A Clinical Therapy for the Treatment of Peri-Implantitis
Reversing the peri-implantitis process can help salvage problem implants
The long-term success and predictability of dental implants has been well documented in the dental literature for more than 40 years.1-3 As their use continues to become more common in contemporary restorative and surgical dental practices, the choice of dental implants in treatment plans now ranges from single tooth replacement to full arch/full mouth reconstruction.4-6
Considerations for Implant Success
Ancillary procedures such as bone grafting, soft tissue replacement, and more advanced surgical techniques have become necessary to restore the proper peri-implant environment, which must consist of an acceptable amount of attached keratinized gingival tissue to protect the alveolar structures and the implant/abutment connection.7-15
Many factors can affect the short- and long-term success of dental implants. A patient’s health status, pre-existing dental infections, and social habits can all play a significant role in the success of dental implants for natural tooth replacement. Patient selection and evaluation, proper pretreatment planning, and effective communication among all members of the implant team (surgeon, restorative dentist, and laboratory technician) also are positive contributors to success. Additionally, success is also dependent to some extent on the proficiency of the surgeon (whether dental specialist or restorative clinician) and the restorative practitioner.
Implant design is also an important factor in success. Surface alterations16 can enhance the integration process and the soft tissue seal at the coronal aspect of the implant and reduce bacterial adherence, whereas abutment designs can minimize their removal, preventing repeated trauma to the hemi-desmisomal seal formed between the implant/abutment complex and surrounding soft tissues. Other implant designs are best for incorporation into immediate restoration and/or loading protocols. Making the right choice for an individual clinical scenario can contribute to more rapid case completion and continued success with dental implants for tooth replacement.
Confronting Peri-Implantitis
Despite the high success rates observed with dental implants in the contemporary dental practice, complications and implant loss can occur. An important complication to dental implant treatment is a pathologic process called peri-implantitis.
Peri-implantitis is a condition caused by peri-implant tissue changes that can have flora present similar to that of periodontitis, causing a mild to aggressive inflammation mimicking that found in cases of mild-moderate to advanced periodontitis.17 The disease state is generally thought to be caused primarily by plaque and biofilm, which has been shown to accumulate on implant surfaces as well as teeth.17,18 Tissue inflammation, bleeding upon probing, purulence, and advanced bone loss is often observed around a dental implant affected with peri-implantitis.19,20
Peri-implantitis has been linked to cases with implant loss.21,22 Looking at survival rates of implant-supported single crowns, Jung and colleagues23 observed that peri-implantitis and soft tissue complications were seen in approximately 10% of cases reviewed.
Additional factors that can contribute to peri-implantitis include a lack of hygiene, which can increase inflammation and bone loss levels,24-26 excusive excessive occlusal load,27 and, as previously mentioned, inadequate zones of attached keratinized tissue.28 The effects of tobacco use on implant surfaces have also been shown to negatively impact implant success rates. Baig and Rajan29 found that in implant patients who smoke, significantly greater marginal bone loss occurs after implant placement and the occurrence of peri-implantitis increases.
Genetic factors have been correlated with occurrence of peri-implantitis. Studies have shown a direct linkage between the IL-1 gene polymorphism and peri-implantitis.30 Plagnat and colleagues31 demonstrated that a similarity existed in the inflammatory processes of tissue surrounding implants and natural teeth, and that elastase and alkaline phosphatase activity could be important markers for bone loss around dental implants.
Corrosion can also play a role in peri-implantitis, particularly if a base metal allergy is observed in direct connection to the titanium implant. This could directly contribute to the deconstruction of osseous tissue, resulting in peri-implantitis.32
Poor surgical technique can also be a cause of peri-implantitis. Bone overheating during the implant site preparation process can lead to bone necrosis and implant surface contamination during implant placement can lead to the development of an apical lesion; these can contribute to the loss of bone as well as inflammation of the peri-implant tissues.33
As implant procedures become more prominent in treatment plans and implant patients present for routine maintenance visits, dental practitioners will most likely be confronted with the challenge of treating peri-implantitis cases. The following case reports outline a protocol to treat peri-implantitis (Table 1) and reverse advanced bone loss around dental implants in occlusal function for greater than 5 years.
