passage, and higher concentrations of bleaching materials will result in greater sensitivity.8In addition, sensitivity can be caused by occlusal problems related to the tray.
Beyond using a low concentration of carbamide peroxide and a proper-fitting tray, the addition of a 10 to 30 minute tray application of 5% potassium nitrate prior to or after bleaching treatment can help reduce associated sensitivity.9 Potassium nitrate is found in most desensitizing toothpastes in the United States, but is less used in other countries. With various professional products available for dispensing potassium nitrate, it can be applied by brushing with a toothpaste, using it in the tray instead of the bleaching material, or incorporating it into the bleaching material. When applied using the tray, a 5% potassium nitrate toothpaste worn for 10 to 30 minutes will successfully reduce sensitivity in more than 90% of bleaching patients. For patients with a history of sensitivity, brushing with a desensitizing toothpaste containing 5% potassium nitrate for 2 weeks prior to beginning bleaching treatment can significantly reduce bleaching sensitivity.10
Sensitivity generally occurs during the first 2 weeks of bleaching treatment. Although the tetracycline-stained patient will require bleaching treatment that lasts for many months, the sensitivity does not usually become progressively worse. To further reduce sensitivity issues, practitioners should wait 2 weeks after a prophylaxis before initiating bleaching, during which time the patient can brush with a desensitizing toothpaste.
Screening Radiographs
Before the initiation of bleaching treatment, a screening radiograph should be taken of the anterior teeth and any individual dark teeth to determine the cause of their discoloration. Oftentimes, an abscessed tooth or internal or external resorption fails to exhibit any associated signs or symptoms other than tooth discoloration. In tetracycline-stained teeth, these color variances are more difficult to recognize. Additionally, calcific metamorphosis, in which the pulp chamber is fully or partially obliterated by the deposition of secondary dentin, can cause a tooth to appear darker. The screening radiograph can also be useful in determining the presence of any interproximal caries or periapical cysts or tumors that could be contributing to the darkened appearance of a tooth or teeth.
Bleaching Materials
Different patient instructions are required for each of the two most used bleaching materials: carbamide peroxide and hydrogen peroxide. Carbamide peroxide is composed of hydrogen peroxide and urea. Because it is active for 6 to 10 hours, carbamide peroxide is best used in overnight tray bleaching.11 Hydrogen peroxide is only active for 30 to 60 minutes, so it is more appropriate for use during the day. A 10% carbamide peroxide product is comparable to a 3.5% hydrogen peroxide product. Because carbamide peroxide has a longer activity time, it requires less treatments to reach the same shade when compared with the use of hydrogen peroxide during the day. For normal teeth, patient preference and lifestyle can determine the most appropriate material and wear time. However, for tetracycline-stained teeth, overnight tray bleaching with 10% carbamide peroxide is the most time-efficient and results in a high rate of patient compliance.
A pH level below 6.8 is necessary for tooth decay to occur in dentin, and a pH level below 5.5 is necessary for it to occur in enamel. Because carbamide peroxide has urea, it elevates the pH level of the mouth above 8.0, significantly reducing patients' ability to develop tooth decay during bleaching treatments. Hydrogen peroxide has a pH of 5, so it does not favor tooth protection. Because of the long-term tray wear necessary in the treatment of tetracycline-stained teeth, the use of 10% carbamide peroxide overnight is recommended.
Tray Fabrication
The bleaching tray design selected should be informed by the product to be used, its concentration, and the specific needs of the patient. Products containing 10% carbamide peroxide were originally designed to treat the gingivae as an oral antiseptic (eg, Gly-Oxide®). Therefore, when planning to use a 10% carbamide peroxide bleaching material, trays can be made that extend 1 to 2 mm onto the gingival tissue for a more comfortable fit and a better seal to retain the material. The gingival health of patients involved in bleaching research projects using this concentration and tray type has been shown to improve during bleaching. The use of carbamide peroxide concentrations higher than 10% requires scalloping the tray so that the material does not come in contact with the gingival tissue and potentially cause a burn. For long-term wear to treat tetracycline-stained teeth, the non-scalloped tray with 10% carbamide peroxide provides the most efficient bleaching possible with the least gingival or tooth sensitivity.
