Treating Peri-Implant Mucositis in the General Practice
Oral irrigators provide an excellent home maintenance option
The diagnosis, treatment, and prevention of peri-implant diseases are essential skills for the modern dental team. It is estimated that as many as 80% of patients restored with implants have peri-implant mucositis and between 28% and 35% have peri-implantitis.1 Because bacteria play a primary role in the pathogenesis of peri-implant conditions, the ability of the patient and the dental professional to remove plaque from the implant area can help reduce the incidence of these diseases.
Case Presentation
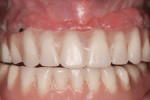
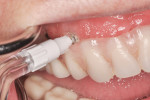
A 53-year-old woman presented for a comprehensive initial examination. Her chief complaint was that her “upper denture sometimes hurts” when eating. A review of her dental history revealed that she received a maxillary “hybrid” prosthesis delivered on five implants approximately 3 years ago. The restoration had been planned for six implants, but one had failed prior to final insertion. The patient’s lower dentition had been replaced with an overdenture on two implants and was stable. Clinical examination revealed moderate plaque accumulation and erythematous, swollen soft tissue (Figure 1). Radiographs showed no clear sign of bone loss around the implants; thus the preliminary diagnosis was generalized peri-implant mucositis of the maxillary implants.
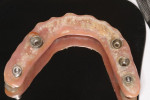
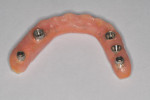
The diagnosis was confirmed upon prosthesis removal. Peri-implant tissues demonstrated bleeding on probing and plaque was found in the peri-implant sulcus. The implants were non-mobile and there was no evidence of purulence. There was a severe accumulation of plaque on the intaglio surface of the prosthesis (Figure 2). The patient explained that her former dental team had failed to educate her about at-home or in-office hygiene procedures. She had never received professional maintenance nor had the prosthesis been removed since it had been inserted approximately 3 years prior.
The treatment plan included mechanical debridement of the maxillary implants using a graphite implant scaler (Premier® Implant Scaler, Premier Products Co., www.premusa.com), followed by local delivery of antibiotics (Arestin® [minocycline HCl], OraPharma, Inc., www.orapharma.com). The prosthesis was brushed to remove the bulk plaque and debris. Finally, the prosthesis was placed in a cleansing solution designed for dentures and set in an ultrasonic device for 10 minutes.
With the peri-implant mucositis managed, the remainder of the appointment was dedicated to patient education and oral hygiene instruction. First, the patient was told that she will require regular hygiene appointments even though she no longer has her natural dentition. A 6-month recall system was instituted with anticipated prosthesis removal every 2 years, depending on clinical presentation. Second, the patient was given a hand mirror and shown proper home care techniques. She was given an oral irrigator (Waterpik® Ultra Water Flosser, Water Pik, Inc., www.waterpik.com) and given a strict protocol to follow.
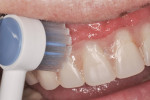
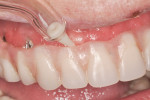
The Waterpik Ultra Water Flosser comes with six tips with individual designs. The Toothbrush Tip was recommended for initial debridement of the surfaces of the prosthesis (Figure 3). The combined action of brush bristles with water irrigation is effective in removing bulk debris. The Pik Pocket™ Tip is flexible and therefore well suited for negotiating around prosthetic flanges (Figure 4). This tip will be able to deliver water and antimicrobial rinse to the tissue in contact with the intaglio surface of the prosthesis. Finally, the Orthodontic Tip is demonstrated around the few areas where metal abutments are exposed to the oral environment (Figure 5).
The patient was appointed for a 2-week follow-up to monitor the tissue response and her home care. The tissue appeared healthy and there was no bleeding on probing. The prosthesis was found to be free from plaque and calculus (Figure 6) and reinserted.
Discussion
Although the treatment of peri-implantitis remains a challenge, peri-implant mucositis can be effectively managed using nonsurgical methods.2 Once the disease is managed, the team must reinforce proper home care.
The design of the prosthesis plays a role in the accumulation of plaque and calculus. A prosthetic flange can block the access of hygiene instruments for both the patient and the dental team.3 Certain prostheses may require removal for adequate maintenance. Based upon the patient’s dental history, the design of the prosthesis, and the patient’s home care, the dental team can design an appropriate recall schedule that includes periodic removal of the prosthesis. This schedule can be modified if the patient fails to follow a strict home care regimen.
Individual patient circumstances should be taken into account when designing a plan for home care. Traditional brushing, rinses, and flossing may be difficult for certain implant prosthetic designs. Only one oral irrigator (Waterpik Water Flosser) has been studied and shown to be superior to regular floss in reducing peri-implant inflammation.4-6
References
1. Ziztmann NU, Berglundh T. Definition and prevalence of peri-implant diseases . J Clin Periodontol. 2008;35(suppl 8):S286-S291.
2. Renvert S, Roos-Jansaker AM, Claffey N. Non-surgical treatment of peri-implant mucositis and peri-implantitis: a literature review . J Clin Periodontol. 2008; 35(suppl 8):S305-S315.
3. Salierno C. The implant dilemma: restorative contours sometimes challenge hygienists . RDH Magazine. 2012;32(6):22-25.
4. Lyle DM. Relevance of the water flosser: 50 years of data . Compend Contin Educ Dent. 2012;33(4) 278-282.
5. Felo A, Shibly O, Ciancio SG, et al. Effects of subgingival chlorhexidine irrigation on peri-implant maintenance . Am J Dent. 1997;10(2):107-110.
6. Magnuson B, Harsono M, Silberstein J, et al. Water flosser vs. floss: comparing reduction in bleeding around implants [abstract 3761]. Presented at: IADR 2013; March 23, 2013; Seattle, Washington.
Chris Salierno, DDS
Private Practice
Melville, New York