Case 1
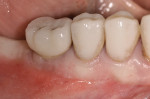
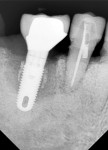
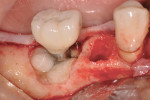
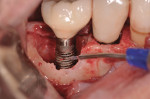
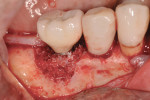
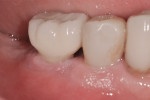
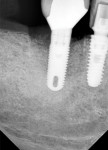
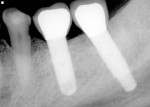
A 64-year-old nonsmoking man presented for treatment of a failing implant at the mandibular right first molar (Figure 1 and Figure 2). The patient had a 4.7-mm x 13-mm Tapered Screw-Vent® titanium plasma-sprayed surface implant (Zimmer Dental, www.zimmerdental.com) placed 8 years prior. Probing depths in excess of 10 mm circumferentially were noted, with bleeding on probing and purulence present. No mobility to the implant structure was present.
The mandibular right second premolar was diagnosed with a guarded-hopeless prognosis, and was slated for removal and replacement with a dental implant by the immediate extraction, placement, and restoration protocol. The patient had wished to keep the implant at the No. 30 position; therefore, the decision was made to attempt repair at the No. 30 site simultaneously with the immediate restoration procedure at tooth No. 29. The patient was given a prescription for 20 tablets of Augmentin (amoxicillin clavulanate) 875 mg to be taken every 12 hours to start the day prior to the surgical therapy.
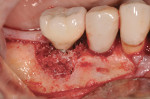
After administration of an appropriate local anesthetic, an intrasulcular incision was made from the distal of the first molar area to the mesial line angle of the mandibular right canine. A full thickness muco-periosteal flap was elevated, and the granulation tissue surrounding the affected implant was removed (Figure 3). The exposed contaminated implant surface, including each exposed thread, was then debrided with a piezoelectric scaler with water irrigation. Mechanical debridement and cleaning of the contaminated surface was achieved. Tooth removal and debridement of the second molar site was then accomplished (Figure 4).
Following tooth removal, citric acid (pH 1) was applied to the exposed implant surface. Initially, the surface was burnished and rinsed with sterile water and then citric acid–saturated cotton pellets were placed around the exposed implant surface for a total of 3 minutes (Figure 5), followed again by rinsing with sterile water. Figure 6 shows the appearance of the exposed implant surface after antiseptic chemical treatment using citric acid.
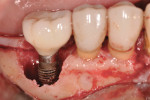
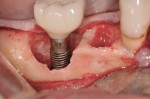
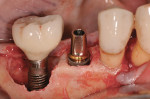
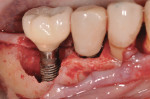
A 4.0-mm x 11.5-mm ETIII implant (Hiossen, www.hiossen.com) was then placed in the No. 29 position to appropriate depth, followed by the placement of a stock titanium abutment (Figure 7) and completion of the immediate provisional restoration (Figure 8). Decontamination of bacteria and inflammatory debris on the implant surface was further treated by surface contact with a neodymium-yttrium-aluminum garnet (Nd:Yag) laser at the 5-watt power setting. The laser beam was in contact with the exposed implant surface, including all exposed threads (Figure 9). Figure 10 shows the appearance of the implant surface post-laser treatment.
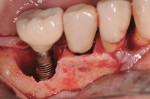
A platelet-rich plasma (PRP) gel was then applied to the implant surface (Figure 11) prior to placement of a PRP/grafting complex into the peri-implant defect (Figure 12), followed by application of a PRP membrane over the grafted site (Figure 13). A continuous sling suturing technique with 5-0 MONOCRYL™ Sutures (Ethicon Inc., www.ethicon360.com) was employed to approximate and close the surgical site (Figure 14). The patient was instructed to finish the antibiotic administered presurgically, and prescribed chlorhexidine rinse 2 times per day for 10 days postsurgically.