In tray fabrication, spacers or reservoirs are not needed to bleach teeth; they are designed to account for tight or ill-fitting trays. A proper alginate impression (ie, the material is allowed to set properly in the tray before use, held in the patient's mouth for the appropriate manufacturer recommended time, and used to pour the model within 15 minutes to 45 minutes without bubbles) can be used to create an excellent fitting, non-scalloped, no-reservoir tray for 10% carbamide peroxide bleaching. Not using reservoirs both conserves the amount of bleaching material used and increases the comfort to the patient, especially during the extended bleaching times required to treat tetracycline-stained teeth.
Composite Bonding
Because bleaching materials penetrate the teeth and release oxygen, when bonding is performed immediately after bleaching, the bond strength between the composite and the enamel is reduced by approximately 50%.12 Therefore, dentists should avoid initiating bonding procedures until 2 weeks after bleaching treatment to give the oxygen time to dissipate. The oxygen released during bleaching also affects the shade of the teeth, so patients should not bleach for 2 weeks prior to a shade being taken for a crown or composite. Because some patients may be using over-the-counter products unknown to the dental office, it is important that the receptionist also question them about any of these products that they may be using before scheduling an appointment that involves bonding or shade matching.
Fees for Extended Bleaching Treatments
The non-specific and longer treatment times required to bleach tetracycline-stained teeth create a unique challenge for the practitioner concerning fees. If quoted a price for the full treatment time, the potential bleaching patient could be discouraged from proceeding with the treatment. One successful model involves charging a reasonable initial bleaching fee for the maxillary arch, while educating the patient that any subsequent treatment and material refill costs will depend on the length of the bleaching process. Nationally, one arch of at-home bleaching averages around $285, which includes the examination, the radiograph, the tray, and the bleaching material. A typical bleaching kit will treat one arch for roughly 4 weeks, and a form can be used by patients to keep track of how quickly they are using the syringes as well as to record any color changes or sensitivity. At the 1-month, follow-up appointment, this form can be used by the dentist to help determine a fair cost for the patient, including the amount of material needed for another month of treatment and an additional office visit. To further reduce the cost, the dentist should use a company that offers refills to sell to patients instead of full kits. Because they can potentially save money, patients tend to become very efficient with the bleaching material. This “pay as you go” approach benefits both the patient and the dentist. The patient can anticipate the monthly cost, and the dentist does not lose money from unanticipated extended treatment times. At each appointment, the dentist and patient can determine if another month of treatment is needed or if it is time to begin bleaching the mandibular arch.
Single Arch Treatment
The dental practice needs to offer a single arch fee. The best way to maximize patient compliance and compare progress from the baseline shade is to treat only one arch at a time. In addition, patients can become discouraged from initiating treatment by a higher fee that includes both arches and may have other reasons for wanting to bleach the maxillary arch only. In bleaching research projects performed free of charge to the participants, surprisingly, some patients choose not to bleach the mandibular arch—even at no cost. The option of wearing only one tray has several other advantages. Because there is only one thickness of material between the teeth, this approach minimizes the impact of tray bleaching on occlusion, which can reduce mechanical tooth sensitivity and eliminate potential joint problems. And because fewer teeth are being treated simultaneously, overall tooth sensitivity can be reduced as well. Single arch bleaching treatment is especially indicated in long-term treatment situations, such as for tetracycline staining, because patients can otherwise lose their reference for how dark their teeth were prior to treatment and become dissatisfied that limited progress is being made. Using a single arch approach provides many advantages, including reduced impact on occlusion, reduced overall sensitivity, and most importantly, increased patient compliance and satisfaction.
Patient Compliance
Maintaining patient compliance for 2 to 6 months of at-home bleaching treatment for tetracycline-stained teeth may seem daunting to the dentist. However, with careful treatment presentation and patient understanding of the benefits, it can be achieved. Much like adjusting to a bruxism splint or an anti-snoring device, with nightly wear, patients can become accustomed to the use of a whitening tray. The use of 10% carbamide peroxide for long-term wear is ideal for a number of reasons. Compliance is better with overnight tray use, and the patient gets the greatest benefit per application due to its longer activity time, which reduces cost. For example, when using carbamide peroxide, approximately 50% of the peroxide is depleted within 1 to 2 hours. However, the remaining material will continue to release peroxide for another 2 to 6 hours. Therefore, if the patient only bleached for 2 hours before removing the tray, he or she would be discarding half of the active material, greatly reducing the efficiency of the individual treatment and unnecessarily increasing the cost of the bleaching process.
