Pulpotomy for Primary Teeth with Tricalcium Silicate Material
Bioactive material provides easy handling and shorter set time
The principal objective of any pulpal therapy is “to maintain the integrity and health of a tooth and its supporting tissues”.1 We strive to preserve the integrity of primary teeth for function, including speech and mastication, as well as to maintain arch length and space maintenance. In primary teeth, pulpal therapy includes vital pulp therapy (including the placement of a protective liner), indirect pulp capping, direct pulp capping, or pulpotomy.
Vital pulp therapy is indicated in cases of a reversible pulpitis. A reversible pulpitis can be defined as a pulp that is capable of healing. Alternatively, pulpal therapy can take the form of non-vital pulp therapy, or pulpectomy. Non-vital pulp therapy would be indicated in cases of irreversible pulpitis. Descriptions of the indications for these pulp therapies are presented in Table 1.2
Clinical Scenario
A hypothetical scenario can help illustrate the dentist’s clinical assessment when considering pulpal therapy. If a child presents with a large carious lesion extending into the pulp on a primary molar, one’s first assessment of the clinical situation may be to consider the child’s actual age and dental age. How much longer until the problematic tooth exfoliates? Is it necessary for the sake of function and space maintenance to restore the tooth, or will the tooth be lost in short order?
For sake of this hypothetical scenario, suppose that the tooth will not exfoliate for several years. One’s second evaluation assesses the size of the lesion. In particular, what remaining tooth structure will be present after excavation of the caries? Let us imagine here that the tooth is restorable and the lesion extends into the pulp.
Thirdly, you will determine if there are any symptoms associated with the lesion. If the tooth is symptomatic, are these signs of a reversible or irreversible pulpitis? In our hypothetical case, the patient is asymptomatic with signs of reversible pulpitis.
In this scenario, therefore, in which a restorable primary molar with a mechanical or carious exposure is asymptomatic and exhibits symptoms of a reversible pulpitis, there is an indication for pulp therapy, more specifically vital pulp therapy.3
Pulpotomies are the most common vital pulp therapy procedure to treat symptom-free carious exposures in primary molars.4 A pulpotomy is the amputation of the coronal pulp and subsequent surface treatment of the remaining radicular vital pulp tissue. The remaining pulp tissue is then treated with a medicament or technique to either devitalize the tooth, conserve the vitality of the tooth, or to promote regeneration of the remaining pulp tissue.5
Medicament Options
Over the years, many different medicaments, pulp dressings, and techniques have been used in pulp therapy procedures. Formocresol, ferric sulfate, calcium hydroxide, glutaraldehyde, mineral trioxide aggregate, laser therapy, and electrosurgery have all been used with varying degrees of success.1 A Cochrane review in 2003 reported no significant difference in superiority among three different medicaments and one technique, namely formocresol, ferric sulfate, zinc oxide eugenol, and electrosurgery.6
The most widely used medicament in North America and the United Kingdom is formocresol.7,8 It has been described as the gold standard of pulp therapy.9 However, there have been significant concerns for the past 25 years surrounding the use of formocresol as a pulpotomy medicament.10 Certainly there are some who present evidence that these concerns are unfounded.11 More recently, the World Health Organization classified formocresol as a known carcinogen.12 As the debate continues, it would behoove dental professionals to search out a reliable biocompatible medicament or technique for vital pulp therapy.
Mineral trioxide aggregate (MTA) has been documented extensively and is a viable option for a biocompatible material for pulp therapy.13-17 Portland cement (PC) has a similar composition and biologic effect to MTA, but needs further investigation before it can be used in dentistry.18,19 Biodentine® (Septodont, www.septodontusa.com) is a new option composed of tricalcium silicate that has been described as a dentin replacement material. It has properties similar to PC and MTA, in that it is bioactive and biocompatible.20,21 Biodentine has the potential to induce apposition of reactionary dentin by stimulating odontoblasts and reparative dentin by induction of cell differentiation.16,22 The pH of Biodentine is very high (pH = 12), giving it bacteriostatic properties. It also has the ability to form a good marginal seal. Finally, Biodentine handles with a creamy, rather than sandy, texture and it sets completely within 12 minutes.23
Clinical Protocol
The technique for a Biodentine pulpotomy is as follows:
1. Prepare tooth for full coverage if exfoliation is expected in more than 2 years or for intracoronal restoration if exfoliation is expected in less than 2 years.24
2. Remove all caries.
3. Access pulp chamber. Be sure to make a wide enough access to visualize the entire coronal pulp.
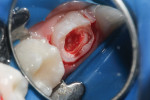
4. Assess health of the pulp tissue. A necrotic or hemorrhagic pulp is not a candidate for a vital pulp therapy procedure. In Figure 1, the tooth is prepared for stainless steel crown full coverage and wide access is made for the pulpotomy procedure.
5. Remove the coronal portion of the pulp with a sharp large spoon excavator.
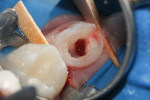
6. Control hemorrhage with pressure from a saline-soaked, sterile cotton pellet for 2 minutes (Figure 2).
7. Rinse chamber with additional saline.
8. Blot dry with a dry cotton pellet.
9. Mix Biodentine according to manufacturer’s directions.
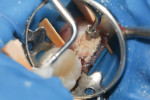
10. Fill chamber with Biodentine, packing gently. Biodentine has a paste-like consistency, similar to the consistency of plaster of Paris. The more that the material is packed, the wetter it will become. Avoid overworking (Figure 3).