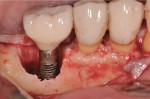
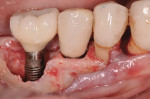
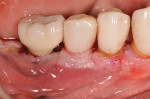
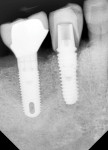
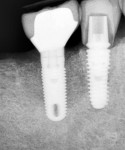
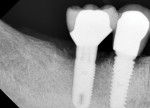
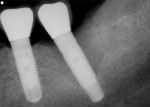
Figure 15 shows the immediate post-operative digital periapical radiograph of the No. 29 and No. 30 sites. A 1-month postoperative digital periapical radiograph can be seen in Figure 16. Figure 17 and Figure 18 show the 2-month post-repair clinical view and periapical radiograph, respectively. Note the post-repair maintenance and band of keratinized attached tissue. A 1-year post-repair periapical radiograph can be seen in Figure 19; bone fill has occurred in the peri-implant structures. Probing depths were reduced to 3 mm or less circumferentially, and the absence of purulence and bleeding upon probing was observed at the 1-year post-repair followup.
Case 2
A 66-year-old nonsmoking man presented for treatment of a large peri-implant defect at the mandibular left second molar site (Figure 20). A 4.7-mm x 15-mm coated Tapered Screw-Vent HA implant was had been placed 7 years prior following immediate extraction of the natural tooth and subsequent peri-implant bone grafting of the natural tooth space. At the current evaluation appointment, 7 years prior to the evaluation appointment, probing depths in excess of 12 mm were noted circumferentially, in addition to purulence observed with palpation of the facial gingival tissues. The implant complex did not exhibit mobility.
The patient preferred to attempt repair of the peri-implant structures over removing and replacing the implant. Following the procedure outlined in Table 1, the repair process was completed. Figure 21 shows the 1-year post repair digital periapical radiograph of the implant in the mandibular left second molar site. Comparing Figure 20 and Figure 21, it is observed that bone fill has occurred following the repair process. At the 1-year post repair evaluation, no bleeding upon probing or purulence was noted. Additionally, probing depths were reduced to 3 mm circumferentially, with the exception of the reading at the distal-buccal of the molar, which registered 5 mm.
Discussion
Bone and tissue loss around dental implants over their life span, placed into occlusal function, will be a problem with which restorative and surgical clinicians will be faced in recall and maintenance programs. As previously outlined in the text of this article, the causes of peri-implantitis can vary from poor surgical technique to genetic predisposition of the host response to periodontal pathogens similar to periodontitis. To achieve resolution of any type of dental infection, the etiologic cause of the problem must first be identified and corrected before any regenerative/reconstructive procedures are attempted. Otherwise, the breakdown of the peri-implant structures would likely recur.
A technique for the treatment of peri-implantitis and subsequent loss of the peri-implant structures has been presented above. The technique, when strictly adhered to, has been shown to replace deep circumferential–type peri-implant defects in more than 20 treated sites, with observation of the successful replacement of bone and soft tissues for more than 1.5 years post-treatment. The author recommends additional clinical trials to substantiate the surgical technique outlined and the clinical findings presented in the case reports.
References
1. Adell R, Lekholm U, Rockier B, Brånemark PI. A 15-year study of osseointegrated implants in the treatment of the edentulous jaw. Int J Oral Surg. 1981;10(6):387-416.
2. Adell R, Eriksson B, Lekholm U, et al. Long-term follow-up study of osseointegrated implants in the treatment of totally edentulous jaws. Int J Oral Maxillofoc Implants. 1990;5(4):347-359.
3. Babbush CA. Dental Implants: The Art and Science. 2nd ed. Philadelphia, PA: WB. Saunders Co; 2001:201-216.
4. Wörhle PS. Single-tooth replacement in the aesthetic zone with immediate provisionalization: fourteen consecutive case reports. Pract Periodontics Aesthet Dent. 1998;10(9):1107-1114.
5. Petrungaro PS. Immediate implant placement and provisionalization in edentulous, extraction, and sinus grafted sites. Compend Contin Educ Dent. 2003;24(2):95-113.