Bleaching vs Veneers
Several factors should be considered when choosing whether or not to bleach tetracycline-stained teeth. Because the teeth lighten from the incisal to the gingival, the location of the darkened areas will have an impact on the final result. Teeth with the darkest discoloration at the cementoenamel junction, especially dark, blue-gray areas, have the poorest prognosis for full lightening, whereas teeth with discoloration in the incisal third have the best prognosis. Teeth that are not able to be fully bleached at the gingival third may be good candidates for porcelain veneers. Bleaching prior to veneer preparation can help determine if veneers are necessary, and when they are, it can better their esthetics by providing lighter teeth as a starting point. Alternatively, teeth can be bleached from the lingual after veneer placement (Figure 13 and Figure 14).13 In cases involving tetracycline-stained teeth, when bleaching treatment is attempted prior to recommending veneers, patients can be confident that the most conservative treatment plan was used to achieve the most esthetic result.
Conclusion
Although the tooth discoloring effects associated with tetracycline ingestion in children have been recognized in the medical field for some time, tooth staining associated with adult ingestion of tetracycline analogs (eg, minocycline for acne treatment) also has been reported.14 Because of the drug's continued use to treat acne and other infections, such as Rocky Mountain spotted fever, tetracycline staining will continue to occur. Due to its financially and clinically conservative approach, bleaching should be the initial treatment for patients with tetracycline-stained teeth.15 Whether the case involves tetracycline-stained teeth or normal teeth, the most effective, cost-efficient, and safest bleaching technique is at-home, overnight bleaching with 10% carbamide peroxide in a non-scalloped, no-reservoir tray.
About the Authors
Van B. Haywood, DMD
Professor
Dental College of Georgia
Augusta University
Augusta, Georgia
Rhoda J. Sword, DMD
Associate Professor
Dental College of Georgia
Augusta University
Augusta, Georgia
References
1. Christensen GJ. Bleaching teeth—which way is best? J Esthet Restor Dent. 2003;15(3):137-139.
2. Haywood VB. The “bottom line” on bleaching 2008. Inside Dent. 2008;4(2):82-89.
3. Haywood VB, Leonard RH, Dickinson GL. Efficacy of six months of nightguard vital bleaching of tetracycline-stained teeth. J Esthet Dent. 1997;9(1):13-19.
4. Mrazek B. “Don't bleach until you see the white of their eyes.” Compend Contin Educ Dent. 2004;25(6):472-476.
5. Haywood VB. Pre-bleaching examination vital for optimum whitening. Compend Contin Educ Dent. 2012;33(1):72-73.
6. Bentley CD, Leonard RH, Crawford JJ. Effect of whitening agents containing carbamide peroxide on cariogenic bacteria. J Esthet Dent. 2000;12(1):33-37.
7. Cooper JS, Bokmeyer TJ, Bowles WH. Penetration of the pulp chamber by carbamide peroxide bleaching agents. J Endod. 1992;18(7):315-317.
8. Matis BA, Wang Y, Jiang T, Eckert GJ. Extended at-home bleaching of tetracycline-stained teeth with concentrations of carbamide peroxide. Quintessence Int. 2002;
33(9):645-655.
9. Haywood VB, Caughman WF, Frazier KB, Myers ML. Tray delivery of potassium nitrate–fluoride to reduce bleaching sensitivity. Quintessence Int. 2001;32(2):105-109.
10. Browning WD. Haywood VB. Hughes N, Cordero R. Prebrushing with a potassium nitrate dentifrice to reduce tooth sensitivity during bleaching evaluated in a practice-based setting. Compend Contin Educ Dent. 2010;31(3):220-225.
11. Matis BA, Cochran MA, Eckert G. Review of the effectiveness of various tooth whitening systems. Oper Dent. 2009;34(2):230-235.
12. Da Silva Machado J, Candido MS, Sundfeld RH, De Alexandre RS, Cardoso JD, Sundefeld ML. The influence of time interval between bleaching and enamel bonding. J Esthet Restor Dent. 2007;19(2):111-118.
13. Haywood VB, Parker MH. Nightguard vital bleaching beneath existing porcelain veneers: a case report. Quintessence Int. 1999;30(11):743-747.
14. Cheek CC, Heymann HO. Dental and oral discolorations associated with minocycline and other tetracycline analogs. J Esthet Dent. 1999;11(1):43-48.
15. Leonard RH, Haywood VB, Caplan DJ, Tart ND. Nightguard vital bleaching of tetracycline-stained teeth 90 months post-treatment. J Esthet Restor Dent. 2003;
15(3):142-153.