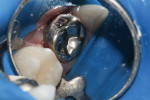
11. Full coverage and cementation can take place immediately. There is no need to wait for complete set to take place, as the material will continue to set after final restoration is cemented (Figure 4).
Final Thought
Biodentine is a clinically practical material for vital pulp therapy in primary molars. It is a very attractive material due to its bioactivity, ease of manipulation, and relatively fast setting time in comparison with other similar materials. It eliminates the need for a filling material for the pulp chamber. In relation to similar materials, it is much more cost effective. In summary, Biodentine pulpotomy is a simple technique for a reliable biocompatible vital pulp procedure.
Disclosure
The author received an honorarium from Septodont for this article.
References
1. American Academy of Pediatric Dentistry. Guideline on pulp therapy for primary and immature permanent teeth. Reference Manual. 2012/2013;
34(6):222-229.
2. Ounsi HF, Debaydo D, Salameh Z, et al. Endodontic considerations in pediatric dentistry: a clinical perspective. International Dentistry SA. 2009;(11)2:40-49.
3. Camp JH, Fuks AB. Pediatric endodontics: endodontic treatment for the primary and young permanent dentition. In: Cohen S, Hargreaves KM, eds. Pathways of the Pulp. 9th ed. St. Louis, MO: Mosby Elsevier; 2006:834-859.
4. Fuks, A. Vital pulp therapy with new materials for primary teeth: new directions and treatment perspectives. J Endod. 2008;34(suppl 7):S18-S24.
5. Ranly DM, Garcia-Godoy F. Current and potential pulp therapies for primary and young permanent teeth. J Dent. 2000;28(3):153-161.
6. Nadin G, Goel BR, Yeung CA, Glenny AM. Pulp treatment for extensive decay in primary teeth. Cochrane Database Syst Rev. 2003;(1):CD003220.
7. Hunter ML, Hunter B. Vital pulpotomy in the primary dentition: attitudes and practices of specialists in paediatric dentistry practising in the United Kingdom. Int J Paediatr Dent. 2003;13(4):246-250.
8. Primosch RE, Glomb TA, Jerrell RG. Primary tooth pulp therapy as taught in pediatric dental programs in the United States. Pediatr Dent. 1997;19(2):118-122.
9. King SR, McWhorter AG, Seale NS. Concentration of formocresol used by pediatric dentists in primary tooth pulpotomy. Pediatr Dent. 2002;24(2):157-159.
10. Casas MJ, Kenny DJ, Judd PL, Johnston DH. Do we still need formocresol in pediatric dentistry? J Can Dent Assoc. 2005;71(10):749-751.
11. Milnes AR. Is formocresol obsolete? A fresh look at the evidence concerning safety issues. J Endod. 2008;34(suppl 7):S40-S46.
12. IARC classifies formaldehyde as carcinogenic to humans [press release]. Lyon, France: International Agency for Research on Cancer. June 15, 2004. www.iarc.fr/en/media-centre/pr/2004/pr153.html. Accessed May 25, 2013.
13. Caicedo R, Abbott PV, Alongi DJ, Alarcon MY. Clinical, radiographic and histological analysis of effects of mineral trioxide aggregate used in direct pulp capping and pulpotomies of primary teeth. Aust Dent J. 2006;51(4):297-305.
14. Erdem AP, Guven Y, Balli B, et al. Success rates of mineral trioxide aggregate, ferric sulfate, and formocresol pulpotomies: a 24-month study. Pediatr Dent. 2011;33(2):165-170.
15. Ansari G, Ranjpour M. Mineral trioxide aggregate and formocresol pulpotomy of primary teeth: a 2-year follow-up. Int Endod J. 2010;43(5):413-418.
16. Doyle TL, Casas MJ, Kenny DJ, Judd PL. Mineral trioxide aggregate produces superior outcomes in vital primary molar pulpotomy. Pediatr Dent. 2010;32(1):41-47.
17. Godhi B, Sood PB, Sharma A. Effects of mineral trioxide aggregate and formocresol on vital pulp after pulpotomy of primary molars: an in vivo study. Contemp Clin Dent. 2011;2(4):296-301.
18. Shayegan A, Jurysta C, Atash R, et al. Biodentine used as a pulp-capping agent in primary pig teeth. Pediatr Dent. 2012;34(7):e202-e208.
19. Sakai V, Moretti A, Oliviera T, et al. Pulpotomy of human primary molars with MTA and Portland cement: a randomised controlled trial. Br Dent J. 2009;207(3):E5.
20. Saidon J, He J, Zhu Q, et al. Cell and tissue reactions to mineral trioxide aggregate and Portland cement. Oral Surg Oral Med Oral Pathol Radil Endod. 2003;95(4):483-489.
21. Laurent P, Camps J, De Meo M, et al Induction of specific cell responses to a Ca3SiO5-based posterior restorative material. Dent Mater. 2008;24(11):1486-1494.
22. Goldberg M, Pradelle-Plasse N, Tran XV, et al. Emerging trends in (bio) material research. In: Goldberg M, ed. Biocompatability or cytotoxic effects of dental composites. Chipping Norton, UK: Coxmoor Publishing Co;2008.
23. Bachoo IK, Seymour D, Brunton P. A biocompatible and bioactive replacement for dentine: is this a reality? The properties and uses of a novel calcium-based cement. Br Dent J. 2013;214(2):E5.
24. Holan G, Fuks AB, Keltz N, Success rate of formocresol pulpotomy in primary molars restored with stainless steel crown vs amalgam. Pediatr Dent. 2002;24(3):212-216.
About the Author
Carla Cohn, DMD
Private Practice
Winnipeg, Manitoba, Canada