6. Petrungaro PS. Implant placement and provisionalization in extraction, edentulous, and sinus grafted sites: a clinical report on 1,500 sites. Compend Contin Educ Dent. 2005;26(12):879-890.
7. Jensen J, Sinder-Pedersen S. Autogenous mandibular bone grafts and osseointegrated implants for reconstruction of the severely atrophied maxilla: a preliminary report. J Oral Maxillofoc Surg. 1991;49(12):1277-1287.
8. Misch CM, Misch CE, Resnik R, Ismail YH. Reconstruction of maxillary alveolar defects with mandibular symphysis grafts for dental implants: a preliminary procedural report. Int J Oral Maxillofoc Implants. 1992;7(3):360-366.
9. Jensen J, Sinder-Pedersen S, Oliver AJ. Varying treatment strategies for reconstruction of maxillary atrophy with implants: results in 98 patients. J Oral Maxillofac Surg. 1994;52(3):210-218.
10. Misch CM, Misch CE. The repair of localized severe ridge defects for implant placement using mandibular bone grafts. Implant Dent. 1995;4(4):261-267.
11. Krauser JT, Rohrer MD, Wallace SS. Human histologic and histomorphometric analysis comparing Osteograf/N with Pepgen P-15 in the maxillary sinus elevation procedure: a case report. Implant Dentistry. 2000;9(4):298-302.
12. Smiler DG. Comparison of an organic bovine mineral with and without synthetic peptide in a sinus elevation: a case study. Implant Dentistry. 2001;10(2):139-142.
13. Mellonig JT, Bowers GM, Bailey RC. Comparison of bone graft materials. Part I. New bone formation with autografts and allografts determined by Srromiurn-85. J Periodontal. 1981;52(6):291-296.
14. Sottosami JS. Aesthetic extractions with calcium sulfate and the principles of guided tissue regeneration. Pract Periodontics Aesthet Dent. 1993;5(5):61-69.
15. Petrungaro P. Platelet-rich plasma for dental implant s and soft tissue grafting. Interview by Arun K. Garg. Dent Implantol Update. 2001;12(6):41-46.
16. Grössner-Schreiber B, Griepentrog M, Haustein I, et al. Plaque formation on surface modified dental implants: an in vitro study. Clin Oral Implants Res. 2001;12(6):543-551.
17. Cortez AR, Ferraz P, Tosta M. Influence of etiologic factors in peri-implantitis: literature review and case report. J Oral Implantol. 2012;38(5):633-637.
18. Heuer W, Elter C, Demling A, et al. Analysis of early biofilm formation on oral implants in man. J Oral Rehabil. 2007;34(5):377-382.
19. George K, Zafiropolous GG, Murat Y, et al. Clinical and microbiological status of osseointegrated implants. J Periodontol. 1994;65(8):766-770.
20. Montes CC, Pereira FA, Thomé G, et al. Failing factors associated with osseointegrated dental implant loss. Implant Dent. 2007;16(4):404-412.
21. Mombelli A, Lang N. The diagnosis and treatment of peri-implantitis. Periodontology. 2000;17(1):63-76.
22. van Winkelhoff AJ, Wolf JW. Actinobacillus actinomycetemcomitans-associated peri-implantitis in an edentulous patient. A case report. J Clin Periodontol. 2000;27(7):531-535.
23. Jung RE, Pjertursson BE, Glauser R, et al. A systematic review of the 5-year survival and complication rates of implant supported single crowns. Clin Oral Implants Res. 2008;19(2):119-130.
24. Lindquist LW, Rockler B, Carlsson GE. Bone resorption around fixtures in edentulous patients treated with mandibular fixed tissue-integrated prostheses. J Prosthet Dent. 1988;59(1):59-63.
25. Sanz M, Alandez J, Lazaro P, et al. Microbial differences in 2 clinically distinct types of failures of osseointegrated implants. Clin Oral Implant Res. 1991;2(3):128-134.
26. Becker W, Becker BE, Newman MG, Nyman S. Clinical and microbiologic findings that may contribute to dental implant failure. Int J Oral Maxillofac Implants. 1990;5(1):31-38.
27. Kozlovsky A, Tal H, Laufer BZ, et al. Impact of implant overloading on the peri-implant bone in inflamed and noninflamed peri-implant mucosa. Clin Oral Implant Res. 2007;18(5):601-610.
28. Apse P, Ellen RP, Overall CM, Zarb GA. Microbiota and crevicular fluid collagenase activity in the osseointegrated dental implant sulcus: a comparison of sites in edentulous and partially edentulous patients. J Periodontal Res. 1989;24(2):96-105.
29. Baig MR, Rajan M. Effects of smoking on the outcome of implant treatment: a literature review. Indian J Dent Res. 2007;18(4):190-195.
30. Bormann KH, Stühmer C, Z’Graggen M, et al. IL-1 polymorphism and periimplantitis. Scheiz Monatsschr Zahnmed. 2010;120(6):510-520.
31. Plagnat D, Giannopoulou C, Carrel A, et al. Elastase, alpha2-macroglobulin and alkaline phosphatase in crevicular fluid from implants with or without periimplantitis. Clin Oral Implants Res. 2002;13(3):227-233.
32. Tengvall P, Elwing H, Sjöqvist L, et al. Interaction between hydrogen peroxide and titanium: a possible role in the biocompatibility of titanium. Biomaterials. 1989;10(2):118-120.
33. Quirynen M, Gijbels F, Jacobs R. An infected jawbone site compromising successful osseointegration. Periodontol 2000. 2003;33(1):129-144.
34. Mouhyi J, Dohan Ehrenfest DM, Albrektsson T. The peri-implantitis: implant surfaces, microstructure, and physiochemical aspects. Clin Implant Dent Relat Res. 2012;14(2):170-183.
35. Petrungaro, P. Treatment of the infected implant site using platelet-rich plasma. Compend Contin Educ Dent. 2002;23(4):363-366, 368.
36. Schwarz F, Sahm N, Iglhaut G, Becker J. Impact of the method of surface debridement and decontamination on the clinical outcome following combined surgical therapy of peri-implantitis: a randomized controlled clinical study. J Clin Periodontol. 2011;38(3):276-284.
37. Renvert S, Lessem J, Dahlen G, et al. Topical minocycline microspheres versus topical chlorhexidine gel as an adjunct to mechanical debridement of incipient peri-implant infections: a randomized clinical trial. J Clin Periodontol. 2006;33(5):362-369.
38. Leonhardt A, Dahlén G, Renvert S. Five-year clinical, microbiological, and radiological outcome following treatment of peri-implantitis in man. J Periodontol. 2003;74(10)1415-1422.
39. Wilson, TG Jr. The positive relationship between excess cement and peri-implant disease: a prospective clinical endoscopic study. J Periodontol. 2009;80(9):1388-1392.
40. Chen S, Darby I. Dental implants: maintenance, care and treatment of peri-implant infection. Aust Dent J. 2003;48(4)212-220.
41. Yamamoto A, Tanabe T. Treatment of peri-implantitis around TiUnite-surface implants using Er:YAG laser microexplosions. Int J Periodontics Restorative Dent. 2013;33(1):21-30.
42. Froum SJ, Froum SH, Rosen PS. Successful management of peri-implantitis with a regenerative approach: a consecutive series of 51 treated implants with 3- to 7.5-year follow-up. Int J Periodontics Restorative Dent. 2012;32(1):11-20.
43. Klinge B, Gustafsson A, Berglundh T. A systematic review of the effect of anti-infective therapy in the treatment of peri-implantitis. J Clinical Periodontol. 2002;29(suppl 3):S213-S215; discussion S232-S233.
44. Claffey N, Clarke E, Polyzois I, Renvert S. Surgical treatment of peri-implantitis. J Clin Periodontol. 2008;35(suppl 8):S316-S332.
About the Author
Paul S. Petrungaro DDS, MS
Founder, Implant Learning Center
Private Practice Periodontics and Implantology
Chicago, Illinois